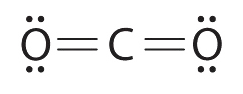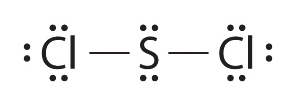4.3 Lewis Dot Structures
- Page ID
- 218256
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Skills to Develop
- To use Lewis dot symbols to explain the stoichiometry of a compound
Using Lewis Dot Symbols to Describe Covalent Bonding
It is usually easier to figure out a problem if you can draw a picture, either mental or real, of what is happening. This is often done in physics and mathematics, and it is especially helpful when looking at the bonding, structure, physical properties, and reactivity of compounds.
The most common picture, or model, of elements and compounds used is the Lewis Dot Structure. These pictures show you the type(s) of atom(s) involved, their position in the molecule, and where their valence electrons are situated.
To draw structures of substances you need to know:
1. the electronegativities of the elements so that you can decide if the atoms form ionic bonds or covalent bonds.
2. the number of valence electrons so that you can determine what ions will likely form (if the bonding will be ionic) or how many electrons will need to be shared in bonds (if the bonding will be covalent.)
GENERAL TERMS FOR LEWIS DOT STRUCTURES:
1. ![]() Dot • one dot represents one valence electron (found on odd-electron particles)
Dot • one dot represents one valence electron (found on odd-electron particles)
2. ![]() Pair of Dots •• a pair of dots represents a nonbonding (lone) pair of electrons that are not involved in a covalent bond and "belong to" only one atom
Pair of Dots •• a pair of dots represents a nonbonding (lone) pair of electrons that are not involved in a covalent bond and "belong to" only one atom
3. Dash each dash represents two electrons that are shared between two atoms as a covalent bond.
One dash — a single bond; one set of two electrons shared; also a sigma (σ) bond
Two dashes = a double bond; two sets of two electrons shared; one sigma (σ) and one pi (π) bond
Three dashes  a triple bond: three sets of two electrons shared; one sigma and two pi (π) bonds
a triple bond: three sets of two electrons shared; one sigma and two pi (π) bonds
The valence electron configurations of the constituent atoms of a covalent compound are important factors in determining its structure, stoichiometry, and properties. For example, chlorine, with seven valence electrons, is one electron short of an octet. If two chlorine atoms share their unpaired electrons by making a covalent bond and forming Cl2, they can each complete their valence shell:
![]()
Each chlorine atom now has an octet. The electron pair being shared by the atoms is called a bonding pair; the other three pairs of electrons on each chlorine atom are called lone pairs. Lone pairs are not involved in covalent bonding.
We can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using Lewis dot symbols:
![]()
The structure on the right is the Lewis electron structure, or Lewis structure, for H2O. With two bonding pairs and two lone pairs, the oxygen atom has now completed its octet. Moreover, by sharing a bonding pair with oxygen, each hydrogen atom now has a full valence shell of two electrons. Chemists usually indicate a bonding pair by a single line, as shown here for our two examples:
![]()
In CHM 110, you should always use lines to represent bonds. The use of dots will be reserved for non-bonding (lone) electron pairs.
The following procedure can be used to construct Lewis electron structures for more complex molecules and ions:
Skills to Develop
THE EASY METHOD PROCEDURE TO DETERMINE A LEWIS DOT STRUCTURE
In this process, the basic concept of this method is that all valence electrons present will be taken away from the individual atoms and pooled together. They will first be doled out to form the requisite number of bonds. The remaining valence electrons will then be added to individual atoms as lone pairs to complete the octet of each atom.
1. Calculate the total number of valence electrons required for each atom to have an octet, or eight valence electrons. The major exception to this rule is that hydrogen wants only two (2) valence electrons. The other two most commonly-occurring exceptions are Be (4) and B (6) valence electrons.
Thus, the process for step one is 2 x (# of H atoms) + 8 x (# of most other atoms) = valence electrons needed
2. Calculate the total number of valence electrons in the given species.
If you have a polyatomic anion, you must add the number of '-' charges because there are extra valence electrons.
If you have a polyatomic cation, you must subtract the number of '+' charges because there are missing valence electrons.
3. Subtract result 2 from result 1. This is the number of valence electrons lacking for each atom to have its own octet. This number of electrons will have to be shared in bonds.
