Fundamentals of Phase Transitions
- Page ID
- 1536
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Phase transition is when a substance changes from a solid, liquid, or gas state to a different state. Every element and substance can transition from one phase to another at a specific combination of temperature and pressure.
Phase Changes
Each substance has three phases it can change into; solid, liquid, or gas(1). Every substance is in one of these three phases at certain temperatures. The temperature and pressure at which the substance will change is very dependent on the intermolecular forces that are acting on the molecules and atoms of the substance(2). There can be two phases coexisting in a single container at the same time. This typically happens when the substance is transitioning from one phase to another. This is called a two-phase state(4). In the example of ice melting, while the ice is melting, there is both solid water and liquid water in the cup.
There are six ways a substance can change between these three phases; melting, freezing, evaporating, condensing, sublimination, and deposition(2). These processes are reversible and each transfers between phases differently:
- Melting: The transition from the solid to the liquid phase
- Freezing: The transition from the liquid phase to the solid phase
- Evaporating: The transition from the liquid phase to the gas phase
- Condensing:The transition from the gas phase to the liquid phase
- Sublimination: The transition from the solid phase to the gas phase
- Deposition: The transition from the gas phase to the solid phase
How Phase Transition works
There are two variables to consider when looking at phase transition, pressure (P) and temperature (T). For the gas state, The relationship between temperature and pressure is defined by the equations below:
Ideal Gas Law:
\[ PV=nRT\]
van der Waals Equation of State:
\[ \left(P+a*\frac{n^2}{V^2}\right)\left(V-nb\right)=nRT\]
Where V is volume, R is the gas constant, and n is the number of moles of gas.
The ideal gas law assumes that no intermolecular forces are affecting the gas in any way, while the van der Waals equation includes two constants, a and b, that account for any intermolecular forces acting on the molecules of the gas.
Temperature
Temperature can change the phase of a substance. One common example is putting water in a freezer to change it into ice. In the picture above, we have a solid substance in a container. When we put it on a heat source, like a burner, heat is transferred to the substance increasing the kinetic energy of the molecules in the substance. The temperature increases until the substance reaches its melting point(2). As more and more heat is transferred beyond the melting point, the substance begins to melt and become a liquid(3). This type of phase change is called an isobaric process because the pressure of the system stays at a constant level.
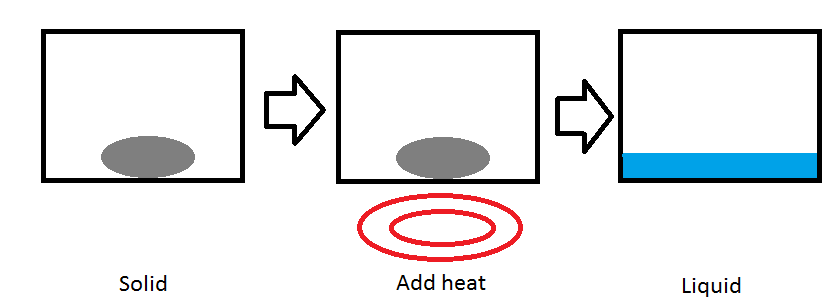
Melting point (Tf)
Each substance has a melting point. The melting point is the temperature that a solid will become a liquid. At different pressures, different temperatures are required to melt a substance. Each pure element on the periodic table has a normal melting point, the temperature that the element will become liquid when the pressure is 1 atmosphere(2).
Boiling Point (Tb)
Each substance also has a boiling point. The boiling point is the temperature that a liquid will evaporate into a gas. The boiling point will change based on the temperature and pressure. Just like the melting point, each pure element has a normal boiling point at 1 atmosphere(2).
