5.4: Phase Changes
- Page ID
- 213242
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define phase change.
- Define melting, freezing, vaporization, condensation, sublimation, and deposition.
- Define complementary phase change.
- Identify complementary phase change pairs.
- Define equilibrium.
The previous section of this text discussed the solid, \(\left( s \right)\), liquid, \(\left( l \right)\), and gaseous, \(\left( g \right)\), states of matter, which are all used to describe the physical form of a single chemical. The following paragraphs will define and describe the six phase changes, or changes in state, through which these physical forms of matter can interconvert.
Phase Changes
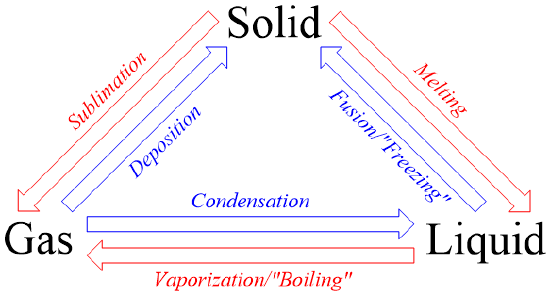
Melting is defined as the process of converting a substance from the solid to the liquid state of matter. A chemical that exists in its liquid state can be changed back into a solid state in a process known as fusion, which is more commonly-known as "freezing." Melting and fusion are complementary phase changes, because both relate the same states of matter.
During condensation, a substance is changed from the gaseous to the liquid state of matter. Vaporization, which is more often referred to as "boiling," is the complementary process in which a chemical is converted from the liquid state of matter to a gaseous physical form. Many people erroneously label this transformation as "evaporation." As stated in the previous section, the primary difference between liquids and gases is the physical proximity of their constituent particles at the molecular level. While the particles in a liquid must remain in direct contact with one another, gaseous atoms or molecules exist independently. Therefore, in order to change a substance from the liquid to the gaseous state of matter, heat must be applied to overcome the attractive forces between the liquid's constituent particles. As single particles separate from others within a liquid, the resultant gas particles escape from the liquid in the form of bubbles, as shown below in the first image in Figure \(\PageIndex{1}\). In contrast, evaporation is a passive transformation, meaning that heat is not required to either initiate or sustain its corresponding processes. As a result, no bubbles are present when a substance evaporates, as shown in the second image in Figure \(\PageIndex{1}\). Because of these subtle, yet significant, differences, "evaporation" is not synonymous with "vaporization," and, therefore, these terms should not be used interchangeably.
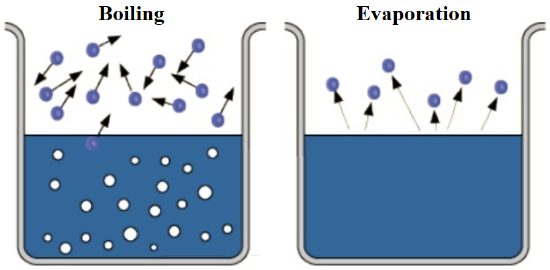
Finally, sublimation is the process in which a solid is change to a gas. Its complement, deposition, is defined as a conversion from the gaseous state of matter to the solid state of matter. While these complementary transformations are the least common among the six phase changes, both can be exemplified by analyzing the transformations of carbon dioxide and water. Solid carbon dioxide, which is commonly-known as "dry ice," does not melt at room temperature, but instead generates a white, "smoky" vapor by subliming directly into its gaseous state. Ice cubes that are left in a freezer for a prolonged period of time also sublime, causing the size of the ice cubes to shrink as solid particles are converted into a gaseous state. Finally, the "freezer burn" that forms on food results from the deposition of water vapor that cannot condense and, therefore, transforms directly into the solid state of matter.
Figure \(\PageIndex{2}\) summarizes the information in the previous paragraphs. This image illustrates which states of matter are converted through each phase change process and highlights the complementary nature of these transformations.
Phase Change Temperatures
Each phase change that is defined above occurs at a singular temperature. For example, the temperature at which a substance freezes is referred to as its freezing point, and the boiling point of a substance corresponds to the temperature at which that substance vaporizes. These phase change temperatures are physical properties, meaning that they are defined based on the identity of the substance that is being considered. The temperature scales that were defined in Section 1.5 were established based on the freezing and boiling points of water, which are summarized below in Table \(\PageIndex{1}\).
| Temperature Scale | Freezing Point | Boiling Point |
|---|---|---|
| degrees Fahrenheit | 32 °F | 212 °F |
| degrees Celsius | 0 °C | 100 °C |
| Kelvin | 273.15 K | 373.15 K |
A substance is present at a temperature below its freezing point will exist exclusively in the solid state of matter. If the temperature exceeds the boiling point of a substance, that chemical will be present only in the gaseous state of matter. Finally, a substance will exist only as a liquid in between these temperatures.
Each phase change process must, by definition, involve two states of matter. For example, when a substance freezes, it exists primarily in the liquid state of matter and begins to transform into the solid state of matter. As the freezing process continues, the relative amount of liquid that is present decreases in proportion to the quantity of solid that is generated. As stated above, a phase change occurs at a singular temperature. Because a state of matter conversion can only transpire at its associated phase change temperature, the temperature of a substance is unable to change while that chemical is transforming from one state of matter to another. As a result, a phase change is classified as an isothermal, or "constant temperature," process, in which two states of matter simultaneously exist at the singular temperature associated with the transformation.
Finally, as indicated above, the six phase changes can be organized into three complementary pairs by considering which states of matter are being interchanged. For example, melting and freezing are complements, because each involve transformations between the solid and liquid states of matter. If both of these states of matter exist at the freezing point of a substance, both freezing, and its complementary process, melting, can transpire at this temperature. This simultaneous occurrence of two opposing processes is known as equilibrium.
Therefore, based on the information in the previous paragraphs, vaporization and its complement, condensation, must also occur at a singular temperature, at which a chemical will exist in both the gaseous and liquid states of matter. Finally, both the solid and gaseous states of matter will be present upon establishment of a sublimation/deposition equilibrium.