4. Divide result 3 by two, because there are two electrons per bond. This is the number of covalent bonds you must use!
5. Connect the atoms in the formula together with the number of bonds from result 4. Helpful hints:
i) H atoms form only one bond.
ii) The first atom in the formula is usually the central atom to which the other atoms bond
iii) If the particle has only three atoms, and each atom is of a different element, the atom in the middle of the formula is usually in the middle of the structure.
iv) species are as symmetrical as possible when there are several central atoms
v) For neutral species:
Group IV atoms usually form four bonds
Group V atoms usually form three bonds
Group VI atoms usually form two bonds
Group VII atoms usually form one bond
6. Add in electrons as non-bonding pairs or lone pairs (groups of two), so that each atom has eight electrons around it.
7. Count the total number of electrons in the structure. The number should be equal to the number of electrons in result 2.
CAUTIONS:
1. Never put a lone pair on an H atom.
2. The only common substances that have a lone pair on a C atom are CO and CN-. Other than those two particles, you should not put a lone pair on a C atom.
Now let’s apply this procedure to some particular compounds, beginning with one we have already discussed.
The central atom is usually the least electronegative element in the molecule or ion; hydrogen and the halogens are usually terminal.
The \(H_2O\) Molecule
- (1 O atom) x (8) + (2 H atoms) x (2) = 12 valence electrons needed
- (1 O atom) x (6) + (2 H atoms) x (1) = 8 valence electrons present.
- 12-8 = 4 electrons short, thus 4 electrons must be shared
- 4 shared electrons/ 2 electrons per bond = 2 bonds
- Connect the each of the two H atoms to the O atom with one single bond each.
- Add the remaining 4 electrons to the oxygen (as two lone pairs) to give the following structure:
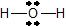
This is the Lewis structure we drew earlier. Because it gives oxygen an octet and each hydrogen two electrons, we do not need to use step 6.
The \(OCl^−\) Ion
- (1 O atom) x (8) + (1 Cl atom) x (8) = 16 valence electrons needed
- (1 O atom) x (6) + (1 Cl atom) x (7) + 1 = 14 valence electrons present.
- 16 - 14 = 2 electrons short, thus 2 electrons must be shared
- 2 shared electrons/ 2 electrons per bond = 1 bond
- Connect the two atoms with one single bond .
- Add the remaining 12 electrons as lone pairs, with 3 pairs on the O atom and 3 pairs on the Cl atom, giving the following structure:
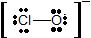
Each atom now has an octet of electrons. The Lewis electron structure is drawn within brackets as is customary for an ion, with the overall charge indicated outside the brackets, and the bonding pair of electrons is indicated by a solid line. OCl− is the hypochlorite ion, the active ingredient in chlorine laundry bleach and swimming pool disinfectant.
The \(CH_2O\) Molecule
- (1 O atom) x (8) + (1 C atom) x (8) + (2 H atoms) x (2) = 20 valence electrons needed
- (1 O atom) x (6) + (1 C atom) x (4) + (2 H atoms) x (1) = 12 valence electrons present.
- 20 - 12 = 8 electrons short, thus 8 electrons must be shared
- 8 shared electrons/ 2 electrons per bond = 4 bonds
- Because carbon is less electronegative than oxygen and hydrogen is normally terminal, C must be the central atom. Connect the two H atoms and the O atom to the C atom with with one single bond each, giving the structure:

6. There is still one more bond that you must use in the structure. At this point, the C atom has only three bonds and the O atom has only one bond. Thus, putting one more bond between the C and the O, creating a double bond, will satisfy all bonding requirements.
7. Add the remaining 4 electrons as two lone pairs on the O atom, giving the following structure:

Both the oxygen and the carbon now have an octet of electrons, so this is an acceptable Lewis electron structure. The O has two bonding pairs and two lone pairs, and C has four bonding pairs. This is the structure of formaldehyde, which is used in embalming fluid.
An alternative structure can be drawn with one H bonded to O. Formal charges, discussed later in this section, suggest that such a structure is less stable than that shown previously.
Example \(\PageIndex{1}\)
Write the Lewis electron structure for each species.
- NCl3
- S22−
- NOCl
Given: chemical species
Asked for: Lewis electron structures
Strategy:
Use the six-step procedure to write the Lewis electron structure for each species.
