9.3: Alkenes
- Page ID
- 85175
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Alkenes contain carbon-carbon double bonds and are unsaturated hydrocarbons with the molecular formula is CnH2n. This is also the same molecular formula as cycloalkanes. Alkenes are named using the same general naming rules for alkanes, except that the suffix is now -ene. There are a few other small differences:
- The main chain of carbon atoms must contain both carbons in the double bond. The main chain is numbered so that the double bond gets the smallest number.
- Before the root name, the number of the carbon atom at which the double bond starts (the smaller number) is written.
- If more than one double bond is present, the prefixes di-, tri-, tetra-, etc. are used before the -ene, and (strangely) the letter "a" is added after the prefix for the number of carbon atoms.
Introduction
Alkenes and alkynes are hydrocarbons which respectively have carbon-carbon double bond and carbon-carbon triple bond functional groups. The molecular formulas of these unsaturated hydrocarbons reflect the multiple bonding of the functional groups:
Alkenes are named as if they were alkanes, but the "-ane" suffix is changed to "-ene". If the alkene contains only one double bond and that double bond is terminal (the double bond is at one end of the molecule or another) then it is not necessary to place any number in front of the name.
butane: C4H10 (CH3CH2CH2CH3)
butene: C4H8 (CH2=CHCH2CH3)
If the double bond is not terminal (if it is on a carbon somewhere in the center of the chain) then the carbons should be numbered in such a way as to give the first of the two double-bonded carbons the lowest possible number, and that number should precede the "ene" suffix with a dash, as shown below.
correct: pent-2-ene (CH3CH=CHCH2CH3)
incorrect: pent-3-ene (CH3CH2CH=CHCH3)
The second one is incorrect because flipping the formula horizontally results in a lower number for the alkene.
If there is more than one double bond in an alkene, all of the bonds should be numbered in the name of the molecule - even terminal double bonds. The numbers should go from lowest to highest, and be separated from one another by a comma. The IUPAC numerical prefixes are used to indicate the number of double bonds.
octa-2,4-diene: CH3CH=CHCH=CHCH2CH2CH3
deca-1,5-diene: CH2=CHCH2CH2CH=CHCH2CH2CH2CH3
Note that the numbering of "2-4" above yields a molecule with two double bonds separated by just one single bond. Double bonds in such a condition are called "conjugated", and they represent an enhanced stability of conformation, so they are energetically favored as reactants in many situations and combinations.
| Alkane | CnH2n+2 | This is the maximum H/C ratio for a given number of carbon atoms. |
|---|---|---|
| Alkene | CnH2n | Each double bond reduces the number of hydrogen atoms by 2. |
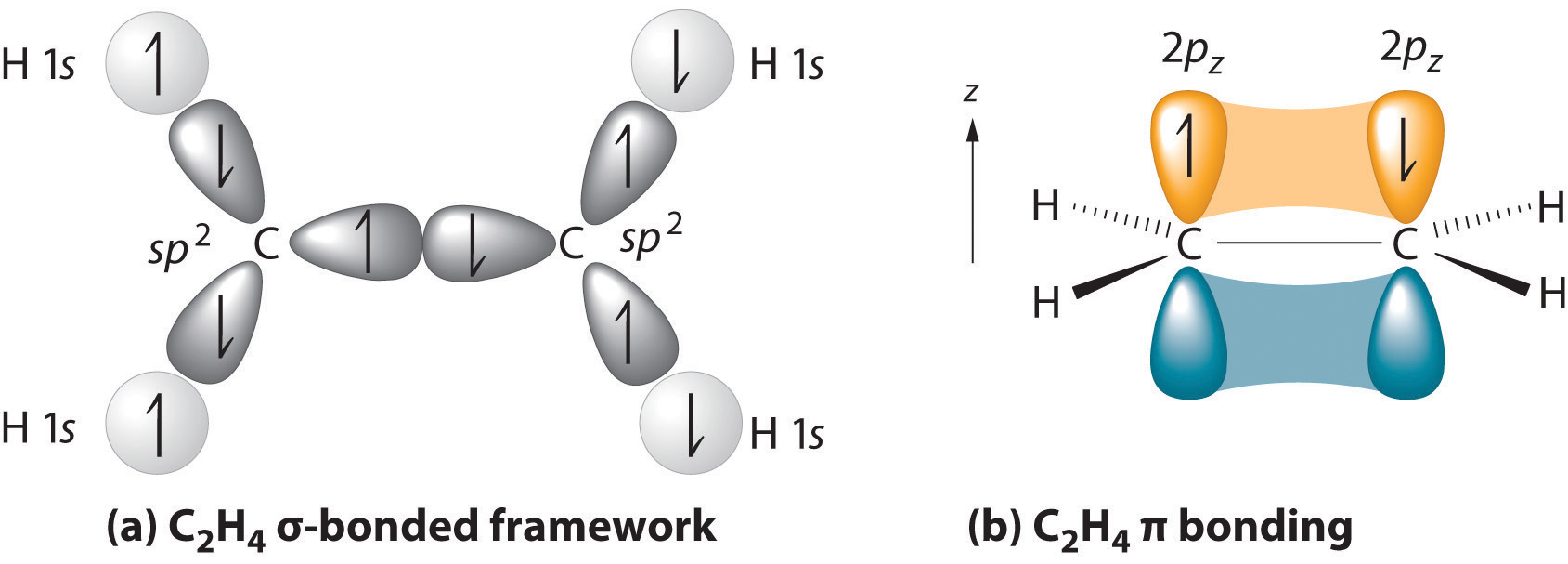
The parent structure is the longest chain containing both carbon atoms of the double bond. The two carbon atoms of a double bond and the four atoms attached to them lie in a plane, with bond angles of approximately 120° A double bond consists of one sigma bond formed by overlap of sp2 hybrid orbitals and one pi bond formed by overlap of parallel 2 p orbitals
