Applications of Photoelectron Spectroscopy
- Page ID
- 1836
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Photoelectron spectroscopy (PES) is a technique used for determining the ionization potentials of molecules. Underneath the banner of PES are two separate techniques for quantitative and qualitative measurements. They are ultraviolet photoeclectron spectroscopy (UPS) and X-ray photoelectron spectroscopy (XPS). XPS is also known under its former name of electron spectroscopy for chemical analysis (ESCA). UPS focuses on ionization of valence electrons while XPS is able to go a step further and ionize core electrons and pry them away.
Photoelectron Instrumentation
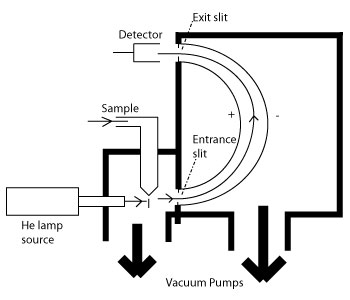
The main goal in either UPS or XPS is to gain information about the composition, electronic state, chemical state, binding energy, and more of the surface region of solids. The key point in PES is that a lot of qualitative and quantitative information can be learned about the surface region of solids. Specifics about what can be studied using XPS or UPS will be discussed in detail below in separate sections for each technique following a discussion on instrumentation for PES experiments. The focus here will be on how the instrumentation for PES is constructed and what types of systems are studied using XPS and UPS. The goal is to understand how to go about constructing or diagramming a PES instrument, how to choose an appropriate analyzer for a given system, and when to use either XPS or UPS to study a system.
There are a few basics common to both techniques that must always be present in the instrumental setup.
- A radiation source: The radiation sources used in PES are fixed-energy radiation sources. XPS sources from x-rays while UPS sources from a gas discharge lamp.
- An analyzer: PES analyzers are various types of electron energy analyzers
- A high vacuum environment: PES is rather picky when it comes to keeping the surface of the sample clean and keeping the rest of the environment free of interferences from things like gas molecules. The high vacuum is almost always an ultra high vacuum (UHV) environment.
Radiation sources
While many components of instruments used in PES are common to both UPS and XPS, the radiation sources are one area of distinct differentiation. The radiation source for UPS is a gas discharge lamp, with the typical one being an He discharge lamp operating at 58.4 nm which corresponds to 21.2 eV of kinetic energy. XPS has a choice between a monocrhomatic beam of a few microns or an unfocused non-monochromatic beam of a couple centimeters. These beams originate from X-Ray sources of either Mg or Al K-? sources giving off 1486 eV and 1258 eV of kinetic energy respectively. For a more versitile light source, synchrotron radiation sources are also used. Synchrotron radiation is especially useful in studying valence levels as it provides continuous, polarized radiation with high energies of > 350 eV.
The main thing to consider when choosing a radiation source is the kinetic energy involved. The source is what sets the kinetic energy of the photoelectrons, so there needs to not only be enough energy present to cause the ionizations, but there must also be an analyzer capable of measuring the kinetic energy of the released photoelectrons.
In XPS experiments, electron guns can also be used in conjunction with x-rays to eject photoelectrons. There are a couple of advantages and disadvantages to doing this, however. With an electron gun, the electron beam is easily focused and the excitation of photoelectrons can be constantly varied. Unfortunately, the background radiation is increased significantly due to the scattering of falling electrons. Also, a good portion of substances that are of any experimental interest are actually decomposed by heavy electron bombardment such as that coming from an electron gun.
Analyzers
There are two main classes of analyzers well-suited for PES - kinetic energy analyzers and deflection or electrostatic analyzers. Kinetic energy analyzers have a resolving power of \(E/\delta{E}\), which means the higher the kinetic energy of the photoelectrons, the lower the resolution of the spectra. Deflection analyzers are able to separate out photoelectrons through an electric field by forcing electrons to follow different paths according to their velocities, giving a resolving power, \(E/\delta{E}\), that is greater than 1,000.
Since the resolving power of both types of analyzer is \(E/\delta{E}\), the resolution is directly dependent on the kinetic energy of the photoelectrons. The intensity of the spectra produced is also dependent on the kinetic energy. The faster the electrons are moving, the lower the resolution and intensity is. In order to actually get well resolved, useful data other components must be introduced into the instrument.
Adding a system of optics (lenses) to a PES instrument helps with this problem immensely. Electron optics are capable of decelerating the photoelectrons through retardation of the electric field. The energy the photoelectrons decelerate to is known as the "pass energy." This has the benefit of significantly raising the resolution, however this does, unfortunately, lower the sensitivity. Optics are also capable of accelerating the electrons as well. The design of any lens system greatly effects the photoelectron counts. These lenses are also capable of focusing on a small area of a particular sample.
