Overlap Integral
- Page ID
- 64948
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The Overlap Integral is a quantitative measure of the overlap of atomic orbitals situated on different atoms. An orbital overlap is the amount of orbitals of adjacent atoms that are in the same regions of space. The overlap of the atomic orbital of an atom A and the atomic orbital of an atom B is called their overlap integral. It is defined as \(S_{AB} = \int_{}^{} \psi^{*}_{A} \psi_{B} dr\) extending over all space.
Basic description
To understand the overlap integral, consider the wave function \(\psi_{+}\) to calculate the energy of \(H^{+}_{2}\) as a function of internuclear separation, R.
The energy associated with \(\psi_{+}\), E+ has the equation
\[\hat{H} \psi_{+} (r; R) = E_{+} \psi_{+} (r; R)\]
Multiply the left by \(\psi^{*}_{+} (r;R)\) and then integrate over the allowed values of r to get E+
\[E_{+} = \dfrac{\int_{}^{} dr \psi^{*}_{+} \hat{H} \psi_{+}}{\int_{}^{} dr \psi^{*}_{+} \psi_{+}}\]
The wave function \(\psi_{+}\) is given by \(\psi_{+} = c(1s_{A} + 1s_{B})\). To normalize this wave function, \(\int_{}^{} dr \psi^{*}_{+} \psi_{+} = 1\), so
\[\int_{}^{} dr \psi^{*}_{+} \psi_{+} = \int_{}^{} dr (1s^{*}_{A} + 1s^{*}_{B})(1s_{A} + 1s_{B})\]
\[\int_{}^{} dr \psi^{*}_{+} \psi_{+} = \int_{}^{} dr (1s^{*}_{A}1s_{A} + 1s^{*}_{A}1s_{B} + 1s^{*}_{B}1s_{A} + 1s^{*}_{B}1s_{B})\]
\[\int_{}^{} dr \psi^{*}_{+} \psi_{+} = (\int_{}^{} dr 1s^{*}_{A}1s_{A} + \int_{}^{} dr 1s^{*}_{A}1s_{B} + \int_{}^{} dr 1s^{*}_{B}1s_{A} + \int_{}^{} dr 1s^{*}_{B}1s_{B})\]
The first and fourth integrals are simply the normalization expressions of the hydrogen atomic orbitals, so
\[\int_{}^{} dr 1s^{*}_{A}1s_{A} = \int_{}^{} dr 1s^{*}_{B}1s_{B} = 1\]
The second and third integrals involve the product of the hydrogen atomic orbital focus on nucleus A and the hydrogen atomic orbital focus on nucleus B. This product is larger when the two atomic orbitals have a larger overlap. These two integrals equal each other because the hydrogen atomic 1s orbital is expressed by a real function, or \(1s^{*} = 1s\), and S is the overlap integral, so
\[\int_{}^{} dr 1s^{*}_{A}1s_{B} = \int_{}^{} dr 1s^{*}_{B}1s_{A} = S\]
This then gives
\[\int_{}^{} dr \psi^{*}_{+} \psi_{+} = 1 + S + S + 1\]
\[\int_{}^{} dr \psi^{*}_{+} \psi_{+} = 2 + 2S\]
Thus, the denominator is
\[\int_{}^{} dr (1s^{*}_{A} + 1s^{*}_{B})(1s_{A} + 1s_{B}) = 2 + 2S\]
The wave function \(\psi_{+}\) is given by \(\psi_{+} = c(1s_{A} + 1s_{B})\). To normalize this wave function, \(\int_{}^{} dr \psi^{*}_{+} \psi_{+} = 1\), so
