Physical Properties of Period 3 Oxides
- Page ID
- 3664
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page explains the relationship between the physical properties of the oxides of Period 3 elements and their structures (including sodium to chlorine; argon is omitted because it does not form an oxide).
A summary of the trends
The oxides:
| Na2O | MgO | Al2O3 | SiO2 | P4O10 | SO3 | Cl2O7 |
| P4O6 | SO2 | Cl2O |
The oxides in the top row are the highest known oxides of the various elements, in which the Period 3 elements are in their highest oxidation states. In these oxides, all the outer electrons in the Period 3 elements are involved in bonding.
- The structures: The metallic oxides on the left adopt giant structures of ions on the left of the period; in the middle, silicon forms a giant covalent oxide (silicon dioxide); the elements on the right form molecular oxides.
- Melting and boiling points: The large structures (the metal oxides and silicon dioxide) have high melting and boiling points because a large amount of energy is needed to break the strong bonds (ionic or covalent) operating in three dimensions. The oxides of phosphorus, sulfur and chlorine consist of individual molecules, simple or polymeric.
The attractive forces between these molecules include van der Waals dispersion and dipole-dipole interactions. These vary in size depending on the size, shape and polarity of the various molecules, but will always be much weaker than the ionic or covalent bonds in a giant structure. These oxides tend to be gases, liquids, or low melting point solids.
- Electrical conductivity: None of the oxides above have any free or mobile electrons, indicating that none of them will conduct electricity when solid. The ionic oxides can, however, undergo electrolysis in a molten state. They can then conduct electricity because of the movement of the ions towards the electrodes and the discharge of the ions when they reach the electrodes.
The metallic oxides
- The structures: Sodium, magnesium and aluminum oxides consist of giant structures containing metal ions and oxide ions. Magnesium oxide is similar in structure to sodium chloride. The other two oxides have more complicated possible arrangements.
- Melting and boiling points: There are strong attractions between the ions in each of the oxides above; breaking these attractions requires much heat energy. These oxides therefore have high melting and boiling points.
- Electrical conductivity: None of the metallic oxides conduct electricity in the solid state, but electrolysis is possible when molten. As discussed above, in a molten state, the oxides conduct electricity because of the movement and discharge of the ions present. The only industrially-important example of this process is the electrolysis of aluminum oxide in the manufacture of aluminum. Whether you can electrolyze molten sodium oxide depends on whether it actually melts (as opposed to subliming or decomposing under ordinary conditions). If it sublimes (experiences a solid-to-vapor transition) , no liquid can electrolyze. Magnesium and aluminum oxides have melting points that are too high for electrolysis under laboratory conditions.
Silicon dioxide (silicon(IV) oxide)
The structure: The electronegativity of the elements increases across the period; silicon and oxygen do not differ enough in electronegativity to form an ionic bond. There are three different crystal forms of silicon dioxide; the most convenient structure to visualize is similar to that of diamond. In silicon dioxide, oxygen atoms fill the empty spaces along the silicon-silicon bond axes, as shown in a representative structure below:
This structure is repeated many times throughout the silicon dioxide substance.
Melting and boiling points: Silicon dioxide has a high melting point that varies depending on the particular structure (the structure given is one of three possible structures), but each is close to 1700°C. Very strong silicon-oxygen covalent bonds must be broken throughout the structure before melting occurs. Silicon dioxide boils at 2230°C. Because two types of bonding are considered, it makes little sense to compare these values directly to those of the metallic oxides; suffice it to say that in both types of structures, the melting and boiling points are very high.
Electrical conductivity: Silicon dioxide has no mobile electrons or ions, and hence does not conduct electricity either as a solid or a liquid.
The molecular oxides
Phosphorus, sulfur and chlorine form molecular oxides. Some of these molecules are fairly simple—others are polymeric. Here the simple structures are considered. Melting and boiling points of these oxides are much lower than those of the metal oxides or silicon dioxide. The intermolecular forces binding one molecule to its neighbors are van der Waals dispersion forces or dipole-dipole interactions. The strength of these vary depending on the size of the molecules. None of these oxides conduct electricity either as solids or as liquids, because none of them contain ions or free electrons.
