Everything You Need to Know About Mechanisms
- Page ID
- 14819
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The Correct Use of Arrows to Indicate Electron Movement
The ability to write an organic reaction mechanism properly is key to success in organic chemistry classes. Organic chemists use a technique called arrow pushing to depict the flow or movement of electrons during chemical reactions. Arrow pushing helps chemists keep track of the way in which electrons and their associated atoms redistribute as bonds are made and broken. The first essential rule to keep in mind is the following:
First rule: Arrows are used to indicate movement of electrons
A regular arrow (double-sided arrowhead) is used to indicate the movement of two electrons, while a line with a single-sided arrowhead (sometimes called a “fish hook arrow”) is used for single electron movement involved with radical reactions that are first described in Chapter 8.

The great majority of reactions that will be discussed in this book involve movement of pairs of electrons, so they are represented by double-sided arrowheads.
Arrow pushing was first introduced in Section 1.8A in the discussion of resonance contributing structures. Recall that when comparing two or more contributing structures, an arrow was used to show how two electrons (lines representing bonds or pairs of dots representing lone pairs) could be redistributed within a single chemical structure to create an alternative Lewis line structure representation of the bonding. By convention, arrows are used to keep track of all pairs of electrons that are in different locations in the two different contributing Lewis line structures, shown here for the acetate anion and benzene molecule.
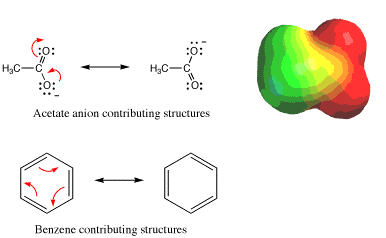
Keep in mind that in the case of resonance, 1) the atoms do not move between contributing structures, and 2) the electrons are not actually moving. The true chemical structure should be thought of as a hybrid of the contributing Lewis line structures. It is worth pointing out that when used with contributing structures, arrows generally indicate only the interconversion of p bonds and lone pairs (acetate ions) or just p bonds (benzene), not the formation or breaking of s bonds.
In chemical reactions, both electrons and atoms change positions as both p and s bonds are formed and broken. Arrow pushing is used to keep track of the movement of all electrons involved with each step of the overall transformation. Because electrons are located in orbitals surrounding atoms, when bonds are formed or broken, the movement of electrons between orbitals is necessarily accompanied by the movement of the associated atoms, which leads to the second rule of arrow pushing when depicting chemical reaction mechanisms:
Second Rule: Arrows are never used to indicate the movement of atoms directly. The arrows only show atom movement indirectly as a consequence of electron movement when covalent bonds are made and broken.
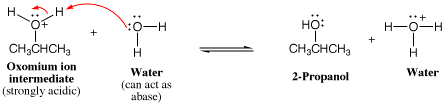
We have already used arrow pushing to show proton transfer several times in Chapter 4. The example below shows the transfer of a proton from the relatively acidic acetic acid molecule to the relatively basic hydroxide anion. We show this process with one arrow (labeled “a” in the diagram) that starts at a lone pair of electrons on the basic oxygen atom of the hydroxide anion, then points to the acidic H atom of acetic acid to indicate formation of the new bond being made. A second arrow originates at the line representing the breaking O-H bond and points to the O atom to denote creation of a lone pair (arrow “b”). In this reaction, the proton is being transferred between molecules, and the arrows indicate movement of the electrons involved.
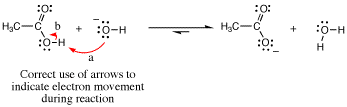
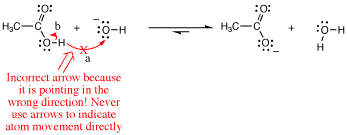
A common mistake beginning students make is that they will erroneously write an arrow pointing from the H of the acetic acid to the O atom of the hydroxide anion. This is wrong, because such an arrow would be indicating the H atom movement directly, not electron movement! Other common mistakes in arrow pushing are given at the end.
Electron Sources and Sinks: How to Predict What Will Occur in an Organic Reaction Mechanism
Combined with the arrows shown for the contributing structures shown previously, we have now seen all three of the situations illustrated by arrows with double-sided arrowheads, namely the redistribution of p bonds and/or lone pairs, formation of a new s bond (generally from a lone pair or sometimes a new p bond), and breaking of a s bond (generally to form a new lone pair or sometimes a new p bond). Often, as in the case of the acetate-hydroxide ion reaction, more than one arrow is used in a given mechanism step. Now that you have seen all of the important types of arrows, we can point out the most important common feature between them:
Third Rule: Arrows always start at an electron source and end at an electron sink.
