24.4 Basicity of Arylamines
- Page ID
- 91032
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- use the concept of resonance to explain why arylamines are less basic than their aliphatic counterparts.
- arrange a given series of arylamines in order of increasing or decreasing basicity.
- discuss, in terms of inductive and resonance effects, why a given arylamine is more or less basic than aniline.
With reference to the discussion of base strength, the traditional explanation for the base‑strengthening effect of electron‑releasing (I) substituents is that such substituents help to stabilize the positive charge on an arylammonium ion more than they stabilize the unprotonated compound, thereby lowering ΔG°.
The electron‑withdrawing (i.e., deactivating) substituents decrease the stability of a positively charged arylammonium ion.
Note that the arylammonium ion derived from aniline, PhNH3+, is commonly referred to as the anilinium ion.
Basicity of Aniline
Aniline is substantially less basic than methylamine, as is evident by looking at the pKa values for their respective ammonium conjugate acids (remember that the lower the pKa of the conjugate acid, the weaker the base).
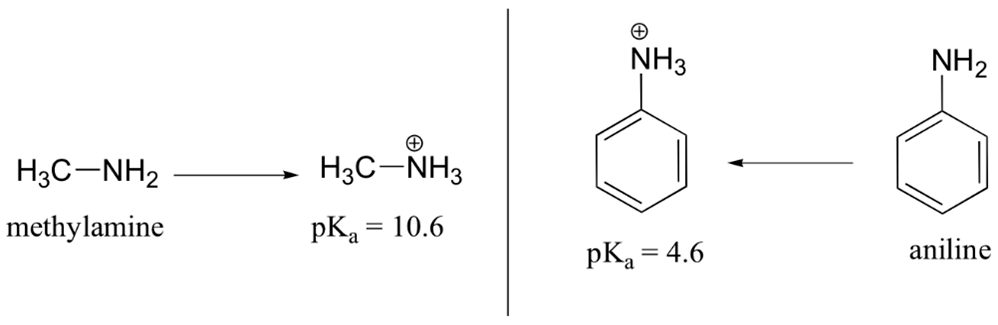
This difference is basicity can be explained by the observation that, in aniline, the lone pair of electrons on the nitrogen are delocalized by the aromatic p system, making it less available for bonding to H+ and thus less basic. The lone pair electrons of aniline are involved in four resonance forms making them more stable and therefore less reactive relative to alkylamines.
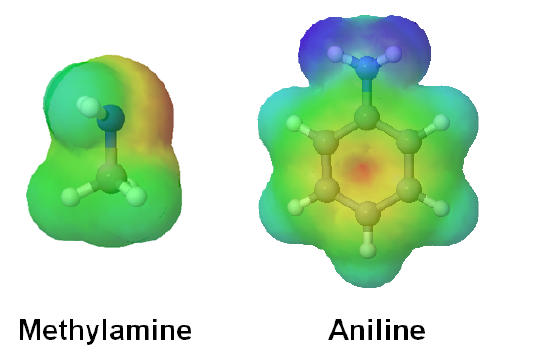
The effect of delocalization can be seen when viewing the electrostatic potential maps of aniline an methyl amine. The nitrogen of methyl amine has a significant amount of electron density on its nitrogen, shown as a red color, which accounts for it basicity compared to aniline. While the electron density of aniline's nitrogen is delocalized in the aromatic ring making it less basic.
Basicity of Substituted Arylamines
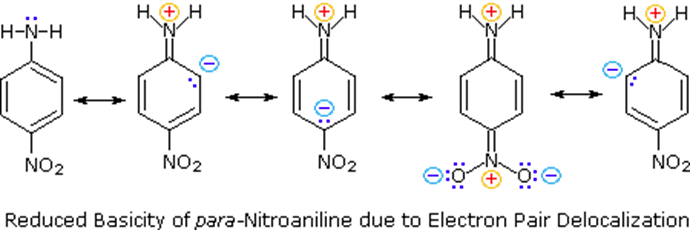
The addition of substituents onto the aromatic ring can can make arylamines more or less basic. Substituents which are electron-withdrawing (-Cl, -CF3, -CN, -NO2) decrease the electron density in the aromatic ring and on the amine making the arylamine less basic. In particular, the nitro group of para-nitroaniline allows for an additional resonance form to be drawn, which further stabilizes the lone pair electrons from the nitrogen, making the substituted arylamine less basic than aniline. This effect is analogous to the one discussed for the acidity of substituted phenols in Section 17.2.
