12.E: The Chemical Bond (Exercises)
- Page ID
- 41338
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)12.1: Lewis Structures
Q12.1
Which of the following molecules has the shortest bond length?- \(O_2\)
- \(Cl_2\)
- \(Br_2\)
- \(I_2\)
Q12.2
Draw a plausible Lewis structure for 1,3-Bisphosphoglyerate (1,3-BPG), (C3H8O10P2).Q12.3
Draw resonance structures for NO3- and ClO3-, please show formal charges in drawings.
Q12.4
Calculate the dipole moment (\(\mu\)) of a two ions of +2e and -2e that are separated by 100 pm. Express dipole moment in both units of \(C\,m\) and \(D\).
S12.4
\[ \mu = Q \times r = 2 ( 1.602 \times 10^{-19})( 8 \times 10^{-10}) = 2.56 \times 10^{-28} C\,m\]
\[(2.56 \times 10^{-28} \cancel{C\,m}) \left( \dfrac{1\,D}{3.336 \times 10^{-30} \cancel{C\,m}} \right) = 76.8 \;D\]
12.2: Valence Bond Theory
Q12.5
What are the resonance of \(NO_3^0\)?
S12.5
Resonance structure in basic means that there are multiple way to draw a Lewis structure. For example:
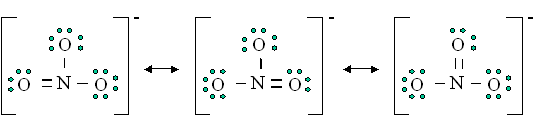
*Source from Michael Blaber
Q12.6
Draw resonance structures for the carbonate ion (\(CO_3^{-2}\)).
S12.6
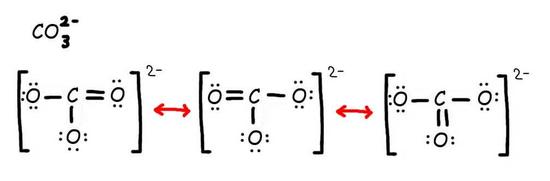
Q12.7a
S12.7a
Q12.7b
Draw the Lewis structure for nitric acid (\(HNO_3\)). This may or may not include resonance structures.S12.7b
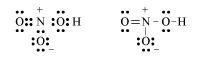
12.3: Hybridization of Atomic Orbitals
Q12.8a
S12.8a
Q12.8b
Describe the pattern of valence electrons across the periodic table (left to right and top to bottom), excluding the transition metals.S12.8b
The number of valence electrons increases going left to right along the same row and stays constant going from top to bottom along the same column.Q12.9a
What is the difference between covalent bond and ionic bond wave functions?S12.9a
An ionic bond wave function takes into account the probability that the electrons of a multiatomic molecule might exist on the same atom; the covalent bond wave function assumes they exist on separate atoms.
Q12.9b
What is the hybridization of HCl?
S12.9b
There is no orbital hybridization in HCl. The \(3p_z\) of Cl can overlap with the 1s of H just fine; they are only 0.06 eV apart in energy However, the 3s of Cl and the 1s of H are too far away in energy to interact, so there is no reason why the 3p of Cl can interact with the 3s of Cl anyway.

Q13.7
Calculate the potential energy of interaction between an electron situated 5.5 Å away from an HF molecule with a dipole moment of 2.5 D.S13.7
\[\mu = 2.5 D \times \dfrac{3.3356\times 10^{30}C m}{D} = 8.39 \times 10^{-30}Cm\]
\[r = 5.5 \; angstroms= 5.5 \times 10^{-10}m\]
\[V = \dfrac{q\mu}{4\pi \varepsilon _{o}r^{2}} = \dfrac{(+1.602\times10^{-19}C)(8.39\times 10^{-30}Cm)}{4\pi (8.851\times 10^{-12}C^{-2}N^{-1}m^{-2})(5.5\times 10^{-10})} = 2.196\times 10^{-11} J\]
Q12.10
Give two types of bonding that present in the molecule CH2. how do you do this?S12.10
Q12.11
Q12.12
Give the bonding scheme in term of molecular orbital H2+, H2, He2+, He. Besides, give the species in the following decreasing stability.Q12.13
Between F2 and F2+ which one has longer bond?S12.13
\(F_2^+\) has bond order 1.5, \(F_2\) has bond order 1.0. The bond lengths are inverse to the bond order, so the order is F2+ < F2 .There is 1 unpaired electron in F2+, 0 unpaired electrons in F2
Q12.14
Which of the following molecules have the shortest bond: F2, N2, O2? Why?S12.14
N2 has the shortest bond because the bond order is 3 while O2 is 2 and F2 is 1.12.5: Molecular Orbital Theory
Q12.15
Draw the molecular orbital diagram for F2 with the atomic orbitals labeled and find the bond order.Q12.16
Comparing benezene and cyclopentadiene, which one has a lesser degree of \(\pi\)-electron delocalization?Q12.19
Draw the molecular orbital energy diagram for the F2 Then compare the relative stability of the molecule to F2+.
Answer