Specific Analyzers
Within the broad picture of two main analyzer classes, there are a variety of specific analyzers in existence that are used in PES. The list below goes over several well-used analyzers, though this list is, by no means, exhaustive. The most common type of analyzer is a hemispherical analyzer, which will be explained in more depth under the spherical deflection analyzer topic.
Plane Mirror Analyzer (PMA)
PMAs, the simplest type of electric analyzer are also known as parallel-plate mirror analyzers. These analyzers are condensers made from two parallel plates with a distance, d, across them. Parabolic trajectories of electrons are obtained due to the constant potential difference, V, between the two plates.
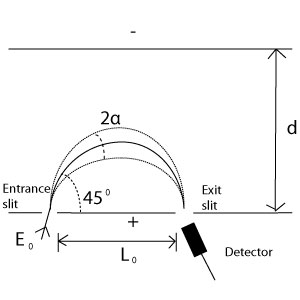
In order for transmission to occur, the potential must be: V=Eod/eLo. Eo=kinetic energy of electron in eV and e=charge of the electron. To obtain better focus, the electron entrance and exit angle is capable of being shifted to 30 degrees, but this is not necessarily a good idea as it sacrifices transmission instead.
Cylindrical Mirror Analyzer (CMA)
CMAs are advantageous over PMAs. They employ 2? geometry to overcome the low transmission with a PMA. A CMA consists of two cylinders having a potential difference, V, between them. The entrance and exit slits are all contained on the inner cylinder.
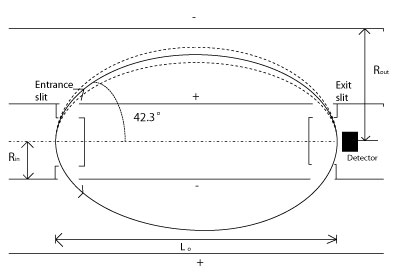
Here: V=1.3E0 ln(Rout/Rin) where L0=6.1(Rin) and E0 is in volts.
They are good for applications that require a high sensitivity with only a moderate resolution.
Cylindrical Deflection Analyzer (CDA)
CDAs consist of two cylinders spanning a 127 degree angle. It is this reason that CDAs are sometimes called "127 degree analyzers."
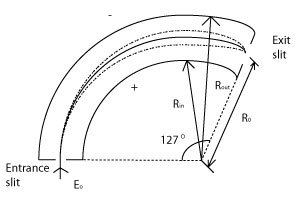
The potential difference in a CDA is: 2V=E0(Rin/Rout) where E0 is the energy of incoming photoelectrons, in eV, that are focused.
These analyzers have high resolution, however their transmission is low.
Spherical Deflection Analyzer (SDA)
SDAs are similar to CDAs, but they consist of two concentric hemispheres instead. In an SDA, the transmission of photoelectrons with initial energy, E0, occurs along a path where R0=(Rin/Rout)/2. Since SDAs are the most common, prevalent type of PES analyzer, they will be discussed in more depth than any of the previous analyzers as a thorough understanding of how they apply to PES is, theoretically, of greater importance.
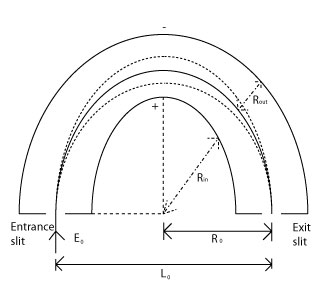
Here, the potential is different for both the inner and the outer hemisphere:
\[V_{in}=E_0[3-2(R_0/R_{in})]\]
and
\[V_{out}=E_0[3-2(R_0/R_{out})]\]
The resolving power of these analyzers is proportional to the radius of the inner and outer hemispheres. These analyzers are also capable of running in two separate modes when coupled with an optical system - fixed analyzer transmission mode (FAT) and fixed retardation energy mode (FRR). In FAT mode, the lens either retards or accelerates the electrons so that all photoelectrons enter the analyzer with the same kinetic energy. For this to occur, the analyzer is also arranged so that only photoelectrons of a specific, fixed kinetic energy will pass through and reach the detector. In this case, the lens is scanned for different energies. In FRR mode, the lens only retards the photoelectrons, and it does so in a uniform manner causing all photoelectrons to be reduced in energy to a fixed value such as 15 eV, 30 eV, or whatever energy is desired. The hemispheres of the analyzer here have a potential difference between them that is varied so that photoelectrons of different kinetic energies can reach the detector. The more common of these two modes is FAT because it provides a greater signal intensity at low electron kinetic energy and is also makes quantification of the spectra simpler.
