1.23: A more complicated bonding example
- Page ID
- 9346
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
As another example, we will use group theory to construct the molecular orbitals of \(H_2O\) (point group \(C_{2v}\)) using a basis set consisting of all the valence orbitals. The valence orbitals are a \(1s\) orbital on each hydrogen, which we will label \(s_H\) and \(s_H'\), and a \(2s\) and three \(2p\) orbitals on the oxygen, which we will label \(s_O\), \(p_x\), \(p_y\), \(p_z\) giving a complete basis \(\begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix}\).
The first thing to do is to determine how each orbital transforms under the symmetry operations of the \(C_{2v}\) point group (\(E\), \(C_2\), \(\sigma_v\) and \(\sigma_v'\)), construct a matrix representation and determine the characters of each operation. The symmetry operations and axis system we will be using are shown below.
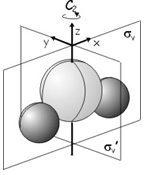
The orbitals transform in the following way
\[\begin{array}{lrcl} E & \begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix} & \rightarrow & \begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix} \\ C_2 & \begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix} & \rightarrow & \begin{pmatrix} s_H', s_H, s_O, -p_x, -p_y, p_z \end{pmatrix} \\ \sigma_v(xz) & \begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix} & \rightarrow & \begin{pmatrix} s_H, s_H', s_O, p_x, -p_y, p_z \end{pmatrix} \\ \sigma_v'(yz) & \begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix} & \rightarrow & \begin{pmatrix} s_H', s_H, s_O, -p_x, p_y, p_z \end{pmatrix} \end{array} \label{23.1}\]
A short aside on constructing matrix representatives
After a little practice, you will probably be able to write matrix representatives straight away just by looking at the effect of the symmetry operations on the basis. However, if you are struggling a little the following procedure might help.
Remember that the matrix representatives are just the matrices we would have to multiply the left hand side of the above equations by to give the right hand side. In most cases they are very easy to work out. Probably the most straightforward way to think about it is that each column of the matrix shows where one of the original basis functions ends up. For example, the first column transforms the basis function \(s_H\) to its new position. The first column of the matrix can be found by taking the result on the right hand side of the above expressions, replacing every function that isn’t \(s_H\) with a zero, putting the coefficient of \(s_H\) (\(1\) or \(-1\) in this example) in the position at which it occurs, and taking the transpose to give a column vector.
Consider the representative for the \(C_2\) operation. The original basis \(\begin{pmatrix} s_H, s_H', s_O, p_x, p_y, p_z \end{pmatrix}\) transforms into \(\begin{pmatrix} s_H', s_H, s_O, -p_x, -p_y, p_z \end{pmatrix}\). The first column of the matrix therefore transforms \(s_H\) into \(s_H'\). Taking the result and replacing all the other functions with zeroes gives \(\begin{pmatrix} 0, s_H, 0, 0, 0, 0 \end{pmatrix}\). The coefficient of \(s_H\) is \(1\), so the first column of the \(C_2\) matrix representative is
\[\begin{pmatrix} 0 \\ 1 \\ 0 \\ 0 \\ 0 \\ 0 \end{pmatrix} \label{23.2}\]
Matrix representation, characters and SALCs