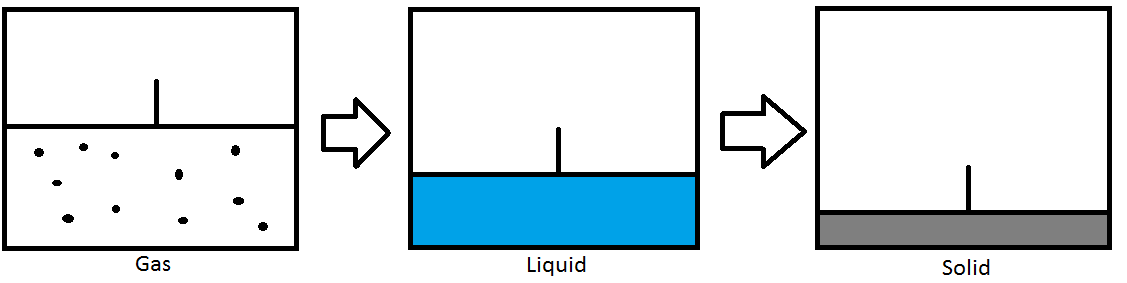
Pressure
Pressure can also be used to change the phase of the substance. In the picture above, we have a container fitted with a piston that seals in a gas. As the piston compresses the gas, the pressure increases. Once the boiling point has been reached, the gas will condense into a liquid. As the piston continues to compress the liquid, the pressure will increase until the melting point has been reached. The liquid will then freeze into a solid. This example is for an isothermal process where the temperature is constant and only the pressure is changing.
A Brief Explanation of a Phase Diagram
Phase transition can be represented with a phase diagram. A phase diagram is a visual representation of how a substance changes phases.
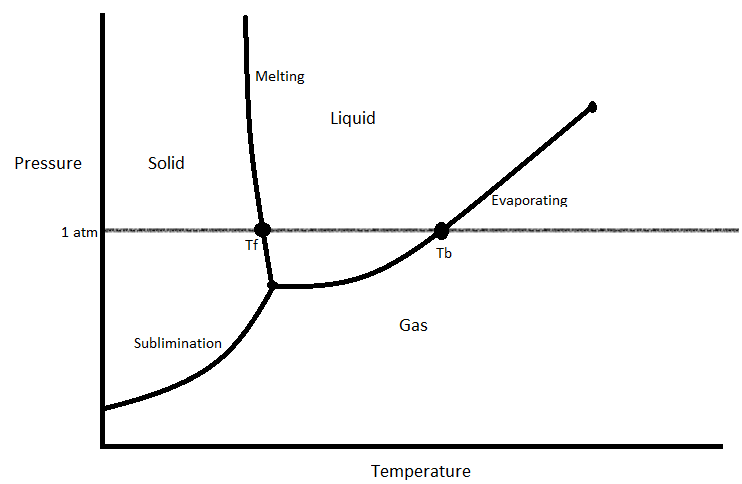
This is an example of a phase diagram. Often, when you are asked about a phase transition, you will need to refer to a phase diagram to answer it. These diagrams usually have the normal boiling point and normal melting point marked on them, and have the pressures on the y-axis and temperatures on the x-axis. The bottom curve marks the temperature and pressure combinations in which the substance will subliminate (1). The left left marks the temperature and pressure combinations in which the substance will melt (1). Finally, the right line marks the conditions under which the substance will evaporate (1).
References
- Olander, Donald R. General Thermodynamics. Boca Raton: CRC, 2008.
- Oxtoby, David W., H. P. Gillis, and Alan Campion. "Phase Transition." Principles of Modern Chemistry. 6th ed. Singapore: Thomson/Brooks/Cole, 2008. 428-30.
- Schmidt, Philip S. Thermodynamics: an Integrated Learning System. Hoboken, NJ: Wiley, 2006.
- Sherwin, Keith. Introduction to Thermodynamics. London: Chapman & Hall, 1994.
Problems
1. Using the phase diagram for carbon dioxide below, explain what phase carbon dioxide is normally in at standard temperature and pressure, 1 atm and 273.15 K.

Phase diagram for CO2.from Wikipedia.
2: Looking at the same diagram, we see that carbon dioxide does not have a normal melting point or a normal boiling point. Explain what kind of a change carbon dioxide makes at 1 atm and estimate the temperature of this point.
Solutions
1: Before we can completely answer the question, we need to convert the given information to match the units in the diagram. First we convert 25 degrees Kelvin into Celsius: \(K=273.15+C\) \[ 298.15-273.25C\] Now we can look at the diagram and determine its phase. At 25 degrees Celsius and 1 atm carbon dioxide is in the gas phase.
2: Carbon dioxide sublimes at 1 atm because it transitions from the solid phase directly to the gas phase. The temperature of sublimation at 1 atm is about -80 degrees Celsius.
Contributors and Attributions
- Kirsten Amdahl (UC Davis)

