1.3.3: Reaction Stoichiometry
- Page ID
- 210594
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Use stoichiometry to calculate the amount (in moles or grams) of a particular substance produced or used in a chemical reaction.
- Use stoichiometry to determine molecular formulas.
A balanced chemical equation provides a great deal of information in a very succinct format. Chemical formulas provide the identities of the reactants and products involved in the chemical change, allowing classification of the reaction. Coefficients provide the relative numbers of these chemical species, allowing a quantitative assessment of the relationships between the amounts of substances consumed and produced by the reaction. These quantitative relationships are known as the reaction’s stoichiometry, a term derived from the Greek words stoicheion (meaning “element”) and metron (meaning “measure”). In this module, the use of balanced chemical equations for various stoichiometric applications is explored.
The general approach to using stoichiometric relationships is similar in concept to the way people go about many common activities. Cooking, for example, offers an appropriate comparison. Suppose a recipe for making eight pancakes calls for 1 cup pancake mix, \(\dfrac{3}{4}\) cup milk, and one egg. The “equation” representing the preparation of pancakes per this recipe is
\[\mathrm{1\:cup\: mix+\dfrac{3}{4}\:cup\: milk+1\: egg \rightarrow 8\: pancakes} \label{4.4.1}\]
If two dozen pancakes are needed for a big family breakfast, the ingredient amounts must be increased proportionally according to the amounts given in the recipe. For example, the number of eggs required to make 24 pancakes is
\[\mathrm{24\: \cancel{pancakes} \times \dfrac{1\: egg}{8\: \cancel{pancakes}}=3\: eggs} \label{4.4.2}\]
Balanced chemical equations are used in much the same fashion to determine the amount of one reactant required to react with a given amount of another reactant, or to yield a given amount of product, and so forth. The coefficients in the balanced equation are used to derive stoichiometric factors (mole bridges) that permit computation of the desired quantity. To illustrate this idea, consider the production of ammonia by reaction of hydrogen and nitrogen:
\[\ce{N2}(g)+\ce{3H2}(g)\rightarrow \ce{2NH3}(g) \label{4.4.3}\]
This equation shows that ammonia molecules are produced from hydrogen molecules in a 2:3 ratio, and stoichiometric factors may be derived using any amount (number) unit:
\[\ce{\dfrac{2NH3 \: molecules}{3H2 \: molecules}\: or \: \dfrac{2 \: doz \: NH3\: molecules}{3\: doz\:H2 \:molecules} \: or \: \dfrac{2\: mol\: NH3\: molecules}{3\: mol\: H2\: molecules}} \label{4.4.4}\]
These stoichiometric factors can be used to compute the number of ammonia molecules produced from a given number of hydrogen molecules, or the number of hydrogen molecules required to produce a given number of ammonia molecules. Similar factors may be derived for any pair of substances in any chemical equation.
Example \(\PageIndex{1}\): Moles of Reactant Required in a Reaction
How many moles of I2 are required to react with 0.429 mol of Al according to the following equation (see Figure \(\PageIndex{2}\))?
\[\ce{2Al + 3I2 \rightarrow 2AlI3} \label{4.4.5}\]

Solution
Referring to the balanced chemical equation, the stoichiometric factor (mole bridge) relating the two substances of interest is \(\ce{\dfrac{3\: mol\: I2}{2\: mol\: Al}}\). The molar amount of iodine is derived by multiplying the provided molar amount of aluminum by this factor:

\[\begin{align*} \mathrm{mol\: I_2} &=\mathrm{0.429\: \cancel{mol\: Al}\times \dfrac{3\: mol\: I_2}{2\:\cancel{mol\: Al}}} \\[4pt] &=\mathrm{0.644\: mol\: I_2} \end{align*}\]
Exercise \(\PageIndex{1}\)
How many moles of Ca(OH)2 are required to react with 1.36 mol of H3PO4 to produce Ca3(PO4)2 according to the equation \(\ce{3Ca(OH)2 + 2H3PO4 \rightarrow Ca3(PO4)2 + 6H2O}\) ?
- Answer
-
2.04 mol
Example \(\PageIndex{2}\): Number of Product Molecules Generated by a Reaction
How many carbon dioxide molecules are produced when 0.75 mol of propane is combusted according to this equation?
Solution
The approach here is the same as for Example \(\PageIndex{1}\), though the absolute number of molecules is requested, not the number of moles of molecules. This will simply require use of the moles-to-numbers conversion factor, Avogadro’s number.