The matrix representatives and their characters are
\[\begin{array}{cccc} E & C_2 & \sigma_v & \sigma_v' \\ \scriptsize{\begin{pmatrix} 1 & 0 & 0 & 0 & 0 & 0 \\ 0 & 1 & 0 & 0 & 0 & 0 \\ 0 & 0 & 1 & 0 & 0 & 0 \\ 0 & 0 & 0 & 1 & 0 & 0 \\ 0 & 0 & 0 & 0 & 1 & 0 \\ 0 & 0 & 0 & 0 & 0 & 1 \end{pmatrix}} & \scriptstyle{\begin{pmatrix} 0 & 1 & 0 & 0 & 0 & 0 \\ 1 & 0 & 0 & 0 & 0 & 0 \\ 0 & 0 & 1 & 0 & 0 & 0 \\ 0 & 0 & 0 & -1 & 0 & 0 \\ 0 & 0 & 0 & 0 & -1 & 0 \\ 0 & 0 & 0 & 0 & 0 & 1 \end{pmatrix}} & \scriptstyle{\begin{pmatrix} 1 & 0 & 0 & 0 & 0 & 0 \\ 0 & 1 & 0 & 0 & 0 & 0 \\ 0 & 0 & 1 & 0 & 0 & 0 \\ 0 & 0 & 0 &1 & 0 & 0 \\ 0 & 0 & 0 & 0 & -1 & 0 \\ 0 & 0 & 0 & 0 & 0 & 1 \end{pmatrix}} & \scriptstyle{\begin{pmatrix} 0 & 1 & 0 & 0 & 0 & 0 \\ 1 & 0 & 0 & 0 & 0 & 0 \\ 0 & 0 & 1 & 0 & 0 & 0 \\ 0 & 0 & 0 & -1 & 0 & 0 \\ 0 & 0 & 0 & 0 & 1 & 0 \\ 0 & 0 & 0 & 0 & 0 & 1 \end{pmatrix}} \\ \chi(E) = 6 & \chi(C_2) = 0 & \chi(\sigma_v) = 4 & \chi(\sigma_v') = 2 \end{array} \label{23.3}\]
Now we are ready to work out which irreducible representations are spanned by the basis we have chosen. The character table for \(C_{2v}\) is:
\[\begin{array}{l|cccc|l} C_{2v} & E & C_2 & \sigma_v & \sigma_v' & h = 4 \\ \hline A_1 & 1 & 1 & 1 & 1 & z, x^2, y^2, z^2 \\ A_2 & 1 & 1 & -1 & -1 & xy, R_z \\ B_1 & 1 & -1 & 1 & -1 & x, xz, R_y \\ B_2 & 1 & -1 & -1 & 1 & y, yz, R_x \\ \hline \end{array}\]
As before, we use Equation (15.20) to find out the number of times each irreducible representation appears.
\[a_k = \dfrac{1}{h}\sum_C n_C \chi(g) \chi_k(g) \label{23.4}\]
We have
\[\begin{array}{rcll} a(A_1) & = & \dfrac{1}{4}(1 \times 6 \times 1 + 1 \times 0 \times 1 + 1\times 4\times 1 + 1\times 2\times 1) & = 3 \\ a(A_2) & = & \dfrac{1}{4}(1\times 6\times 1 + 1\times 0\times 1 + 1\times 4\times -1 + 1\times 2\times -1) & = 0 \\ a(B_1) & = & \dfrac{1}{4}(1\times 6\times 1 + 1\times 0\times -1 + 1\times 4\times 1 + 1\times 2\times -1) & = 2 \\ a(B_2) & = & \dfrac{1}{4}(1\times 6\times 1 + 1\times 0\times -1 + 1\times 4\times -1 + 1\times 2\times 1) & = 1 \end{array} \label{23.5}\]
so the basis spans \(3A_1 + 2B_1 + B_2\). Now we use the projection operators applied to each basis function \(f_i\) in turn to determine the SALCs \(\phi_i = \Sigma_g \chi_k(g) g f_i\)
The SALCs of \(A_1\) symmetry are:
\[\begin{array}{rclll} \phi(s_H) & = & s_H + s_H' + s_H + s_H' & = & 2(s_H + s_H') \\ \phi(s_H') & = & s_H' + s_H + s_H' + s_H & = & 2(s_H + s_H') \\ \phi(s_O) & = & s_O + s_O + s_O + s_O & = & 4s_O \\ \phi(p_x) & = & p_x - p_x + p_x - p_x & = & 0 \\ \phi(p_y) & = & p_y - p_y + p_y - p_y & = & 0 \\ \phi(p_z) & = & p_z + p_z + p_z + p_z & = & 4p_z \end{array} \label{23.6}\]
The SALCs of \(B_1\) symmetry are:
\[\begin{array}{rclll} \phi(s_H) & = & s_H - s_H' + s_H - s_H' & = & 2(s_H - s_H') \\ \phi(s_H') & = & s_H' - s_H + s_H' - s_H & = & 2(s_H' - s_H) \\ \phi(s_O) & = & s_O - s_O + s_O - s_O & = & 0 \\ \phi(p_x) & = & p_x + p_x + p_x + p_x & = & 4p_x \\ \phi(p_y) & = & p_y + p_y - p_y - p_y & = & 0 \\ \phi(p_z) & = & p_z - p_z + p_z - p_z & = & 0 \end{array} \label{23.7}\]
The SALCs of \(B_2\) symmetry are:
\[\begin{array}{rclll} \phi(s_H) & = & s_H - s_H' -s_H = s_H' & = & 0 \\ \phi(s_H') & = & s_H' - s_H - s_H' + s_H & = & 0 \\ \phi(s_O) & = & s_O - s_O - s_O + s_O & = & 0 \\ \phi(p_x) & = & p_x + p_x - p_x - p_x & = & 0 \\ \phi(p_y) & = & p_y + p_y + p_y + p_y & = & 4p_y \\ \phi(p_z) & = & p_z - p_z - p_z + p_z & = & 0 \end{array} \label{23.8}\]
