29.7: Addition Polymers
- Page ID
- 22398
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)We have discussed the synthesis and properties of a considerable number of addition polymers in this and previous chapters. Our primary concern here will be with some aspects of the mechanism of addition polymerization that influence the character of the polymer formed.
Alkene Polymerization
The most important type of addition polymerization is that of alkenes (usually called vinyl monomers) such as ethene, propene, ethenylbenzene, and so on. In general, we recognize four basic kinds of mechanisms for polymerization of vinyl monomers - radical, cationic, anionic, and coordination. The elements of the first three of these have been outlined (Section 10-8). The possibility, in fact the reality, of a fourth mechanism is essentially forced on us by the discovery of the Ziegler and other (mostly heterogeneous) catalysts, which apparently do not involve "free" radicals, cations, or anions, and which can and usually do lead to highly stereoregular polymers. With titanium-aluminum Ziegler catalysts, the growing chain has a \(\ce{C-Ti}\) bond; further monomer units then are added to the growing chain by coordination with titanium, followed by an intramolecular rearrangement to give a new growing-chain end and a new vacant site on titanium where a new molecule of monomer can coordinate:
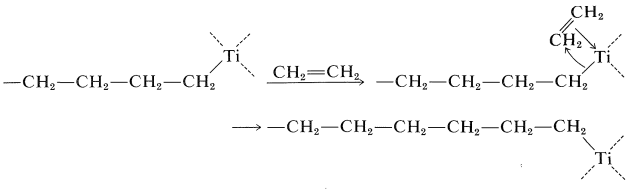
In the coordination of the monomer with the titanium, the metal is probably behaving as an electrophilic agent and the growing-chain end can be thought of as being transferred to the monomer as an anion. Because this mechanism gives no explicit role to the aluminum, it is surely oversimplified. Ziegler catalysts polymerize most monomers of the type \(\ce{RCH=CH_2}\), provided the \(\ce{R}\) group is one that does not react with the organometallic compounds present in the catalyst. More reactions of the type that occur in Ziegler polymerizations will be discussed in Chapter 31.
Radical Polymerization
In contrast to coordination polymerizations, formation of vinyl polymers by radical chain mechanisms is reasonably well understood - at least for the kinds of procedures used on the laboratory scale. The first step in the reaction is the production of radicals; this can be achieved in a number of different ways, the most common being the thermal decomposition of an initiator, usually a peroxide or an azo compound:
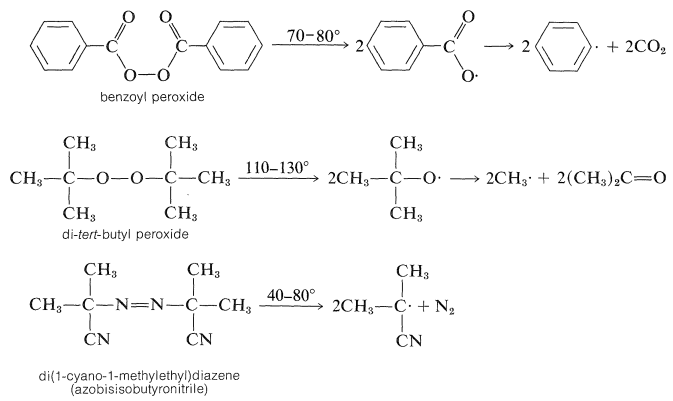
Many polymerizations are carried out on aqueous emulsions of monomers. For these, water-soluble inorganic peroxides, such as ammonium peroxysulfate, often are employed.