The balanced equation shows that carbon dioxide is produced from propane in a 3:1 ratio:
Using this stoichiometric factor, the provided molar amount of propane, and Avogadro’s number,

\[\mathrm{0.75\: \cancel{mol\: C_3H_8}\times \dfrac{3\: \cancel{mol\: CO_2}}{1\:\cancel{mol\:C_3H_8}}\times \dfrac{6.022\times 10^{23}\:CO_2\:molecules}{\cancel{mol\:CO_2}}=1.4\times 10^{24}\:CO_2\:molecules} \label{4.4.8}\]
Exercise \(\PageIndex{1}\)
How many NH3 molecules are produced by the reaction of 4.0 mol of Ca(OH)2 according to the following equation:
\[\ce{(NH4)2SO4 + Ca(OH)2 \rightarrow 2NH3 + CaSO4 + 2H2O} \label{4.4.9} \nonumber\]
- Answer
-
4.8 × 1024 NH3 molecules
These examples illustrate the ease with which the amounts of substances involved in a chemical reaction of known stoichiometry may be related. Directly measuring numbers of atoms and molecules is, however, not an easy task, and the practical application of stoichiometry requires that we use the more readily measured property of mass.
Example \(\PageIndex{3}\): Relating Masses of Reactants and Products
What mass of sodium hydroxide, NaOH, would be required to produce 16 g of the antacid milk of magnesia [magnesium hydroxide, Mg(OH)2] by the following reaction?
Solution
The approach used previously in Examples \(\PageIndex{1}\) and \(\PageIndex{2}\) is likewise used here; that is, we must derive an appropriate stoichiometric factor from the balanced chemical equation and use it to relate the amounts of the two substances of interest. In this case, however, masses (not molar amounts) are provided and requested, so additional steps of the sort learned in the previous chapter are required. The calculations required are outlined in this flowchart:
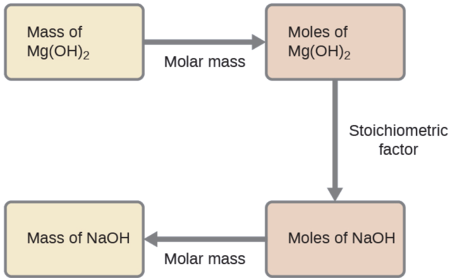
Exercise \(\PageIndex{3}\)
What mass of gallium oxide, Ga2O3, can be prepared from 29.0 g of gallium metal? The equation for the reaction is \(\ce{4Ga + 3O2 \rightarrow 2Ga2O3}\).
- Answer
-
39.0 g
Example \(\PageIndex{4}\): Relating Masses of Reactants
What mass of oxygen gas, O2, from the air is consumed in the combustion of 702 g of octane, C8H18, one of the principal components of gasoline?
Solution
The approach required here is the same as for the Example \(\PageIndex{3}\), differing only in that the provided and requested masses are both for reactant species.
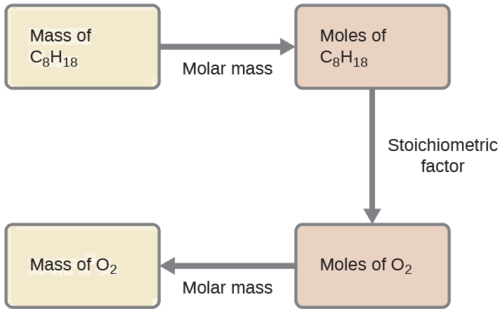
\(\mathrm{702\:\cancel{g\:\ce{C8H18}}\times \dfrac{1\:\cancel{mol\:\ce{C8H18}}}{114.23\:\cancel{g\:\ce{C8H18}}}\times \dfrac{25\:\cancel{mol\:\ce{O2}}}{2\:\cancel{mol\:\ce{C8H18}}}\times \dfrac{32.00\: g\:\ce{O2}}{\cancel{mol\:\ce{O2}}}=2.46\times 10^3\:g\:\ce{O2}}\)
Exercise \(\PageIndex{4}\)
What mass of CO is required to react with 25.13 g of Fe2O3 according to the equation \(\ce{Fe2O3 + 3CO \rightarrow 2Fe + 3CO2}\)?
- Answer
-
13.22 g
These examples illustrate just a few instances of reaction stoichiometry calculations. Numerous variations on the beginning and ending computational steps are possible depending upon what particular quantities are provided and sought (volumes, solution concentrations, and so forth). Regardless of the details, all these calculations share a common essential component: the use of stoichiometric factors derived from balanced chemical equations. Figure \(\PageIndex{2}\) provides a general outline of the various computational steps associated with many reaction stoichiometry calculations.
