5.1: Q-NMR for purity determination of macrolide antibiotic reference standards- Comparison with the mass balance method
- Page ID
- 77797
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
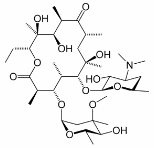
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Macrolide antibiotics are derived from microbial fermentations. As a class the macrolide antibiotics contain a 14- or 16-membered macrocyclic lactone, containing amino groups and deoxy sugars. The antibiotic activity of these compounds results from the inhibition of bacterial protein synthesis. Because macrolides accumulate within leukocytes, they are transported to the site of infection. Some macrolides also have immunomodulatory effects that can reduce inflammation. One of the best known macrolide antibiotics is erythromycin, the structure of which is shown here.
This application examines the use of quantitative 1H NMR measurements for determining the purity of macrolide antibiotic reference standards. Samples of clarithrmycin, roxithromycin, azithrimycin, dirithromycin and midecamycin were tested. The Q-NMR experiments were performed at 500 MHz using 1,4-dinitrobenzene as an internal standard. Rather than determining T1 relaxation times, the relaxation delay (64 s) was selected by comparing the peak area for the internal standard using d1 delays of 1, 5,10, 20, 32, 64 and 256 s.
The Q-NMR results were compared with the more conventional approach of determining mass balance, shown in Table 1 below. In mass balance determinations, the content is calculated as shown in Equation \(\ref{E1}\).
\[\mathrm{Content\: \% = (1 - impurity\%)(1 - water\% - volatile\: material\: \% - sulfated\: ash\%) \times 100} \label{E1}\]
In this determination the % impurity is determined by HPLC-UV. Water content is determined by Karl Fischer titration and residual solvents are measured using gas chromatography. The amount of sulfated ash (primarily a European designation) corresponds to the amount of residue remaining after ignition of the sample. One of the drawbacks of HPLC-UV for these measurements is the lack of chromophore with a characteristic absorption wavelength. In addition, a general problem with this approach is that the UV absorption coefficient of each impurity is different, and in many cases unknown.
Purity determinations using NMR rely on the comparison of the integrals measured for analyte’s resonances (Ix) with the integral of a quantitation standard (Istd). In this case an internal standard was used. Because the masses of the sample (mx) and the internal standard (mstd) are known, the content of the analyte can be determined. Note that the underlying assumption in this method is that resonances of impurities do not overlap with the resonances used to measure the analyte content. The purity of the analyte, Px, is calculated as shown in Equation \(\ref{E2}\):
\[P_x = \dfrac{I_x}{I_{std}} \dfrac{N_{std}}{N_x} \dfrac{M_x}{M_{std}} \dfrac{m_{std}}{m_x} P_{std} \label{E2}\]
where Mx and Mstd are the molar masses of the analyte and the standard, respectively, Pstd is the purity of the standard, and Nstd and Nx are the number of spins responsible for the integrated standard and analyte signals, respectively. The content results, summarized in Table 1, for the macrolide antibiotics obtained with 1H NMR and by the mass balance method are in good agreement. The main source of uncertainty in the Q-NMR method was in the sample weight, about 15 mg in these experiments. Use of a larger mass would decrease the uncertainty in the results but would consume larger amounts of deuterated solvents. An informative feature of this report is the detailed error analysis it contains.
| 1H Q-NMR method (%) | Mass balance method (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Average Contenta | RSD | Uexpanded | Impurity/u1 | Water/u2 b | Residual Solvents/u3 b | Sulfated ash/u4 b | Content/ Uexpanded | |
| Clarithromycin | 96.3 | 0.49 | 1.89 | 3.35/0.0299 | 1.4/0.0148 | <0.001/<8.4 x 10-6 | 0/0 | 95.3/2.64 |
| Roxithromycin | 95.7 | 0.44 | 1.82 | 2.50/0.0223 | 2.2/0.0233 | <0.001/<8.4 x 10-6 | 0/0 | 95.4/2.64 |
| Azithromycin | 94.3 | 0.50 | 1.36 | 1.59/0.0142 | 4.5/0.0486 | <0.001/<8.4 x 10-6 | 0.02/0.0198 | 94.0/2.75 |
| Dirithromycin | 96.9 | 0 | 1.81 | 3.30/0.0295 | 0.6/0.00636 | <0.001/<8.4 x 10-6 | 0/0 | 96.1/2.67 |
| Midecamycin | 97.1 | 2.0 | 1.96 | 3.94/0.0352 | 0.16/0.000035c | 0/0 | 95.9/1.71 | |
|
||||||||
Reference
Liu, S.-Y.; Hu, C.-Q. “A comparative uncertainty study of the calibration of macrolide antibiotic reference standards using quantitative nuclear magnetic resonance and mass balance methods” Anal. Chim. Acta 2007, 602, 114-121.