These analyzers are a particularly good class of deflection analyzer. The slits in an SDA define the acceptable range of entrance and exit trajectories a photoelectron may have when entering or leaving the analyzer. The photoelectrons that do make it through the entrance slits will then only exit if they follow a specific, curved path down the middle of the two hemispheres. The path they follow has the "correct energy" for exit to occur, and is determined by the selection of Vin and Vout. Photoelectrons that are of higher or lower kinetic energy than what is defined by the hemispheres will be lost through collisions with the walls.
Detection & Spectra
Detection relies on the ability of the instrument to measure energy and photoelectron output. One type of energy measured is the binding energy, which is calculated through the following equation:
\[ K_e = h\nu - BE - \phi \]
where:
- Ke= Kinetic energy, this is measured
- \( h\nu \) = Photon energy from the radiation source, this is controlled by the source
- \( \phi \)= Work function of the spectrometer, this is found through calibration
- BE= Binding energy, this is the unknown of interest and can be calculated from the other three variables
Another part of PES detection is in the use of electron multipliers. These devices act as electron amplifiers because they are coated with a material that produce secondary photoelectrons when they are struck by an electron. Typically, they are able to produce two to three photoelectrons per every electron they are hit with. Since the signals in PES are low, the huge amplification, up to 107 and higher when run in series so the secondary electrons from one multiplier strike the next, they greately improve the signal strength from these instruments.
One type of spectra in these experiments is recorded by varying the potential difference between the plates or hemispheres of the analyzer. The output is known as an electron kinetic energy spectrum and is obtained by measuring the photoelectron current at the detector as a function of the voltage applied to the hemispheres or plates. The voltage is then used in the calculation of kinetic energy.
Further detail on the spectra produced in PES experiments and the analysis of said spectra is planned for a future module on the interpreation of photoelectron spectroscopy.
Limitations
The main limitation in a PES instrument is the resolution. The problems in resolution come from four main areas: the dimensions of the analyzer, the widths of the entrance and exit slits, other charges such as outside electronic fields or outside magnetic fields, and local charges inside the instrument itself arising from things such as contamination in the analyzer. Steps can be taken to improve the resolution, but some methods then sacrifice other factors such as the sensitivity. Obtaining high spatial resolution and high energy resolution always comes at the expense of the signal intensity.
One resolution improving technique that, then, messes with the sensitivity is changing the width of the entrance and/or exit slits. For example, in an SDA these slits are what define the range of trajectories photoelectrons may have when entering or exiting the analyzer. Decreasing the widths will certainly cause the resolution to go up, but the smaller slit size will decrease the number of photoelectrons allowed in and out of the analyzer, therefore lowering the sensitivity.
Another technique which was discussed above given it's relevance to the discussion on analyzers, is the addition of electron optics to the instrument.
A third method of improving resolution is specific to XPS and is the addition of an x-ray monochromator to the system. These monochromators eliminate satellite radiation from x-rays and are capable of narrowing the x-ray line width from ~1eV to ~0.2eV. The use of monochromatic x-rays also serves to simplify the spectrum.
Ultraviolet Photoelectron Spectroscopy - UPS
In early UPS, the sample was a gas or a vapor that is irradiated with a narrow beam of UV radiation. More modern UPS instruments are now capable of studying solids as well. The photoelectrons produced are passed through a slit into a vacuum region where they are then deflected by magnetic or electrostatic fields to give an energy spectrum. UPS is sensitive to the very near surface region, up to around 10 nm in depth.
There are two main areas UPS is used to study:
1. Electronic structure of solids
2. Adsorbed molecules on metals
Specific examples of UPS studies include:
1. The measurement of molecular orbital energies that can be compared to theortical values calculated from quantum chemistry
2. Determination and assignment of bonding, nonbonding, and/or antibonding molecular orbitals
3. The binding and orientation of adsorbed species on the surface of solids
4. Band structure mapping in k-space with angle-resolved techniques
Spectral output
Briefly, the spectrum produced from a UPS experiment has peaks that correspond to the ionization potentials of the molecule. These also correspond to the orbital energies. Because of this, UPS can also give information on the vibrational energy levels of the ions formed.
Limitations
UPS is capable only of ionizing valence electrons, which limits the range and depth of UPS surface experiments. Conventional UPS has relatively poor resolution.
