Kinetics and Catalysis of Drug-Target Binding in Biology
- Page ID
- 418900
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This Exemplar will teach the following concept(s) from the ACS Examinations Institute General Chemistry ACCM:
VII.A.2.a Chemical reactions may occur at a wide range of rates, and a key aspect of rate is related to the concentration of species involved in the reaction.
VII.A.1.b Once known, a rate law can be used to quantitatively predict concentrations of species involved in a reaction as a function of time.
Drug Degradation, Effectiveness, and Kinetics
While the effectiveness of pharmaceuticals is impacted by a variety of different biological and chemical factors, kinetics plays a large role in determining the success of a particular molecule as a therapeutic. The rate of degradation, or breakdown, of a drug in the body impacts how long it remains effective, how long traces can be found in the body, and even what doses are appropriate. Beyond this, drug kinetics can even have impacts on the severity of side effects or how quickly drugs are able to produce an effect in the body.
Rates of Degradation
One of the main aspects of kinetics involved in pharmaceuticals is the rate of degradation of drugs. Based on how long a drug remains in the body before breaking down, it can have varying levels of impact. Dosages for drugs also take this degradation rate into account to ensure drug levels in the body do not exceed safe levels.
Rate laws link the concentrations of reactants and the rate of reaction. The chemical equation \(A → B + C\) represents a decomposition reaction where substance A gets broken down into components \(B\) and \(C\). A rate law to represent this degradation could be \(rate = k[A]\), where \(k\) is the rate constant that represents the proportionality of \([A]\) and the rate of reaction. In this case, \(k\) would have units of 1/s because the rate law is of the first order, represented by the concentration of reactants, \([A]\), adding up to the power of 1. Reaction orders indicate the number of reactants in the equation that have concentrations that affect the rate of reaction and the amount to which a change in concentration of a reactant would amplify the rate of reaction. Although higher reaction orders are possible, we usually deal with zero, first, second, and third-order reaction orders. Zero-order reactions suggest no relationship between reactant concentration and rate of reaction.
Most pharmaceuticals operate as first or zero-order reactions. In dealing with the human body, one must consider the elimination of the drug from the system. With zero- order reactions, the body reduces the amount of the drug in the system based on time. With first-order reactions, the more common of the two for pharmaceuticals, the rate at which the body eliminates the drug is proportional to the concentration of the drug consumed.
Aspirin, or acetylsalicylic acid, is activated in the body through hydrolysis (the addition of water to split molecules). The breakdown of aspirin produces salicylic acid, which is the molecule that works to reduce fever and inflammation in the body. Therefore, the rate at which aspirin is hydrolyzed affects the speed at which aspirin can start producing an effect. To represent the breakdown of aspirin as an equation, it can be simplified into the equation:
Aspirin + H2O → Salicylic acid + Acetic acid
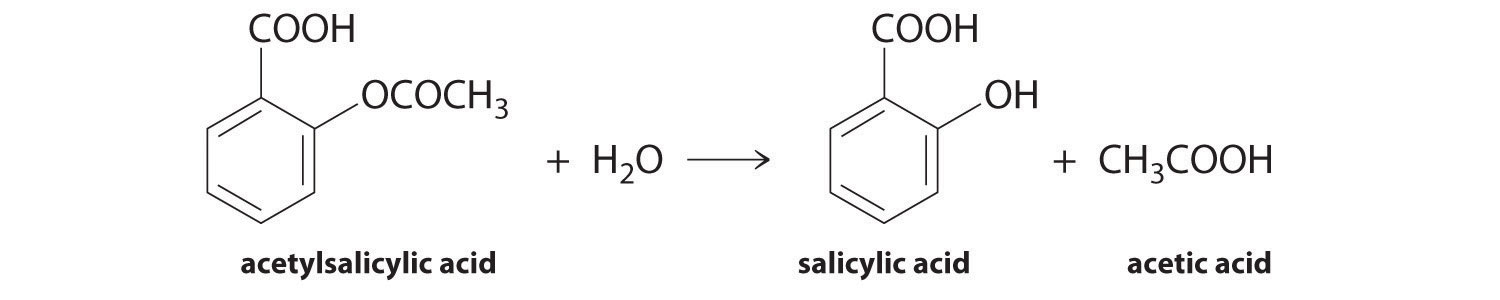
Figure 1. Hydrolysis of Aspirin reaction
A structural diagram for an aspirin hydrolysis reaction.
Libretexts. 14.2: Reaction rates. https://chem.libretexts.org/Bookshel...Reaction_Rates (accessed Dec 6, 2022).
The rate law is based on the concentrations of the reactants, specifically aspirin. Even though water is a reactant in the reaction, it is not included in the rate law because it is present in extreme excess compared to the amount of aspirin present. So, the rate law would then be
\[ rate = k [A]^m \]
Given the rate law above, determine the order of the reaction based on the data below. This data shows the hydrolysis of aspirin at pH= 7 and 60 C. Experimental data has been rounded or clarity.
|
Time |
[Acetylsalicylic acid] (mM) |
Rate (mM/min) |
|
40 |
6.6 |
0.069 |
|
80 |
5.5 |
0.058 |
|
120 |
4.4 |
0.046 |
|
160 |
3.6 |
0.038 |
|
190 |
3.1 |
0.032 |
Solution
In order to determine the reaction order, we need to study how the rate changes with changes in the concentration of aspirin. This can be done by comparing ratios of aspirin concentration and rate for two different points in the reaction.