Other ways of obtaining initiator radicals include high-temperature decomposition of the monomer and photochemical processes, often involving a ketone as a photosensitizer. Addition of the initiator radicals to monomer produces a growing-chain radical that combines with successive molecules of monomer until, in some way, the chain is terminated. Addition to an unsymmetrical monomer can occur in two ways. Thus for ethenylbenzenes:
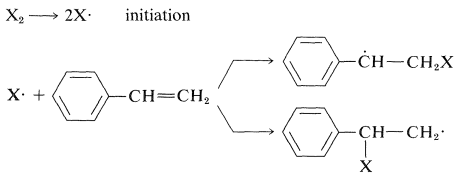
All evidence on addition of radicals to ethenylbenzene indicates that the process by which \(\ce{X} \cdot\) adds to the \(\ce{CH_2}\) end of the double bond is greatly favored over addition at the \(\ce{CH}\) end. This direction of addition is in accord with the considerable stabilization of the phenylmethyl radicals relative to the alkyl radicals (see Sections 14-3C and 26-4D). Polymerization then will result in the addition of monomer units to give phenyl groups only on alternate carbons ("head-to-tail" addition):
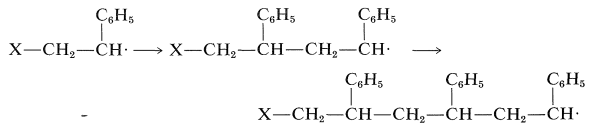
In general, we predict that the direction of addition of an unsymmetrical monomer will be such as to give always the most stable growing-chain radical. Similar considerations were discussed previously (Section 21-11) in respect to how [2 + 2] cycloadditions occur.
The process of addition of monomer units to the growing chain can be interrupted in different ways. One is chain termination by combination or disproportionation of radicals. Explicitly, two growing-chain radicals can combine to form a carbon-carbon bond, or disproportionation can occur with a hydrogen atom being transferred from one chain to the other:
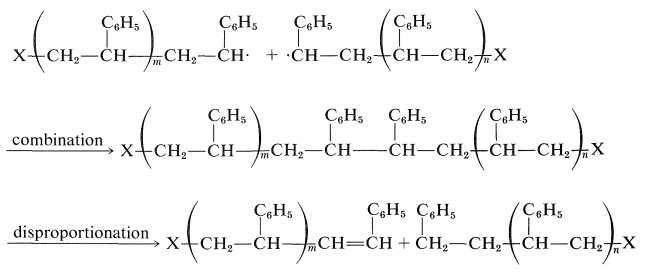
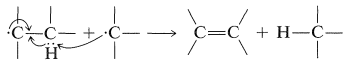
The disproportionation reaction is the radical equivalent of the \(E2\) reaction:
Which mode of termination occurs can be determined by measuring the number of initiator fragments per polymer molecule. If there are two initiator fragments in each molecule, termination must have occurred by combination. One initiator fragment per molecule indicates disproportionation. Apparently, ethenylbenzene polymerizations terminate by combination, but with methyl 2-methylpropenoate, both reactions take place, disproportionation being favored.
Another very important way that a growing chain may be terminated is by chain transfer. This stops the chain but starts a new one. Thiols, such as phenylmethanethiol and dodecanethiol, are efficient chain-transferring agents. The reactions involved are as follows (where \(\ce{M}\) represents monomer and \(\ce{RSH}\) represents the chain-transfer reagent):
\[\begin{align} \ce{X-M-(M)}_n \ce{-M} \cdot + \ce{RSH} &\rightarrow \ce{X-M-(M)}_n \ce{-M-H} + \ce{RS} \cdot \\ \ce{RS} \cdot + \ce{M} &\rightarrow \ce{RS-M} \cdot \\ \ce{RS-M} \cdot + \left( n + 1 \right) \ce{M} &\rightarrow \ce{RS-M-(M)}_n \ce{-M} \cdot \: \text{(new growing chain)} \\ \ce{RS-M-(M)}_n \ce{-M} \cdot + \ce{RSH} &\rightarrow \ce{RS-M-(M)}_n \ce{-M-H} + \ce{RS} \cdot \: \text{etc.} \end{align}\]
Chain transfer reduces the average molecular weight of the polymer without wasting initiator radicals. Dodecanethiol has considerable use in the manufacture of GRS rubber (Section 13-4) as a regulator to hold down the molecular weight in the emulsion polymerization of 1,3-butadiene and ethenylbenzene.
