Addition Polymers
- Page ID
- 1127
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Polymers are long chain giant organic molecules are assembled from many smaller molecules called monomers. Polymers consist of many repeating monomer units in long chains. A polymer is analogous to a necklace made from many small beads (monomers). Many monomers are alkenes or other molecules with double bonds which react by addition to their unsaturated double bonds.
Introduction
The electrons in the double bond are used to bond two monomer molecules together. This is represented by the red arrows moving from one molecule to the space between two molecules where a new bond is to form. The formation of polyethylene from ethylene (ethene) may be illustrated in the graphic on the left as follows. In the complete polymer, all of the double bonds have been turned into single bonds. No atoms have been lost and you can see that the monomers have just been joined in the process of addition. A simple representation is -[A-A-A-A-A]-. Polyethylene is used in plastic bags, bottles, toys, and electrical insulation.
- LDPE - Low Density Polyethylene: The first commercial polyethylene process used peroxide catalysts at a temperature of 500 C and 1000 atmospheres of pressure. This yields a transparent polymer with highly branched chains which do not pack together well and is low in density. LDPE makes a flexible plastic. Today most LDPE is used for blow-molding of films for packaging and trash bags and flexible snap-on lids. LDPE is recyclable plastic #4.
- HDPE - High Density Polyethylene: An alternate method is to use Ziegler-Natta aluminum titanium catalysts to make HDPE which has very little branching, allows the strands to pack closely, and thus is high density. It is three times stronger than LDPE and more opaque. About 45% of the HDPE is blow molded into milk and disposable consumer bottles. HDPE is also used for crinkly plastic bags to pack groceries at grocery stores. HDPE is recyclable plastic #2.
Other Addition Polymers
- PVC (polyvinyl chloride), which is found in plastic wrap, simulated leather, water pipes, and garden hoses, is formed from vinyl chloride (H2C=CHCl). The reaction is shown in the graphic on the left. Notice how every other carbon must have a chlorine attached.
- Polypropylene: The reaction to make polypropylene (H2C=CHCH3) is illustrated in the middle reaction of the graphic. Notice that the polymer bonds are always through the carbons of the double bond. Carbon #3 already has saturated bonds and cannot participate in any new bonds. A methyl group is on every other carbon.
- Polystyrene: The reaction is the same for polystrene where every other carbon has a benzene ring attached. Polystyrene (PS) is recyclable plastic #6. In the following illustrated example, many styrene monomers are polymerized into a long chain polystyrene molecule. The squiggly lines indicate that the polystyrene molecule extends further at both the left and right ends.
- Blowing fine gas bubbles into liquid polystyrene and letting it solidify produces expanded polystyrene, called Styrofoam by the Dow Chemical Company.
- Polystyrene with DVB: Cross-linking between polymer chains can be introduced into polystyrene by copolymerizing with p-divinylbenzene (DVB). DVB has vinyl groups (-CH=CH2) at each end of its molecule, each of which can be polymerized into a polymer chain like any other vinyl group on a styrene monomer.
| Monomer | Polymer Name | Trade Name | Uses |
|---|---|---|---|
| F2C=CF2 | polytetrafluoroethylene | Teflon |
Non-stick coating for cooking utensils, chemically-resistant specialty plastic parts, Gore-Tex |
| H2C=CCl2 | polyvinylidene dichloride | Saran | Clinging food wrap |
| H2C=CH(CN) | polyacrylonitrile | Orlon, Acrilan, Creslan | Fibers for textiles, carpets, upholstery |
| H2C=CH(OCOCH3) | polyvinyl acetate | Elmer's glue - Silly Putty Demo | |
| H2C=CH(OH) | polyvinyl alcohol | Ghostbusters Demo | |
| H2C=C(CH3)COOCH3 | polymethyl methacrylate | Plexiglass, Lucite | Stiff, clear, plastic sheets, blocks, tubing, and other shapes |
Addition polymers from conjugated dienes
Polymers from conjugated dienes usually give elastomer polymers having rubber-like properties.
| Monomer | Polymer name | Trade name | Uses |
|---|---|---|---|
| H2C=CH-C(CH3)=CH2 | polyisoprene | natural or some synthetic rubber | applications similar to natural rubber |
| H2C=CH-CH=CH2 | polybutadiene | polybutadiene synthetic rubber | select synthetic rubber applications |
| H2C=CH-CCl=CH2 | polychloroprene | Neoprene | chemically-resistant rubber |
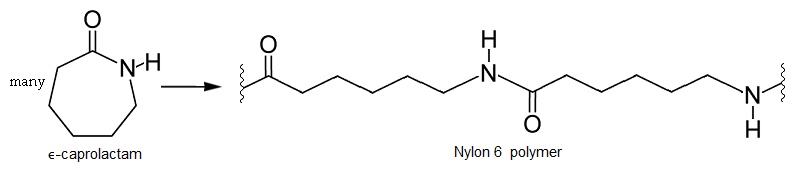
Ring opening polymerization
In this kind of polymerization, molecular rings are opened in the formation of a polymer. Here epsilon-caprolactam, a 6-carbon cyclic monomer, undergoes ring opening to form a Nylon 6 homopolymer, which is somewhat similar to but not the same as Nylon 6,6 alternating copolymer.
References
- Gorodetsky, Malka. "Electroplating of polyethylene." J. Chem. Educ. 1978, 55, 66.
Contributors
Charles Ophardt (Professor Emeritus, Elmhurst College); Virtual Chembook