\[1 = \int_{}^{} dr \psi^{*}_{+} \psi_{+}\]
\[1 = \int_{}^{} dr \space c(1s^{*}_{A} + 1s^{*}_{B}) c(1s_{A} + 1s_{B})\]
\[1 = c^{2} \int_{}^{} dr (1s^{*}_{A} + 1s^{*}_{B})(1s_{A} + 1s_{B})\]
\[1 = c^{2} \int_{}^{} dr (1s^{*}_{A}1s_{A} + 1s^{*}_{A}1s_{B} + 1s^{*}_{B}1s_{A} + 1s^{*}_{B}1s_{B})\]
\[1 = c^{2} (\int_{}^{} dr 1s^{*}_{A}1s_{A} + \int_{}^{} dr 1s^{*}_{A}1s_{B} + \int_{}^{} dr 1s^{*}_{B}1s_{A} + \int_{}^{} dr 1s^{*}_{B}1s_{B})\]
Using the concepts of the Overlap Integral, \(\int_{}^{} dr 1s^{*}_{A}1s_{A} = \int_{}^{} dr 1s^{*}_{B}1s_{B} = 1\) and \(\int_{}^{} dr 1s^{*}_{A}1s_{B} = \int_{}^{} dr 1s^{*}_{B}1s_{A} = S\), so
\[1 = c^{2} (1 + S + S + 1)\]
\[1 = c^{2} (2 + 2S)\]
\[1 = c^{2} 2(1 + S)\]
\[c^{2} = \dfrac{1}{2(1 + S)}\]
\[c = \sqrt{\dfrac{1}{2(1 + S)}} = \dfrac{1}{\sqrt{2(1 + S)}}\]
Thus, the normalized wave function is
\[\psi_{+} = c(1s_{A} + 1s_{B})\]
\[\psi_{+} = \dfrac{1}{\sqrt{2(1 + S)}} (1s_{A} + 1s_{B})\]
Advanced description
If the wave functions do not overlap, then the overlap integral is zero. The integral can also be zero if the wave functions have positive and negative aspects that cancel out. If the overlap integral is zero, then the wave functions are called orthogonal. As the distance of the functions get closer to R = 0, then the overlap integral gets closer to the maximum overlap integral of S = 1.
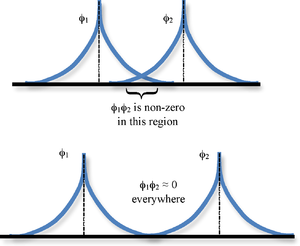
Overall, the full equation for the overlap integral, which is hard to calculate but is explained here, is
\[S(R)= \left \langle 1s_A | 1s_B \right \rangle = e^{-R/a_0} \left (1 + \dfrac{R}{a_0} + \dfrac{R^2}{3a_0^2} \right)\]
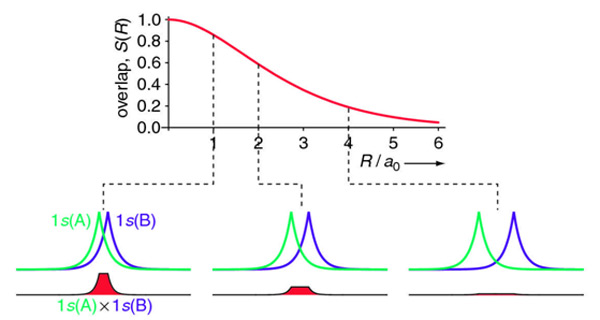
\[S(R)= e^{-R/a_0} \left( 1 +\dfrac{R}{a_0} + \dfrac{R^2}{3a_0^2} \right)\]
Calculating the different separations of Figure 1.2 above:
At \(\dfrac{R}{a_0}\) = 0,
\[S(0) = e^{-0} (1 + 0 + \dfrac{0^{2}}{3}) = e^{0}(1 + 0 + 0) = 1(1) = 1\]
At \(\dfrac{R}{a_0}\) = 1,
\[S(1) = e^{-1} (1 + 1 + \dfrac{1^{2}}{3}) = e^{-1}(1 + 1 + \dfrac{1}{2}) = e^{-1}(\dfrac{5}{2}) = 0.9197\]
At \(\dfrac{R}{a_0}\) = 2,
\[S(2) = e^{-2} (1 + 2 + \dfrac{2^{2}}{3}) = e^{-2}(1 + 2 + \dfrac{4}{2}) = e^{-2}(1 + 2 + 2) = e^{-2}(5) = 0.6767\]
At \(\dfrac{R}{a_0}\) = 4,
\[S(4) = e^{-4} (1 + 4 + \dfrac{4^{2}}{3}) = e^{-4}(1 + 4 + \dfrac{16}{2}) = e^{-4}(1 + 2 + 8) = e^{-4}(11) = 0.2015\]
As evident from these calculations, the distance of R/a0 = 0 or R = 0 gives the maximum overlap integral of S = 1. After that, the overlap integral S decreases exponentially with distance R.
Contributors and Attributions
- Marilyn McBryan