Polymerization inhibitors stop or slow down polymerization by reacting with the initiator or growing-chain radicals. A wide variety of substances can behave as inhibitors: quinones, hydroquinones, aromatic nitro compounds, aromatic amines, and so on. In cases where the inhibitor is a hydrogen donor (symbolized here by \(\ce{InH}\)), then for inhibition to occur, the radical resulting from hydrogen transfer \(\left( \ce{In} \cdot \right)\) must be too stable to add to monomer. If it does add to monomer and starts a new chain, chain transfer occurs instead of inhibition. For perfect inhibition, the \(\ce{In} \cdot\) radicals must combine with themselves (or initiator radicals) to give inert products:
\[\begin{align} \ce{X-(M)}_n \ce{-M} \cdot + \ce{InH} &\rightarrow \ce{X-(M)}_n \ce{-M-H} + \ce{In} \cdot \\ 2 \ce{In} \cdot &\rightarrow \text{inert products (inhibition)} \\ \ce{In} \cdot + \ce{M} &\rightarrow \ce{In-M} \cdot \: \text{(chain transfer)} \end{align}\]
Many compounds are known that fall in the intermediate zone between chain transfer and inhibition reagents.
Some inhibitors such as 2,3,5,6-tetrachloro-1,4-benzenedione (tetrachlorobenzoquinone) act as inhibitors by adding to the growing chain radicals to give radicals too stable to continue the chain:
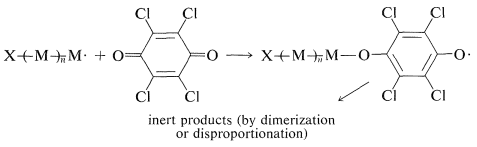
Again, for inhibition to be effective there must be destruction of the stable radicals by dimerization or disproportionation.
The reactive vinyl monomers usually are stabilized against polymerization, while in storage, by addition of \(0.1\) to \(1\%\) of an inhibitor. 1,4-Benzenediol (hydroquinone), 2,6-di-tert-butyl-4-methylbenzenol, and 4-tert-butyl-1,2-benzenediol are used for this purpose. These substances are especially effective at scavenging \(\ce{RO} \cdot\) radicals, which are formed by oxidation of the monomer with atmospheric oxygen.
Cationic Polymerization
Polymerization by the cationic mechanism is most important for 2-methylpropene (isobutylene), which does not polymerize well by other methods, and was discussed previously in considerable detail (Section 10-8B).
Anionic Polymerization
In general, we expect that anionic polymerization will be favorable when the monomer carries substituents that will stabilize the anion formed when a basic initiator such as amide ion adds to the double bond of the monomer:
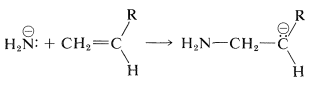
Cyano and alkoxycarbonyl groups are favorable in this respect and propenenitrile and methyl 2-methylpropenoate can be polymerized with sodium amide in liquid ammonia. Ethenylbenzene and 2-methyl-1,3-butadiene undergo anionic polymerization under the influence of organolithium and organosodium compounds, such as butyllithium and phenylsodium.
An important development in anionic polymerization has been provided by M. Szwarc's "living polymers". The radical anion, sodium naphthalenide (Section 27-9), transfers an electron reversibly to ethenyl benzene to form a new radical anion, \(1\), in solvents such as 1,2-dimethoxyethane or oxacyclopentane:
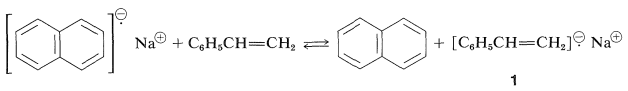
Dimerization of the sodium naphthalenide radical anion would result in a loss of aromatic stabilization, but this is not true for \(1\), which can form a \(\ce{C-C}\) bond and a resonance-stabilized bis-phenylmethyl dianion, \(2\) (Section 26-4C).