The phosphorus oxides
Phosphorus has two common oxides, phosphorus(III) oxide, P4O6, and phosphorus(V) oxide, P4O10.
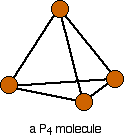
Phosphorus(III) oxide: Phosphorus(III) oxide is a white solid, melting at 24°C and boiling at 173°C. To understand its structure, consider a tetrahedral P4 molecule:

The structure is expanded to display the bonds:
The phosphorus-phosphorus bonds are interrupted with oxygen atoms, in a bent shape similar to water, as shown below:
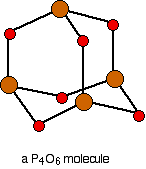
Only 3 of the valence electrons of phosphorus (the 3 unpaired p electrons) are involved in the phosphorus-oxygen bonds.
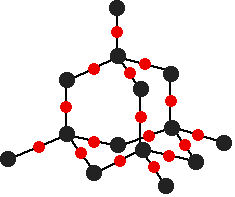
Phosphorus(V) oxide: Phosphorus(V) oxide is also a white solid, which sublimes at 300°C. In this case, the phosphorus uses all five of its outer electrons in the bonding. Solid phosphorus(V) oxide exists in several different forms - some of them polymeric. We are going to concentrate on a simple molecular form, and this is also present in the vapor. This is most easily drawn starting from P4O6. The other four oxygens are attached to the four phosphorus atoms via double bonds.
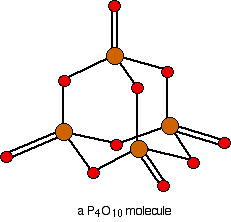
The sulfur oxides
Sulfur has two common oxides, sulfur dioxide (sulfur(IV) oxide), SO2, and sulfur trioxide (sulfur(VI) oxide), SO3.
Sulfur dioxide: S ulfur dioxide is a colorless gas at room temperature with an easily recognized rotten-egg smell. It consists of simple SO2 molecules as shown:
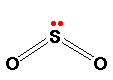
The sulfur uses four of its six valence electrons to form the double bonds with oxygen, leaving the other two as a lone pair on the sulfur. The bent shape of SO2 is due to this lone pair.
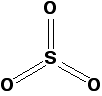
Sulfur trioxide: Pure sulfur trioxide is a white solid with a low melting and boiling point. It reacts very rapidly with water vapour in the air to form sulfuric acid. Under laboratory conditions, it forms a white sludge which fumes dramatically in moist air (forming a fog of sulfuric acid droplets). Gaseous sulfur trioxide consists of simple SO3 molecules in which all six of the sulfur's outer electrons are involved in the bonding.
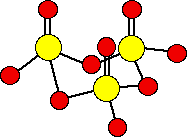
There are various forms of solid sulfur trioxide. The simplest one is a trimer, S3O9, in which three SO3 molecules are joined in a ring.
There are also other polymeric forms in which the SO3 molecules join together in long chains. For example:
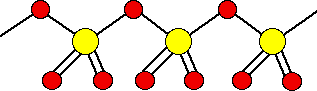
The tendency of sulfur trioxide to form polymers determines to its solid nature.
The chlorine oxides
Chlorine forms several oxides. Two are considered here: chlorine(I) oxide, Cl2O, and chlorine(VII) oxide, Cl2O7.
Chlorine(I) oxide: Chlorine(I) oxide is a yellowish-red gas at room temperature. It consists of simple, small molecules.

The physical properties of chlorine (I) oxide are consistent with those predicted for a molecule of its size.
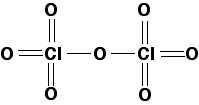
Chlorine(VII) oxide: In chlorine(VII) oxide, the chlorine involves all of its seven valence electrons in bonds with oxygen. This produces a molecule much larger than chlorine (I) oxide, suggesting higher melting and boiling points. Chlorine(VII) oxide is a colorless oily liquid at room temperature. The diagram below shows a simple structural formula, neglecting three-dimensional structure; the geometry is tetrahedral around both chlorine atoms, and V-shaped around the central oxygen.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