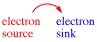
An electronsource is a bond or a lone pair of electrons. It is either a p bond or a lone pair on an atom of relatively high electron density in a molecule or ion, or a bond that must break during a reaction. An electron sink is an atom on a molecule or ion that can accept a new bond or lone pair of electrons.
Learning to identify the characteristic sources and sinks in different functional groups is the key to learning organic chemistry reaction mechanisms. For example, for arrows that depict the formation of new s bonds, the electron source is often readily identified as being a lone pair on the most electron rich atom of a molecule or ion, and the electron sink is readily identified as the most electron poor atom of a molecule or ion. Thus, the prediction of many of the most important electron sources and sinks comes down to lessons concerning the differences in electronegativity between atoms that were presented in Section 1.2, which allow you to identify partial and formal negative and positive charges in molecules. As an aid to your analysis, the red and blue colors of the various electrostatic surface maps given throughout this book indicate the negative and positive regions of molecules. We will have more to say about this reactivity pattern a little bit later.
This leads us to another commonly encountered type of process that deserves mention. As you will see in this and many later chapters, making a new bond to an electron sink often requires the simultaneous breaking of one of the bonds present at the sink atom to avoid overfilling its valence orbitals, a situation referred to as hypervalence.
Fourth rule: Breaking a bond will occur to avoid overfilling valence (hypervalence) on an atom serving as an electron sink.
In these cases, the electron source for the arrow is the bond being broken, and the sink is an atom able to accommodate the electrons as a lone pair, generally an electronegative atom such as an O atom or a halogen. If an ion is created, that ion is often stabilized by resonance delocalization or other stabilizing interactions.
Returning to the proton transfer reaction between acetic acid and hydroxide, we can now summarize our analysis of this simple one-step mechanism.
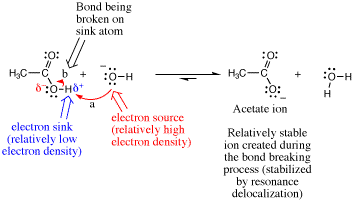
Viewed in the context of the third rule, when considering the arrow used to make a new s bond (arrow a), the hydroxide O atom is the electron source (most negatively charged atom) and the acetic acid H atom is the electron sink (atom with highest partial positive charge). This is illustrated using the electrostatic molecular surfaces shown below the reaction equation. The O atom of hydroxide ion has the greatest localized negative charge as indicated by the most intense red color and the acetic acid proton being transferred has the most intense positive charge character indicated by the most intense blue color. In order to avoid overfilling the valence of the H atom during the reaction (fourth rule), the O-H bond of acetic acid must be broken (arrow “b”). In so doing, the acetate ion is formed. Note that the acetate ion is stabilized by resonance delocalization.
Based on our analysis of the reaction between acetic acid and the hydroxide anion, you should now appreciate that the transfer of a proton (a so-called Brønsted acid-base reaction) is really just a special case of the common pattern of reactivity between an electron source (the base) and the proton as an electron sink, combined with breaking a bond to satisfy valence and create a relatively stable ion.
The addition or removal of protons during chemical reactions is so common that proton transfer steps are referred to by name directly, and we will use phrases such as “add a proton” or “take a proton away” when referring to them. However, proton transfer reactions are not the only case in which we use special names to describe a particular type of common reaction that involves arrows between electron sources and electron sinks.
As briefly mentioned in Section 4.7, a broader terminology is applied to the very common case of reactions in which new s bonds form between electron rich and electron poor regions of molecules. Nucleophiles (meaning nucleus seeking) are molecules that have relatively electron rich p bonds or lone pairs that act as electron sources for arrows making new bonds. Electrophiles (meaning electron seeking) are molecules with relatively electron poor atoms that serve as sinks for these arrows. Analogously, a molecule, or region of a molecule, that is a source for such an arrow is called nucleophilic, while a molecule or region of a molecule that is a sink for these arrows is referred to as being electrophilic. Based on this description, it should be clear that nucleophiles are analogous to Lewis bases and electrophiles are analogous to Lewis acids. Chemists use these terms interchangeably, although nucleophile and electrophile are more commonly used in kinetics discussions while Lewis acid and Lewis base are more commonly used in discussions about reaction thermodynamics. We will use all of these terms throughout the rest of the book.
