19.8 Nucleophilic Addition of Amines: Imine and Enamine Formation
- Page ID
- 90971
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- write equations to describe the reactions that occur between aldehydes or ketones and primary or secondary amines.
- identify the product formed from the reaction of a given aldehyde or ketone with a given primary or secondary amine.
- identify the aldehyde or ketone, the amine, or both, required in the synthesis of a given imine or enamine.
- write the detailed mechanism for the reaction of an aldehyde or ketone with a primary amine.
- write the detailed mechanism for the reaction of an aldehyde or ketone with a secondary amine.
- explain why the rate of a reaction between an aldehyde or ketone and a primary or secondary amine is dependent on pH.
Make certain that you can define, and use in context, the key terms below.
- 2,4‑dinitrophenylhydrozone
- enamine
- imine
An imine is a compound that contains the structural unit
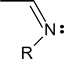
An enamine is a compound that contains the structural unit
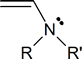
Both of these types of compound can be prepared through the reaction of an aldehyde or ketone with an amine.
You may have the opportunity to observe the reaction of an aldehyde and ketone with 2,4‑dinitrophenylhydrazine (Brady’s reagent) to form a 2,4‑dinitrophenylhydrozone in the laboratory. This is a classical organic chemistry test to confirm the presence of a carbonyl group. The reaction produces very colourful and bright precipitates of yellow, orange and red.
If you can understand why the two reactions of imine and enamine formation are essentially identical, and can write a detailed mechanism for each one, you are well on the way to mastering organic chemistry. If you understand how and why these reactions occur, you can keep the amount of material that you need to memorize to a minimum.
Reaction with Primary Amines to form Imines
The reaction of aldehydes and ketones with ammonia or 1º-amines forms imine derivatives, also known as Schiff bases (compounds having a C=N function). Water is eliminated in the reaction.
Predicting the Products of an Imine Forming Reaction
During imine formation, the carbonyl oxygen is completely removed. The nitrogen of the 1o amine reactant replaces the carbonyl oxygen to form the imine C=N bond. During the process the nitrogen of the 1o amine loses both of its hydrogens.
Examples of Imine Forming Reactions
Mechanism of Imine Formation
Imine formation is a reversible process that starts with the nucleophilic addition of a primary amine to the carbonyl group of an aldehyde or ketone. Next, a proton transfer forms a neutral amino alcohol called a carbinolamine. Acid protonation of the carbinolamine oxygen converts it into a better leaving group which is subsequently eliminated as water producing an iminium ion. Deprotonation of nitrogen gives the final imine product.
1) Nucleophilic addition
2) Protron transfer
3) Protonation
4) Water is eliminated to form an iminium ion.
5) Deprotonation
Reversibility of Imine Forming Reactions
Imines can be hydrolyzed back to the corresponding 1o amine under acidic conditions.
Reactions Involving other Reagents of the type Y-NH2
A wide variety of substances with −NH2 groups can react with aldehydes and ketones by an addition-elimination sequence to yield compounds with a carbon-nitrogen double bond. Imines are sometimes difficult to isolate and purify due to their sensitivity to hydrolysis. Consequently, other reagents of the type Y–NH2 have been studied, and found to give stable products (R2C=N–Y) useful in characterizing the aldehydes and ketones from which they are prepared. Some of these reagents are listed below, together with the structures and names of their carbonyl reaction products. Hydrazones are used as part of the Wolff-Kishner reduction and will be discussed in more detail in Section 19.9.
With the exception of unsubstituted hydrazones, these derivatives are easily prepared and are often crystalline solids - even when the parent aldehyde or ketone is a liquid. Since melting points can be determined more quickly and precisely than boiling points, derivatives such as these are useful for comparison and identification of carbonyl compounds. It should be noted that although semicarbazide has three nitrogen groups (–NH2) only one of them is a reactive amine. The other two are similar to amides and are deactivated by resonance with the adjacent carbonyl group.
Example
Cinnamaldehyde reacting with hydroxylamine to form cinnamaldehyde oxime
Acetone reacting with semicarbazide to form acetone semicarbazide
Reaction with Secondary Amines to form Enamines
Most aldehydes and ketones react with 2º-amines to give products known as enamines. It should be noted that, like acetal formation, these are acid-catalyzed reversible reactions in which water is lost. Consequently, enamines are easily converted back to their carbonyl precursors by acid-catalyzed hydrolysis.
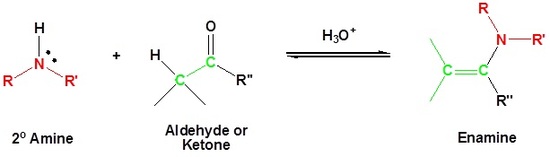
Mechanism
1) Nuleophilic attack
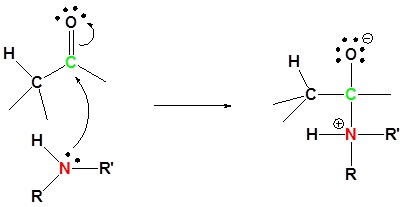
2) Proton transfer
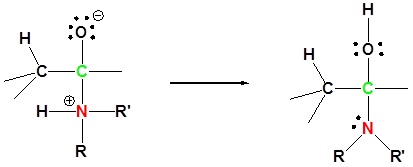
3) Protonation of OH
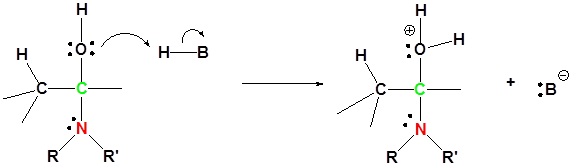
4) Removal of water
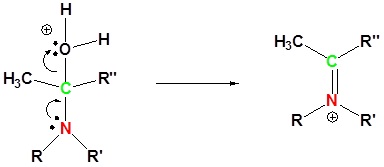
5) Deprotonation
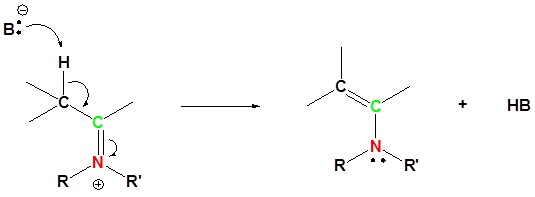
Reversibility of Enamines
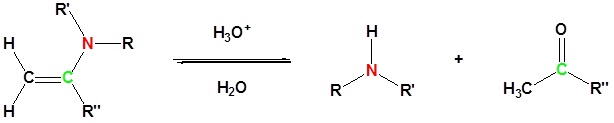
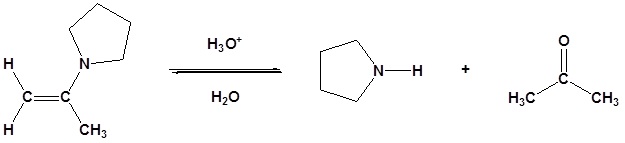
Exercises
Please draw the products of the following reactions.
- Answer
-
Please draw the structure of the reactant needed to produce the indicated product.
- Answer
-
Please draw the products of the following reaction.
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)