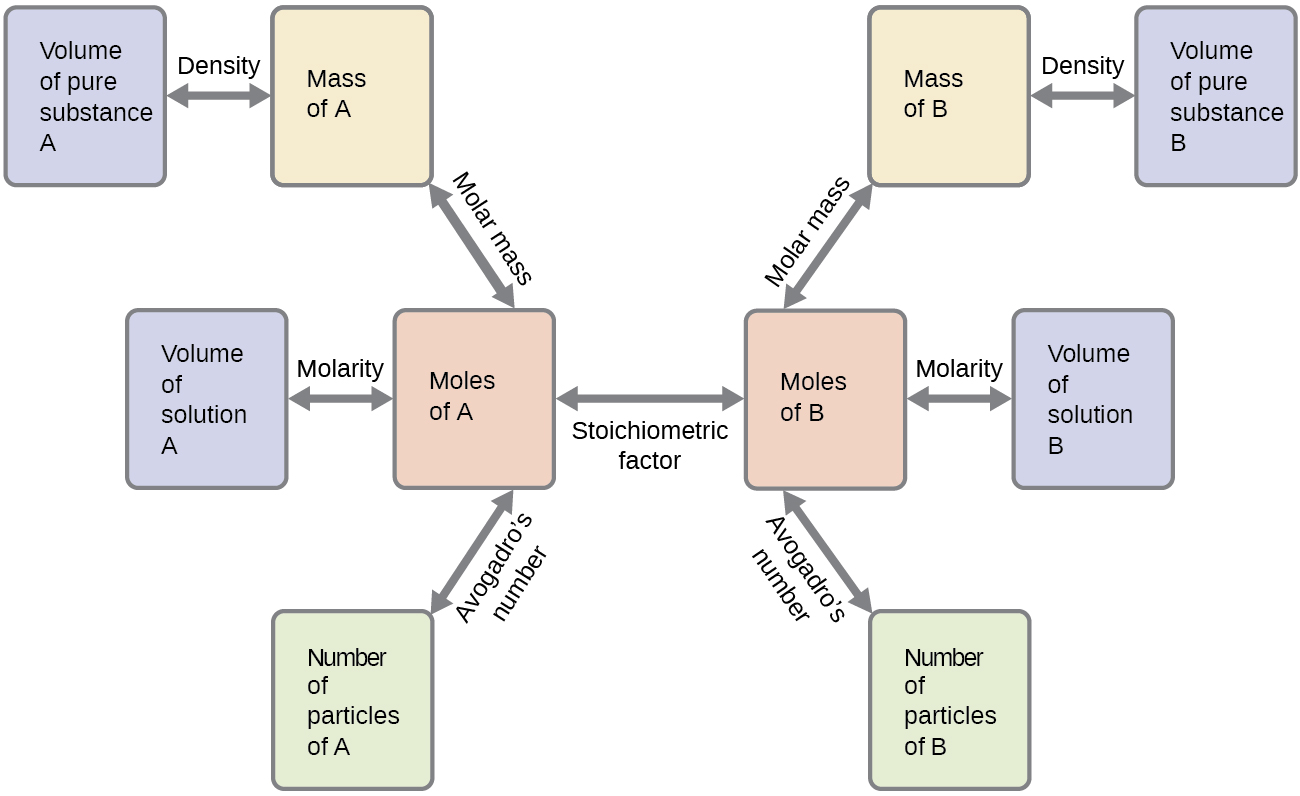
Figure \(\PageIndex{2}\): The flowchart depicts the various computational steps involved in most reaction stoichiometry calculations.
Using Stoichiometry to Determine Molecular Formulas
Since a chemical formula is the mole ratios of elements when they combine, if we are given the mass of the elements that reacted with each other, we should be able to figure the moles from this, and then the ratios.
Example \(\PageIndex{4}\): Determining A Molecular Formula from Mole Ratios
If 1.5875 g of copper react with 0.8875 g of chlorine to form copper chloride, write the chemical formula for the copper chloride.
Step 1: Determine the number of moles of an element which reacted. In this case, Cu.
\[\mathrm{1.5875\:\cancel{g\: Cu} \times \dfrac{1\:\cancel{mol\: Cu}}{63.5\:\cancel{g\: Cu}}=0.025\: mol\: Cu}\]
Step 2: Determine the number of moles of the other element which reacted. In this case, Cl.
\[\mathrm{0.8875\:\cancel{g\: Cl} \times \dfrac{1\:\cancel{mol\: Cl}}{35.5\:\cancel{g\: Cl}}=0.025\: mol\: Cl}\]
Step 3: Make a ratio of moles one element in a compound to moles of the other element in the compound (as a general rule, the larger number will go on top of the ratio.) Since we are trying to find the formula of copper chloride, we would use moles copper to and moles chlorine.
0.025 moles Cu / 0.025 moles Cl = 1 mole Cu / 1 mole Cl or Cu Cl