\(\frac{[A_1]}{[A_1]} = \frac{6.6 mM}{4.4mM} = 1.5 \)
\(\frac{rate_1}{rate_2} = \frac{.069 mM/min}{0.032 mM/min} = 1.5 \)
The results show that when the concentration of aspirin is decreased by a factor of 1.5, the rate also decreases at the same rate. This indicates that the rate is dependent on concentration, and the reaction is a first-order reaction (m=1). Repeating this with different data points produces the same result.
How would the rate of aspirin breakdown differ if the reaction was zero-order? Draw a sample table like the one shown in the previous question.
Solution
If the hydrolysis of aspirin were a zero-order reaction, the rate would be constant even with changes in aspirin concentration. Unlike the table above, the rate wouldn’t decrease as the reaction progresses, instead looking more like this:
|
Time |
[Acetylsalicylic acid] (mM) |
Rate (mM/min) |
|
40 |
6.6 |
0.070 |
|
80 |
5.5 |
0.070 |
|
120 |
4.4 |
0.070 |
|
160 |
3.6 |
0.070 |
|
190 |
3.1 |
0.070 |
Drug Side Effects
While differential rate laws connect the concentration of reactants with the rate of reaction over time, integrated rate laws delve into the relationship between an initial and measured concentration of the reactant(s) and the rate of reaction at a specific time.
The integrated rate laws for zero, first, and second-order reactions are shown below.
| Order | Integrated Rate Law |
| 0 | \([A]_t = -kt + [A]_0 \) |
| 1 | \( ln[A]_t = -kt + ln [A]_0 \) |
| 2 | \( \frac{1}{[A]_t} = kt + \frac{1}{[A]_0} \) |
Kinetics of drugs is also involved in studying the negative side effects that certain drugs can produce. The negative side effects of antipsychotic drugs (APDs) are currently being explored in relation to kinetics. Antipsychotic drugs commonly result in negative symptoms that occur due to binding to the target, referred to as extrapyramidal symptoms. These can impair communication and motor skills, with severe effects that can be debilitating. As a result, scientists have been studying the association and dissociation rates of different APDs to determine relationships between these rates and the severity of extrapyramidal symptoms. kon is the rate constant for the association or binding to the target, and koff is the dissociation rate constant.
The kon and koff values for several antipsychotic medications have been experimentally determined to study extrapyramidal symptoms. The values for two drugs, Haloperidol and Chlorpromazine, are shown in the table below.
|
Drug |
kon (1/M*min) |
koff (1/min) |
|
Haloperidol |
109.1 |
10-3 |
|
Chlorpromazine |
109.3 |
100.1 |
a. If the units for kon are 1/M*min, what is the overall order for the association reaction? The units for koff are 1/min, so what is the dissociation reaction’s overall order?
b. Based on the overall order for each, what would be the integrated rate law for both reactions?
c. What do the kon values for Haloperidol and Chlorpromazine indicate about their association rates?
Solution
a. The units for k vary with the rate order, \( M^{1-order} * min^{-1} \).
\( M^{-1} * min^{-1} \) would be a second order reaction, and \( s^{-1}\) would be first order.
b. The integrated rate law for on would be: \( \frac{1}{[I]} = kt + \frac{1}{[I]_0} \)
The integrated rate law for off would be:
| \( ln[I] = -kt + ln [I]_0 \) |
c. If haloperidol had a lower kon value than chlorpromazine, it would have a slower association rate.

Figure 2. APD drug affinities
Shows characteristics of Haloperidol and Chloropromazine
Sykes, D. A.; Moore, H.; Stott, L.; Holliday, N.; Javitch, J. A.; Lane, J. R.; Charlton, S. J. Extrapyramidal Side Effects of Antipsychotics Are Linked to Their Association Kinetics at Dopamine D2 Receptors. Nature Communications 2017, 8 (1). https://doi.org/10.1038/s41467-017-00716-z (accessed Nov 09, 2022).
Studies have also shown that larger kon values are involved in rapid onset of drug action (where the drug begins producing the desired effect). For example, indacaterol (a bronchodilator used to treat COPD) has a relatively high kon value of 1.5 * 10-6 1/Ms and produces an effect in the body in only 7.8 minutes. So, kinetics is a major area of focus in drug development as a means of making drugs more effective.
References
Borowy, C. S.; Ashurst, J. V. Physiology, Zero and First Order Kinetics https://www.ncbi.nlm.nih.gov/books/NBK499866/ (accessed Nov 09, 2022).
Differential and integrated rate laws - laney college. https://laney.edu/abraham-reyes/wp-content/uploads/sites/229/2015/08/Differential-and-Integrated-Rate-
Laws1.pdf (accessed Nov 09, 2022).
IJzerman, A. P.; Guo, D. Drug–Target Association Kinetics in Drug Discovery. Trends in Biochemical Sciences 2019, 44 (10), 861–871.
https://doi.org/10.1016/j.tibs.2019.04.004 (accessed Nov 09, 2022).
Libretexts. 14.2: Reaction rates. https://chem.libretexts.org/Bookshel...Reaction_Rates (accessed Dec 6, 2022).
Sykes, D. A.; Moore, H.; Stott, L.; Holliday, N.; Javitch, J. A.; Lane, J. R.; Charlton, S. J. Extrapyramidal Side Effects of Antipsychotics Are Linked to Their Association
Kinetics at Dopamine D2 Receptors. Nature Communications 2017, 8 (1). https://doi.org/10.1038/s41467-017-00716-z (accessed Nov 09, 2022).
Tonge, P. J. Drug–Target Kinetics in Drug Discovery. ACS Chemical Neuroscience 2017, 9 (1), 29–39. https://doi.org/10.1021/acschemneuro.7b00185 (accessed Nov
09, 2022).
W Cary Mobley; Amiji, M. M.; Cook, T. J. Applied Physical Pharmacy; Mcgraw-Hill Education: New York, 2019.