Advantages
Ultraviolet radiation has a very narrow line width and a high flux of photons available from simple discharge sources. Higher resolution UPS scans allow for the observation of the fine structures that are due to vibrational levels of the molecular ion which, then, allows molecular orbital assignment of specific peaks.
X-Ray Photoelectron Spectroscopy - XPS
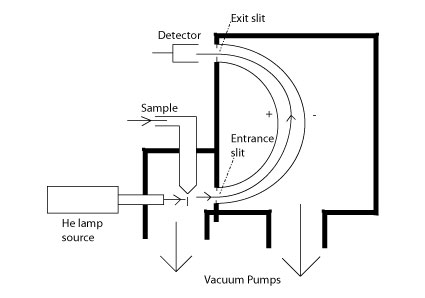
A diagram of a typical XPS instrument was shown at the beginning in figure 1. XPS is extremely good for surfaces. This is because the kinetic energy of the escaping photoelectrons limits the depth able to be probed. The samples studied are all solids of some type ranging from metals to frozen liquids. When the sample is irradiated, the electrons ejected are from the inner shells of the atoms.
There are several areas suited to measurement by XPS:
1. Elemental composition
2. Empirical formula determination
3. Chemical state
4. Electronic state
5. Binding energy
6. Layer thickness in the upper portion of surfaces
Some specific examples of systems studied by XPS are:
- Analysis of stains and residues on surfaces
- Reactive frictional wear of solid-solid reactions
- Silicon oxynitried thickness and measurements of dosage
- Depth profiling: In depth profiling, a sputter source is used. This removes successive layers from the surface of a sample and allows for the quanitation of element depth profiles to be recorded in the near-surface region. This is useful in the composition of thin films.
- Angle dependence measurements: When the angle of measurement is changed, the depth of the information gathered can be varied by 1-10 nm. The usefulness here is in determining the concentration of additives in the surface region.
- Imaging of surfaces
- Utilizing a special imaging mode, the distribution of elements in surface structures can be determined. This technique is useful in dimensions up to about 3 um.
Spectral output
Briefly, the spectrum from an XPS experiment is a graph of emission intensity vs binding energy. This allows elements on the surface to be identified based on the unique binding energy each element has. The peak areas on these spectra can also be used to obtain the concentration of the elements on the surface as well. Detailed information on the interpretation of XPS spectra is planned for a future module.
Limitations
Despite the many benefits to XPS, nothing is foolproof, nor is anything without limitations. The smallest analytical area XPS can measure is ~10 um. Samples for XPS must be compatible with the ultra high vacuum environment. Because XPS is a surface technique, there is a limited amount of organic information XPS can provide. XPS is limited to measurements of elements having atomic numbers of 3 or greater, making it unable to detect hydrogen or helium. XPS spectra also take a long time to obtain. The use of a monochromator can also reduce the time per experiment.
Advantages
XPS has a greater range of potential application than UPS since it can probe down to core electrons. XPS is good for identifying all but two elements, identifying the chemical state on surfaces, and is good with quantitative analysis. XPS is capable of detecting the difference in chemical state between samples. XPS is also able to differentiate between oxidations states of molecules.
UPS vs. XPS
One question that is always of consideration when multiple techniques are available for use is which technique is the best for the system or sample of interest.
Here is a brief table of some of the experimental applications of PES and the suitability to either XPS, UPS, or both.
| Application | XPS | UPS |
|---|---|---|
| Materials on surfaces | X | X |
| Depth profiling | X | |
| Angle dependent studies | X | X |
| Binding energy | X | X |
| Valence band fine structure | X | |
| Elemental composition | X | |
| Empirical formulas | X | |
| Electron energy levels | X | X |
References
- Hufner, Stefan. Photoelectron Spectroscopy: Principles and Applications. Springer 2003
- Skoog. Principles of Instrumental Analysis. Brooks/Cole
- Oxford Dictionary of Chemistry, 6th ed.. Oxford University Press
- Somasundaran, P. ed. Encyclopedia of Surface and Colloid Science. Taylor and Francis
- Ellis, Andrew M.; Feher, Miklos; Wright, Timothy G. Electronic and Photoelectron Spectroscopy: Fundamentals and Case Studies. Cambridge University Press. 2005
Problems
- What are the limitations involved with PES analyzers?
- Is it possible to obtain both high sensitivity and high resolution with XPS? Why or why not?
- Name three methods for improving the signal output from a PES instrument.
- Can you study a system using both UPS and XPS? What are the advantages to using both techniques? Are there any disadvantages?
- Why is the SDA the most widely used analyzer for PES experiments?