After normalization, our SALCs are therefore:
A1 symmetry
\[\begin{array}{rcl} \phi_1 & = & \dfrac{1}{\sqrt{2}}(s_H + s_H') \\ \phi_2 & = & s_O \\ \phi_3 & = & p_z \end{array} \label{23.9}\]
B1 symmetry
\[\begin{array}{rcl} \phi_4 & = & \dfrac{1}{\sqrt{2}}(s_H - s_H') \\ \phi_5 & = & p_x \end{array} \label{23.10}\]
B2 symmetry
\[\begin{array}{rcl} \phi_6 & = & p_y \end{array} \label{23.11}\]
Note that we only take one of the first two SALCs generated by the \(B_1\) projection operator since one is a simple multiple of the other (i.e. they are not linearly independent). We can therefore construct three molecular orbitals of \(A_1\) symmetry, with the general form
\[\begin{array}{rcll} \Psi(A_1) & = & c_1 \phi_1 + c_2 \phi_2 + c_3 \phi_3 & \\ & = & c_1'(s_H + s_H') + c_2 s_O + c_3 p_z & \text{where} \: c_1' = \dfrac{c_1}{\sqrt{2}} \end{array} \label{23.12}\]
two molecular orbitals of \(B_1\) symmetry, of the form
\[\begin{array}{rcl} \Psi(B_1) & = & c_4 \phi_4 + c_5 \phi_5 \\ & = & c_4'(s_H - s_H') + c_5 p_z \end{array} \label{23.13}\]
and one molecular orbital of \(B_2\) symmetry
\[\begin{array}{rcl} \Psi(B_2) & = & \phi_6 \\ & = & p_y \end{array} \label{23.14}\]
To work out the coefficients \(c_1\) - \(c_5\) and determine the orbital ener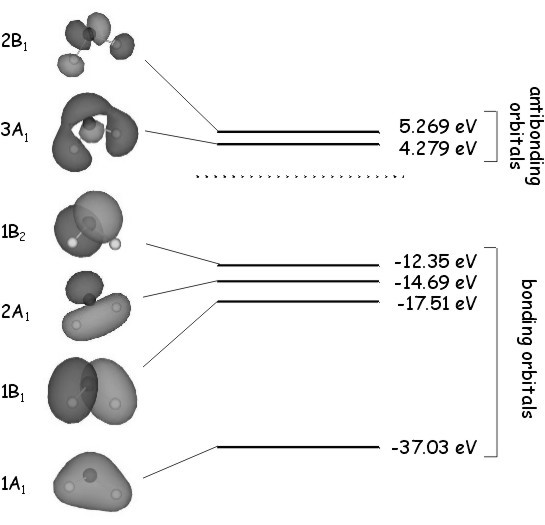 gies, we would have to solve the secular equations for each set of orbitals in turn. We are not dealing with a conjugated \(p\) system, so in this case Hückel theory cannot be used and the various \(H_{ij}\) and \(S_{ij}\) integrals would have to be calculated numerically and substituted into the secular equations. This involves a lot of tedious algebra, which we will leave out for the moment. The LCAO orbitals determined above are an approximation of the true molecular orbitals of water, which are shown on the right. As we have shown using group theory, the \(A_1\) molecular orbitals involve the oxygen \(2s\) and \(2p_z\) atomic orbitals and the sum \(s_H + s_H'\) of the hydrogen \(1s\) orbitals. The \(B_1\) molecular orbitals involve the oxygen \(2p_x\) orbital and the difference \(s_H -s_H'\) of the two hydrogen \(1s\) orbitals, and the \(B_2\) molecular orbital is essentially an oxygen \(2p_y\) atomic orbital.
gies, we would have to solve the secular equations for each set of orbitals in turn. We are not dealing with a conjugated \(p\) system, so in this case Hückel theory cannot be used and the various \(H_{ij}\) and \(S_{ij}\) integrals would have to be calculated numerically and substituted into the secular equations. This involves a lot of tedious algebra, which we will leave out for the moment. The LCAO orbitals determined above are an approximation of the true molecular orbitals of water, which are shown on the right. As we have shown using group theory, the \(A_1\) molecular orbitals involve the oxygen \(2s\) and \(2p_z\) atomic orbitals and the sum \(s_H + s_H'\) of the hydrogen \(1s\) orbitals. The \(B_1\) molecular orbitals involve the oxygen \(2p_x\) orbital and the difference \(s_H -s_H'\) of the two hydrogen \(1s\) orbitals, and the \(B_2\) molecular orbital is essentially an oxygen \(2p_y\) atomic orbital.
Contributors and Attributions
Claire Vallance (University of Oxford)