BOND ORDER OF F2 = (8-6)/2 =1
BOND ORDER OF F2+ (8-5)/2 =1.5
Based on bond order F2+ is stable than F2
Q12.21
Diatomic carbon (C2) exists at very high temperatures. What type or types of bonds do you expect a molecule of diatomic carbon to have?
S12.21
1. Draw a molecular orbital diagram.
Answer: The molecular orbital diagram predicts that there will be a double bond consisting of two π bonds.
Q12.21
Determine the second period for molecule O2 by using molecular orbital?
S12.21

Based on general diagram for molecular orbital of second row of O2 molecule, the 

Q12.30
Though there are some exceptions, the double bond rule states that atoms of period 3 and greater do not form double or triple bonds. Explain why this is generally true. Hint: What properties of elements change with increasing period?
S12.30
Pi bonds are usually weaker than sigma bonds because there is less orbital overlap. The larger atomic radius of period 3 and greater elements means that there is even less overlap between the orbitals that would form pi bonds. There is not enough overlap to form the pi bonds that would make for double or triple bonds.
Q12.30
Explain why the Si=Si double bond is not stable than the C=C. Given the covalent radius of carbon is 77 pm and Silicon is 111pm?
S12.30
Silicon is significantly bigger than carbon. Thus the Si-Si bond is longer than C-C bond. However, the longer bond result in lowering the effective overlap of 3p orbital to form a π bonds. In contrast, C atoms can form the π bond from 2p orbital. Thus the Si=Si is less stable despite its larger size.
Q12.33
Myohemerythrin is an iron-containing protein that binds oxygen in marine invertebrates. The protein is monomeric (has one subunit) and contains 0.8107% iron by mass. Estimate the molar mass of myohemerythrin. Is your answer the same as the known molar mass of myohemerythrin, 13,780 grams per mole? If it is not, account for the difference between your estimate and the known value.

Myohemerythrin ribbon schematic, a 4-helix bundle protein structure with a bound iron. Hand drawn by Jane Richardson in 1980, from PDB file 2MHR.
S12.33
1. Find the mass of the protein if there is one atom of iron per protein.
\[ 55.85\ g\ Fe/mol \times \dfrac{100.0\ g\ protein}{0.8107\ g\ Fe}\]
\[ 6889 g/mol \]
2. This is much less than the known molar mass. In fact, it is half.
\[ \dfrac{13780\ g/mol}{6889\ g/mol} = 2.000 \]
We know the protein is monomeric, so there must be two atoms of iron in each protein.
Q12.19
S12.19
Based on the molecular orbital diagram, there are more anti-bonding than bonding state. The bonding order of Ne2 is:
BO = 1/2 (8-8) = 0
Therefore, \(Ne_2\) does not exist in nature.
Q12.21
S12.21

Q12.30
Be is in the second period, and very rare Be2 exist in nature. Explain the instability of Be2 , and calculate the bond order.
S12.30
Be2 molecule has 2 valence electrons. The bonding order, BO= 1/2 (2-2)= 0 Therefore, diatomic Be2 is not stable! Not exist in nature12.4: Electronegativity and Dipole Moment
Q12.36
Will the dipole moment of a cis-dichloroethylene molecule increase or decrease upon heating? Why?
S12.36
The dipole moment will decrease because cis-trans isomerization will occur when heated. This is because the trans isomer of the molecule does not have a dipole moment present.
Q12.37
Draw the \(\sigma\) and \(\sigma^*\) molecular orbital of \(CO\). Draw the MO energy level diagram and write the electron coefficient. Calculate the Bond order.
S12.37
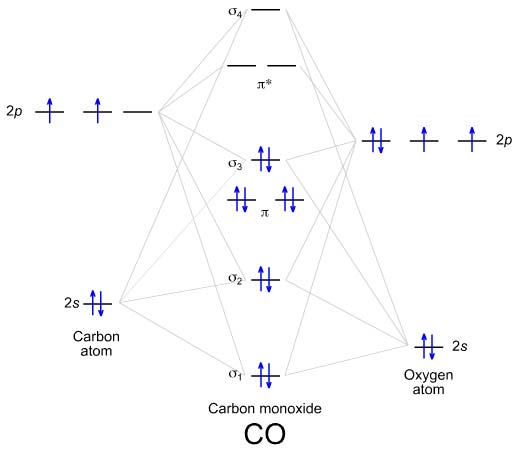
Electron Configuration:
(1s)2(
*1s)2(
2s)2(
*2s)2(
x)2(
y)2(
2sp)2
Bond Order: 1/2(Bonding Electrons-Antibonding Electrons)
=1/2(8-2) = 3