Solution:
- NCl3 Determine number of bonds needed
- (1 N atom) x (8) + (3 Cl atoms) x (8) = 32 valence electrons needed
- (1 N atom) x (5) + (3 Cl atoms) x (7) = 26 valence electrons present.
- 32 - 26 = 6 electrons short, thus 6 electrons must be shared
- 6 shared electrons/ 2 electrons per bond = 3 bonds
- Nitrogen is less electronegative than chlorine, and halogen atoms are usually terminal, so nitrogen is the central atom. Connect the three Cl atoms with the central N atom with three single bonds.
- You have used 6 electrons. Add the remaining 20 electrons on to the structure as lone pairs, to ensure that each atom has an octet (1 lone pair for N, and 3 lone pairs for each Cl.)
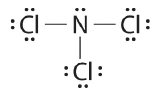
Nitrogen trichloride is an unstable oily liquid once used to bleach flour; this use is now prohibited in the United States.
b. S22- ion Determine number of bonds needed
1. (2 S atoms) x (8) = 16 valence electrons needed
2. (2 S atoms) x (6) + 2 = 14 valence electrons present.
3. 16-14 = 2 electrons short, thus 2 electrons must be shared
4. 2 shared electrons/ 2 electrons per bond = 1 bond
5. Connect the each of the two S atoms with one single bond each.
6. You have used two electrons. Add the remaining 12 electrons to the S atoms (as three lone pairs per S atom) to give the following structure:

c. NOCl Determine number of bonds needed