Substituents which are electron-donating (-CH3, -OCH3, -NH2) increase the electron density in the aromatic ring and on the amine making the arylamine more basic. In the case of para-methoxyaniline, the lone pair on the methoxy group donates electron density to the aromatic system, and a resonance contributor can be drawn in which a negative charge is placed on the carbon adjacent to the nitrogen, which makes the substituted arylamine more basic than aniline.
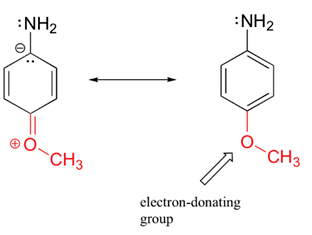
Increased Basicity of para-Methoxyaniline due to Electron-Donation
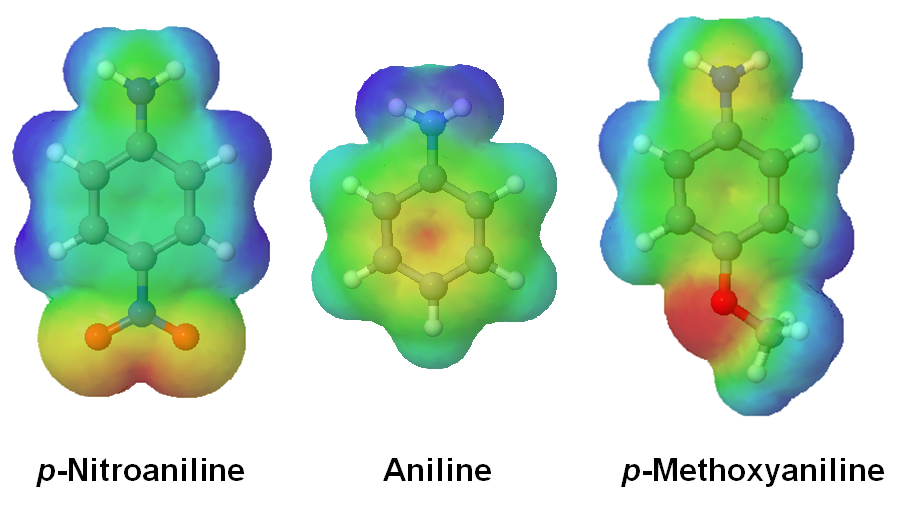
The shifting electron density of aniline, p-nitroaniline, and p-methoxyaniline are seen in their relative electrostatic potential maps. For p-Nitroaniline virtually all of the electron density, shown as a red/yellow color. is pulled toward the electron-withdrawing nitro group. In p-methoxyaninline the electron donating methoxy group donates electron density into the ring. The amine in p-methoxyaniline is shown to have more electron density, shown as a yellow color, when compared to the amine in aniline.
Exercise
Exercise 24.4.1
Using the knowledge of the electron donating or withdrawing effects of subsituents gained in Section 16.6, rank the following compound in order of decreasing basicity.
a) p-Nitroaniline, methyl p-aminobenzoate, p-chloroaniline
b) p-Bromoaniline, p-Aminobenzonitrile, p-ethylaniline
c) p-(Trifluoromethyl)aniline, p-methoxyaniline, p-methylaniline
- Answers
-
a) p-Chloroaniline, methyl p-aminobenzoate, p-nitroaniline
b) p-Ethylaniline, p-Bromoaniline, p-aminobenzonitrile
c) p-Methoxyaniline, p-methylaniline, p-(trifluoromethyl)aniline
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