The molecular formula of a hydrocarbon provides information about the possible structural types it may represent. For example, consider compounds having the formula C5H8. The formula of the five-carbon alkane pentane is C5H12 so the difference in hydrogen content is 4. This difference suggests such compounds may have a triple bond, two double bonds, a ring plus a double bond, or two rings. Some examples are shown here, and there are at least fourteen others!
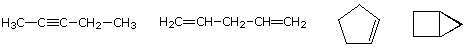
IUPAC Rules for Alkene and Cycloalkene Nomenclature
- The ene suffix (ending) indicates an alkene or cycloalkene.
- The longest chain chosen for the root name must include both carbon atoms of the double bond.
- The root chain must be numbered from the end nearest a double bond carbon atom. If the double bond is in the center of the chain, the nearest substituent rule is used to determine the end where numbering starts.
- The smaller of the two numbers designating the carbon atoms of the double bond is used as the double bond locator. If more than one double bond is present the compound is named as a diene, triene or equivalent prefix indicating the number of double bonds, and each double bond is assigned a locator number.
- In cycloalkenes the double bond carbons are assigned ring locations #1 and #2. Which of the two is #1 may be determined by the nearest substituent rule.
- Substituent groups containing double bonds are:
H2C=CH– Vinyl group
H2C=CH–CH2– Allyl group
In this diagram this is a cis conformation. It has both the substituents going upward. This molecule would be called (cis) 5-chloro-3-heptene.)
Trans would look like this
v. On the other hand if there are 3 or 4 non-hydrogen different atoms attached to the alkene then use the E, Z system.
E (entgegen) means the higher priority groups are opposite one another relative to the double bond.
Z (zusammen) means the higher priority groups are on the same side relative to the double bond.
(You could think of Z as Zame Zide to help memorize it.)
Solution
In this example it is E-4-chloro-3-heptene. It is E because the Chlorine and the CH2CH3 are the two higher priorities and they are on opposite sides.
vi. A hydroxyl group gets precedence over th double bond. Therefore alkenes containing alchol groups are called alkenols. And the prefix becomes --enol. And this means that now the alcohol gets lowest priority over the alkene.
vii. Lastly remember that alkene substituents are called alkenyl. Suffix --enyl.
Here is a chart containing the systemic name for the first twenty straight chain alkenes.
| Name | Molecular formula |
| Ethene | C2H4 |
| Propene | C3H6 |
| Butene | C4H8 |
| Pentene | C5H10 |
| Hexene | C6H12 |
| Heptene | C7H14 |
| Octene | C8H16 |
| Nonene | C9H18 |
| Decene | C10H20 |
| Undecene | C11H22 |
| Dodecene | C12H24 |
| Tridecene | C13H26 |
| Tetradecene | C14H28 |
| Pentadecene | C15H30 |
| Hexadecene | C16H32 |
| Heptadecene | C17H34 |
| Octadecene | C18H36 |
| Nonadecene | C19H38 |
| Eicosene | C20H40 |
Did you notice how there is no methene? Because it is impossible for a carbon to have a double bond with nothing.
Geometric Isomers
Double bonds can exist as geometric isomers and these isomers are designated by using either the cis / trans designation or the modern E / Z designation.
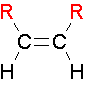
cis Isomers
.The two largest groups are on the same side of the double bond.