- (1 N atom) x (8) + (1 O atom) x (8) + (1 Cl atom) x (8) = 24 valence electrons needed
- (1 N atom) x (5) + (1 O atom) x (6) + (1 Cl atom) x (7)= 18 valence electrons present.
- 24-18 = 6 electrons short, thus 6 electrons must be shared
- 6 shared electrons/ 2 electrons per bond = 3 bonds
- Because nitrogen is less electronegative than oxygen or chlorine, it is the central atom. Also, this molecule falls into the category of a particle that has only three atoms, and each atom is of a different element, so the atom in the middle of the formula is usually in the middle of the structure. Placing one bonding pair of electrons between each pair of bonded atoms uses 4 electrons and gives the following:
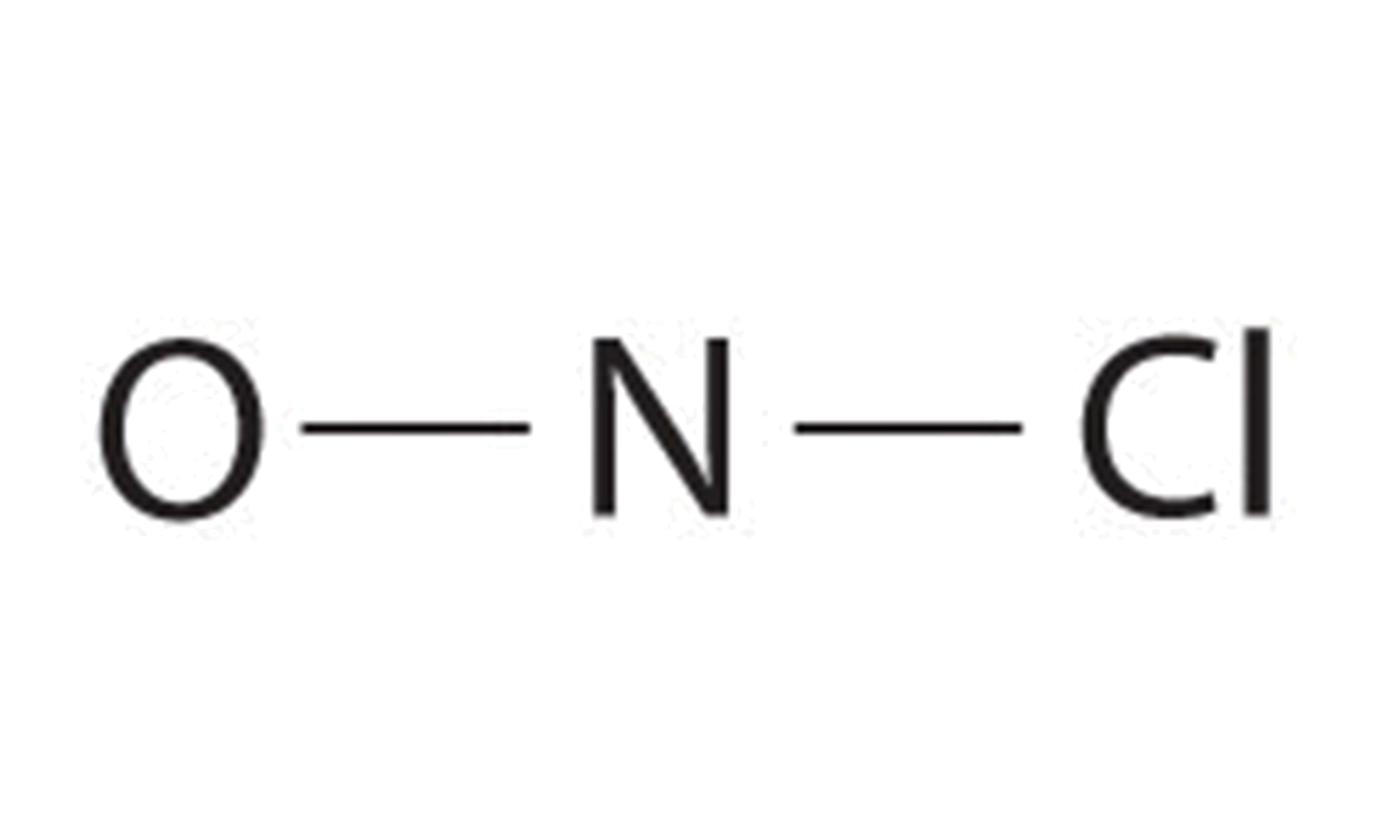
6. There is still one more bond that you must use in the structure. At this point, the N atom has only two bonds and the O atom has only one bond. Thus, putting one more bond between the N and the O, creating a double bond, will satisfy all bonding requirements. The Cl, as a halogen, is stable with just one bond.
7. You have used 6 electrons. Add the remaining 12 electrons as two lone pairs on the O atom, one lone pair on the N atom, and three lone pairs ion the Cl atom giving the following structure:
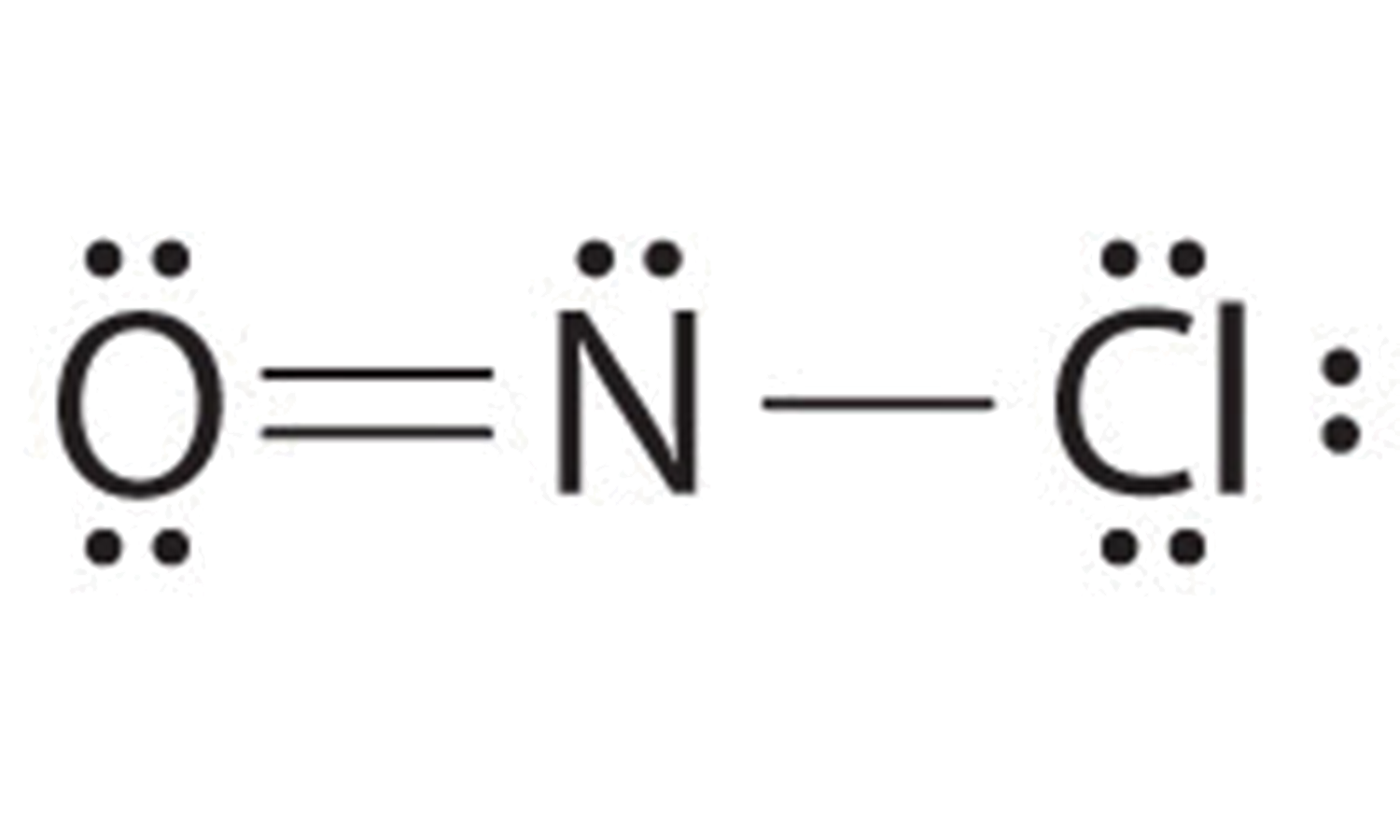
All atoms now have octet configurations. This is the Lewis electron structure of nitrosyl chloride, a highly corrosive, reddish-orange gas.