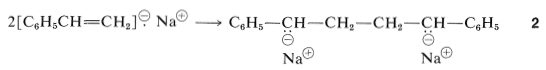
The anionic ends of \(2\) are equivalent and can add ethenylbenzene molecules to form a long-chain polymer with anionic end groups, \(3\):
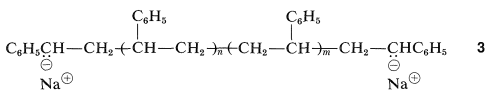
If moisture and oxygen are rigorously excluded, the anionic groups are stable indefinitely, and if more monomer is added polymerization will continue. Hence the name "living polymer", in contrast to a radical-induced polymerization, which only can be restarted with fresh monomer and fresh initiator, and even then not by growth on the ends of the existing chains.
The beauty of the Szwarc procedure is that the chains can be terminated by hydrolysis, oxidation, carboxylation with \(\ce{CO_2}\), and so on, to give polymer with the same kind of groups on each end of the chain. Also, it is possible to form chains in which different monomers polymerize well by the anion mechanism and contain no groups or impurities that will destroy the active ends. Thus one can start with ethenylbenzene \(\left( \ce{S} \right)\), and when the reaction is complete, add methyl 2-methylpropenoate \(\left( \ce{M} \right)\) to obtain a block copolymer of the type
\[\ce{M-M-M-M-M-S-S-S-S-S-S-M-M-M-M-M}\]
Copolymers
When polymerization occurs in a mixture of monomers there will be competition between the different kinds of monomers to add to the growing chain and produce a copolymer. Such a polymer will be expected to have physical properties quite different from those of a mixture of the separate homopolymers. Many copolymers, such as GRS, ethene-propene, Viton rubbers, and Vinyon plastics are of considerable commercial importance.
The rates of incorporation of various monomers into growing radical chains have been studied in considerable detail. The rates depend markedly on the nature of the monomer being added and on the character of the radical at the end of the chain. Thus a 1-phenylethyl-type radical on the growing chain reacts about twice as readily with methyl 2-methylpropenoate as it does with ethenylbenzene; a methyl 2-methylpropenoate end shows the reverse behavior, being twice as reactive toward ethylbenzene as toward methyl 2-methylpropenoate. This kind of behavior favors alternation of the monomers in the chain and reaches an extreme in the case of 2-methylpropene and butenedioic anhydride. Neither of these monomers separately will polymerize well with radical initiators. Nonetheless, a mixture polymerizes very well with perfect alternation of the monomer units. It is possible that, in this case, a 1:1 complex of the two monomers is what polymerizes.
In general however, in a mixture of two monomers one is considerably more reactive than the other and the propagation reaction tends to favor incorporation of the more reactive monomer, although there usually is some bias toward alternation. Ethenylbenzene and 2-methyl-1,3-butadiene mixtures are almost unique in having a considerable bias toward forming the separate homopolymers.
One of the more amazing copolymerizations is that of ethenylbenzene and oxygen gas, which at one atmosphere oxygen pressure gives a peroxide with an average molecular weight of 3000 to 4000 and a composition approaching \(\ce{C_8H_8O_2}\):
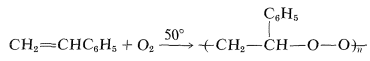
When heated rapidly in small portions the product undergoes a mild explosion and gives high yields (\(80\%\) to \(95\%\)) of methanal and benzenecarbaldehyde. The mechanism may be a kind of unzipping process, starting from a break in the chain an spreading toward each end:
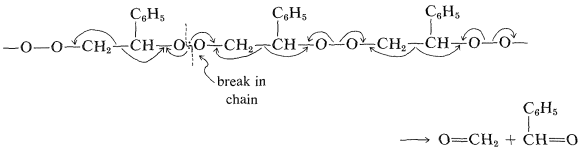
Another interesting copolymerization is of ethene and carbon monoxide by the radical mechanism. The polymer contains \(\ce{-CH_2-CH_2-CO-CH_2-CH_2}-\) units, which are broken apart at a \(\ce{CH_2} \vdots \ce{C=O}\) bond on absorption of ultraviolet light, thereby giving a polymer that has the possibility of degrading in the environment through the action of sunlight (see Section 28-2A).
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