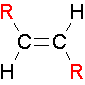
trans Isomers
...The two largest groups are on opposite sides of the double bond.
E/Z nomenclature
E = entgegan ("trans") Z = zusamen ("cis")
Priority of groups is based on the atomic mass of attached atoms (not the size of the group). An atom attached by a multiple bond is counted once for each bond.
fluorine atom > isopropyl group > n-hexyl group
deuterium atom > hydrogen atom
-CH2-CH=CH2 > -CH2CH2CH3
Try to name the following compounds using both conventions...
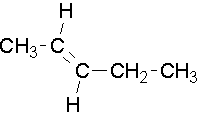
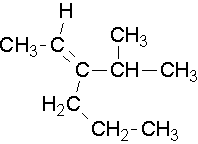
Common names
Remove the -ane suffix and add -ylene. There are a couple of unique ones like ethenyl's common name is vinyl and 2-propenyl's common name is allyl. That you should know are...
- vinyl substituent H2C=CH-
- allyl substituent H2C=CH-CH2-
- allene molecule H2C=C=CH2
- isoprene
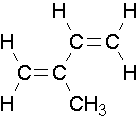
Endocyclic Alkenes
Endocyclic double bonds have both carbons in the ring and exocyclic double bonds have only one carbon as part of the ring.

Cyclopentene is an example of an endocyclic double bond.

Methylenecylopentane is an example of an exocyclic double bond.
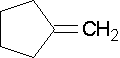
Name the following compounds...
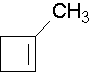
1-methylcyclobutene. The methyl group places the double bond. It is correct to also name this compound as 1-methylcyclobut-1-ene.
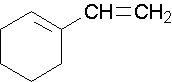
1-ethenylcyclohexene, the methyl group places the double bond. It is correct to also name this compound as 1-ethenylcyclohex-1-ene. A common name would be 1-vinylcyclohexene.
Try to draw structures for the following compounds...
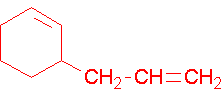
- 2-vinyl-1,3-cyclohexadiene
Outside links
References
- Vollhardt, Peter, and Neil E. Schore. Organic Chemistry: Structure and Function. 5th Edition. New York: W. H. Freeman & Company, 2007.
Problems
Try to name the following compounds...

1-pentene or pent-1-ene

2-ethyl-1-hexene or 2-ethylhex-1-ene
Try to draw structures for the following compounds...
- 2-pentene
CH3–CH=CH–CH2–CH3
- 3-heptene
CH3–CH2–CH=CH–CH2–CH2–CH3
b. Give the double bond the lowest possible numbers regardless of substituent placement.
• Try to name the following compound...
 J
J
• Try to draw a structure for the following compound...
4-methyl-2-pentene J
Name the following structures:
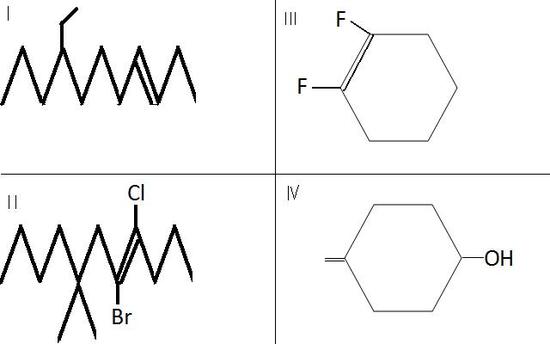
v. Draw (Z)-5-Chloro-3-ethly-4-hexen-2-ol.
Answers
I. trans-8-ethyl-3-undecene
II. E-5-bromo-4-chloro-7,7-dimethyl-4-undecene
III. Z-1,2-difluoro-cyclohexene
IV. 4-ethenylcyclohexanol.
V. 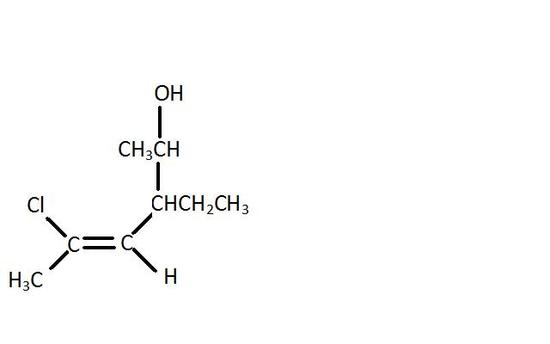
Contributors and Attributions
- S. Devarajan (UCD)
Richard Banks (Boise State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
- Jonathan Mooney (McGill University)