Exercise \(\PageIndex{1}\)
Write Lewis electron structures for CO2 and SCl2, a vile-smelling, unstable red liquid that is used in the manufacture of rubber.
Answer:
Using Lewis Electron Structures to Explain Stoichiometry
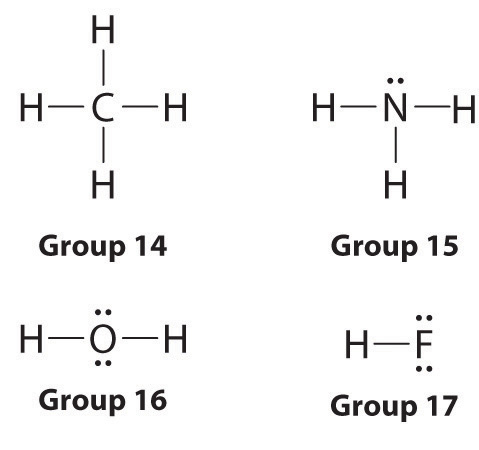
Lewis dot symbols provide a simple rationalization of why elements form compounds with the observed stoichiometries. In the Lewis model, the number of bonds formed by an element in a neutral compound is the same as the number of unpaired electrons it must share with other atoms to complete its octet of electrons. For the elements of Group VIIA (the halogens), this number is one; for the elements of Group VIA (the chalcogens), it is two; for Group VA elements, three; and for Group IVA elements four. These requirements are illustrated by the following Lewis structures for the hydrides of the lightest members of each group:
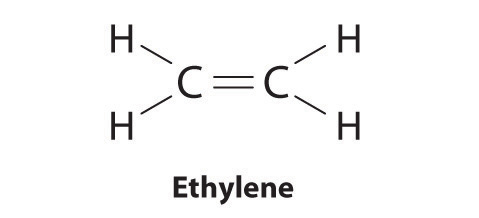
Elements may form multiple bonds to complete an octet. In ethylene, for example, each carbon contributes two electrons to the double bond, giving each carbon an octet (two electrons/bond × four bonds = eight electrons). Neutral structures with fewer or more bonds exist, but they are unusual and violate the octet rule.
Lewis structures explain why the elements of groups IVA–VIIA form neutral compounds with four, three, two, and one bonded atom(s), respectively.
