6.2: Solutions
- Page ID
- 431291
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To understand what causes solutions to form.
A solution is another name for a homogeneous mixture. A mixture as a material composed of two or more substances. In a solution, the combination is so intimate that the different substances cannot be differentiated by sight, even with a microscope. Compare, for example, a mixture of salt and pepper and another mixture consisting of salt and water. In the first mixture, we can readily see individual grains of salt and the flecks of pepper. A mixture of salt and pepper is not a solution. However, in the second mixture, no matter how carefully we look, we cannot see two different substances. Salt dissolved in water is a solution.
The major component of a solution, called the solvent, is typically the same phase as the solution itself. Each minor component of a solution (and there may be more than one) is called the solute. It is the component dissolves in solution. For example, in a solution of salt in water, the solute is salt, and solvent is water.
Solutions come in all phases, and the solvent and the solute do not have to be in the same phase to form a solution (such as salt and water). For example, air is a gaseous solution of about 80% nitrogen and about 20% oxygen, with some other gases present in much smaller amounts. An alloy is a solid solution consisting of a metal (like iron) with some other metals or nonmetals dissolved in it. Steel, an alloy of iron and carbon and small amounts of other metals, is an example of a solid solution. Table \(\PageIndex{1}\) lists some common types of solutions, with examples of each.
| Solvent Phase | Solute Phase | Example |
|---|---|---|
| gas | gas | air |
| liquid | gas | carbonated beverages |
| liquid | liquid | ethanol (C2H5OH) in H2O (alcoholic beverages) |
| liquid | solid | saltwater |
| solid | gas | H2 gas absorbed by Pt metal |
| solid | liquid | Hg(ℓ) in dental fillings |
| solid | solid | steel alloys |
What causes a solution to form? The simple answer is that the solvent and the solute must have similar intermolecular interactions. When this is the case, the individual particles of solvent and solute can easily mix so intimately that each particle of solute is surrounded by particles of solute, forming a solution. However, if two substances have very different intermolecular interactions, large amounts of energy are required to force their individual particles to mix intimately, so a solution does not form.
Thus two alkanes like heptane, C7H16, and hexane, C6H14, are completely miscible in all proportions. The C7H16 and C6H14 molecules are both nonpolar, so they interact with each other via London's Dispersion Forces. They are however insoluble in polar water and hence hydrophobic (water-fearing).
For a similar reason, methanol, CH3OH, is completely miscible with water. In this case both molecules are polar and can form hydrogen bonds among themselves, and with each other. CH3OH dipoles can align with H2O dipoles, and CH3OH molecules can hydrogen bond to H2O molecules, and so methanol is hydrophilic (water-loving).
This process leads to a simple rule of thumb: like dissolves like. Solvents that are very polar will dissolve solutes that are very polar or even ionic. Solvents that are nonpolar will dissolve nonpolar solutes. Thus water, being polar, is a good solvent for ionic compounds and polar solutes like ethanol (C2H5OH). However, water does not dissolve nonpolar solutes, such as many oils, greases, and hydrocarbons.
We use the word soluble to describe a solute that dissolves in a particular solvent, and the word insoluble for a solute that does not dissolve in a solvent. Thus, we say that sodium chloride is soluble in water but insoluble in hexane (C6H14). If the solute and the solvent are both liquids and soluble in any proportion, we use the word miscible, and the word immiscible if they are not.
Water is considered a polar solvent. Which substances should dissolve in water?
- methanol (CH3OH)
- sodium sulfate (Na2SO4)
- octane (C8H18)
Solution
Because water is polar, substances that are polar or ionic will dissolve in it.
- Because of the OH group in methanol, we expect its molecules to be polar. Thus, we expect it to be soluble in water. As both water and methanol are liquids, the word miscible can be used in place of soluble.
- Sodium sulfate is an ionic compound, so we expect it to be soluble in water.
- Like other hydrocarbons, octane is nonpolar, so we expect that it would not be soluble in water.
Toluene (C6H5CH3) is widely used in industry as a nonpolar solvent. Which substances should dissolve in toluene?
- water (H2O)
- sodium sulfate (Na2SO4)
- octane (C8H18)
- Answer
-
Octane only.
Predict which of the following compounds will be most soluble in water:
- \(\underset{\text{Ethanol}}{\mathop{\text{CH}_{\text{3}}\text{CH}_{\text{2}}\text{OH}}}\,\)
- \(\underset{\text{Hexanol}}{\mathop{\text{CH}_{\text{3}}\text{CH}_{\text{2}}\text{CH}_{\text{2}}\text{CH}_{\text{2}}\text{CH}_{\text{2}}\text{CH}_{\text{2}}\text{OH}}}\,\)
Solution
Since ethanol contains an OH group, it can hydrogen bond to water. Although the same is true of hexanol, the OH group is found only at one end of a fairly large molecule. The rest of the molecule can be expected to behave much as though it were a nonpolar alkane. This substance should thus be much less soluble than the first. Experimentally we find that ethanol is completely miscible with water, while only 0.6 g hexanol dissolves in 100 g water.
Exercise \(\PageIndex{2}\)
Would I2 be more soluble in CCl4 or H2O?
- Answer
-
I2 is nonpolar. Of the two solvents, CCl4 is nonpolar and H2O is polar, so I2 would be expected to be more soluble in CCl4.
Electrolytes
A characteristic property of ionic compounds dissolved in water to form a solution is their electrical conductivity. The figure below shows three experiments in which two electrodes that are connected to a light bulb are placed in beakers containing three different substances.
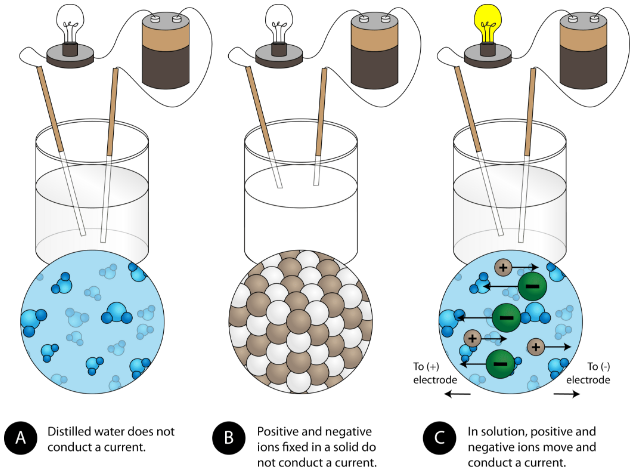
In the first beaker, distilled water does not conduct a current because water is a molecular compound. In the second beaker, solid sodium chloride also does not conduct a current. Despite being ionic and thus composed of charged particles, the solid crystal lattice does not allow the ions to move between the electrodes. Mobile charged particles are required for the circuit to be complete and the light bulb to light up. In the third beaker, the NaCl has been dissolved into the distilled water. Now the crystal lattice has been broken apart and the individual positive and negative ions can move. Cations move to one electrode, while anions move to the other, allowing electricity to flow. Ionic compounds conduct an electric current when dissolved in water. Such a solution is known as an electrolyte.
Key Takeaway
- Solutions form because a solute and a solvent experience similar intermolecular interactions.