Allotropes of an element can have very different physical and chemical properties because of different three-dimensional arrangements of the atoms; the number of bonds formed by the component atoms, however, is always the same. Diamond is a hard, transparent solid; graphite is a soft, black solid; and the fullerenes have open cage structures. Despite these differences, the carbon atoms in all three allotropes form four bonds, in accordance with the octet rule.
Elemental phosphorus also exists in three forms: white phosphorus, a toxic, waxy substance that initially glows and then spontaneously ignites on contact with air; red phosphorus, an amorphous substance that is used commercially in safety matches, fireworks, and smoke bombs; and black phosphorus, an unreactive crystalline solid with a texture similar to graphite (Figure \(\PageIndex{3}\)). Nonetheless, the phosphorus atoms in all three forms obey the octet rule and form three bonds per phosphorus atom.
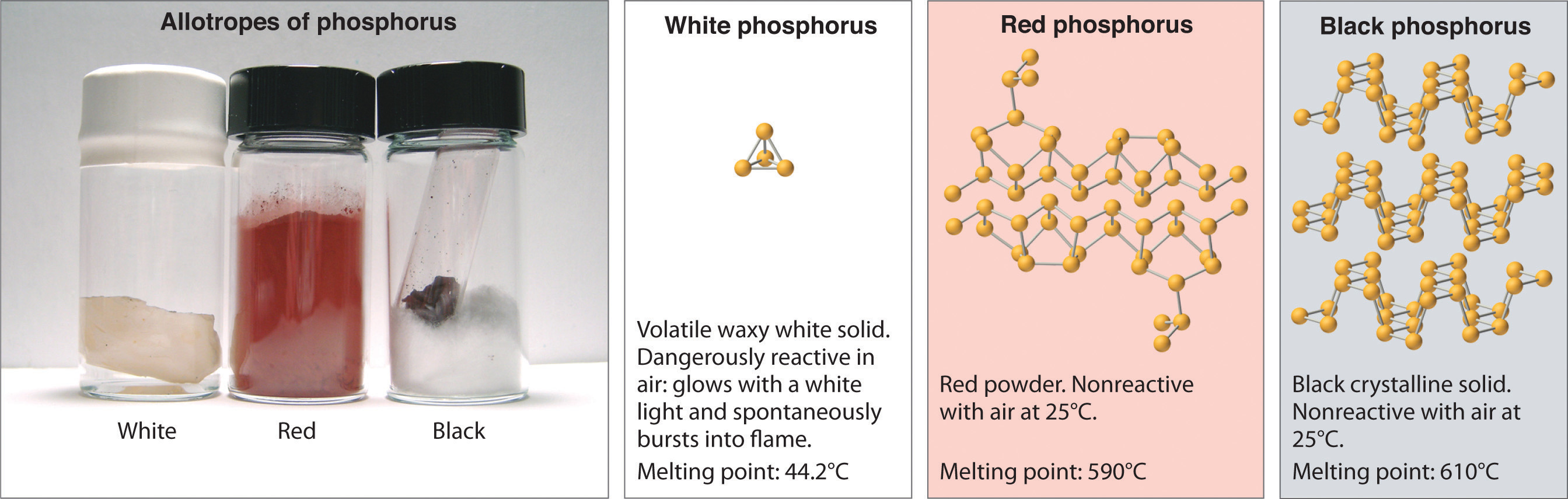
Figure \(\PageIndex{3}\): The Three Allotropes of Phosphorus: White, Red, and Black. ll three forms contain only phosphorus atoms, but they differ in the arrangement and connectivity of their atoms. White phosphorus contains P4 tetrahedra, red phosphorus is a network of linked P8 and P9 units, and black phosphorus forms sheets of six-membered rings. As a result, their physical and chemical properties differ dramatically.
Summary
Lewis dot symbols provide a simple rationalization of why elements form compounds with the observed stoichiometries. In Lewis electron structures, we encounter bonding pairs, which are shared by two atoms, and lone pairs, which are not shared between atoms. Lewis structures are an attempt to rationalize why certain stoichiometries are commonly observed for the elements of particular families. Neutral compounds of group IVA elements typically contain four bonds around each atom (a double bond counts as two, a triple bond as three), whereas neutral compounds of group VA elements typically contain three bonds.
Contributors
Modified by Joshua Halpern (Howard University)
- Modified by Tom Neils (Grand Rapids Community College)