It is helpful to summarize the appropriate use of key terms associated with arrow pushing and reaction mechanisms. The terms “source” and “sink” are used to identify the start and end of each reaction mechanism arrow, which is indicating the change in location of electron pairs. The terms “nucleophile” and “electrophile” (as well as “Lewis base” and “Lewis acid”) are used to describe molecules based on their chemical reactivity and propensity to either donate or receive electrons when they interact. Protons can be thought of as a specific type of electrophile, and for reactions in which a proton is transferred, the nucleophile is called a base.
| Example 1: I see you |
|---|
|
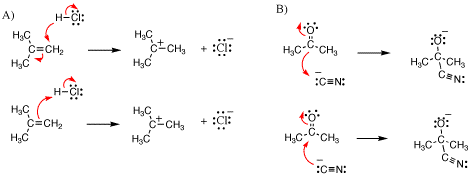
The following two sets of reactions (A and B) show possibilities for arrow pushing in individual reaction steps. Identify which is wrong and explain why. Next, using arrow pushing correctly, label which molecule is the nucleophile and which is the electrophile.
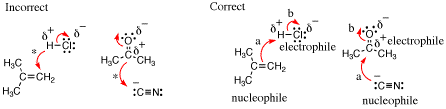
SolutionIn each case the first arrow pushing scenario is wrong. The arrows shown below with stars over them do not start at a source of electrons, but rather they start at positions of relative positive charge, which is incorrect.
In the correct arrow pushing, the arrow labeled “a” depicts the interaction of a region of relative high negative charge (a p-bond or lone pair) with an atom of relatively high partial positive charge on the other reactant. Therefore, the molecule acting as the source for arrow the s bond-forming arrow “a” is the nucleophile while the molecule containing the sink atom is the electrophile. The arrow labeled “b” is needed to satisfy valence, and is not considered when defining the nucleophile and electrophile. |
Putting it All Together: It Comes Down to a Multiple Choice Situation
In the sections and chapters that follow, many different reaction mechanisms will be described in a stepwise fashion. Each arrow can be classified according to one of the three overall situations we have already encountered (redistribution of \(\pi\) bonds and/or lone pairs, formation of a new s bond from a lone pair or \(\pi\) bond, breaking a s bond to give a new lone pair or \(\pi\) bond).
When learning new mechanisms, first focus on the overall transformation that takes place. It might be a reaction in which atoms or groups are added (an addition reaction), a reaction in which atoms or groups are removed (an elimination reaction), a reaction in which atoms or groups replace an atom or group (a substitution reaction), or other processes we will encounter. Often, the overall process is composed of multiple steps. Once you have the overall process in mind, it is time to think about the individual steps that convert starting material(s) into product(s). Predicting complete multi-step mechanisms, then, comes down to learning how to predict the individual steps.
Understanding, as opposed to memorizing, mechanisms is critical to mastering organic chemistry. Although the mechanisms you encounter throughout the course may seem entirely different, they are actually related in fundamental ways. In fact, almost all of the organic reaction mechanisms you will learn are composed of only a few different individual elements (steps) that are put together in various combinations. Your job is to learn these individual mechanism elements, and then understand how to assemble them into the steps of the correct mechanism for the overall reaction.
Fortunately, there are a surprisingly small number of different types of characteristic mechanism elements (patterns of arrows) to be considered when trying to predict individual steps of even complex chemical reactions. For this reason, you should view the prediction of each step in an organic mechanism as essentially a multiple choice situation in which your most common choices are the following:
- Make a new bond between a nucleophile (source for an arrow) and an electrophile (sink for an arrow). Use this element when there is a nucleophile present in the solution as well as an electrophile suitable for reaction to occur.
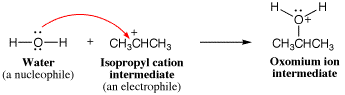
- Break a bond so that relatively stable molecules or ions are created Use this element when there is no suitable nucleophile-electrophile or proton transfer reaction, but breaking a bond can create neutral molecules or relatively stable ions, or both.
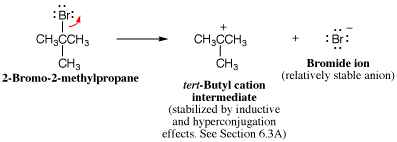
- Add a proton Use this element when there is no suitable nucleophile-electrophile reaction, but the molecule has a strongly basic functional group or there is a strong acid present.
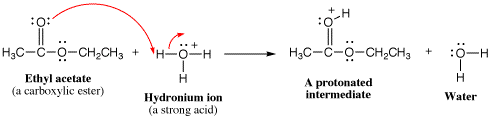
- Take a proton away Use this element when there is no suitable nucleophile-electrophile reaction, but the molecule has a strongly acidic proton or there is a strong base present.
The situation is even simpler than you might expect because 1. and 2. are the functional reverse of each other, as are 3. and 4. in many cases.
Many times, more than one of the four choices occurs simultaneously in the same mechanism step and there are some special situations in which unique or different processes such as electrophilic addition or 1,2 shifts occur. These different processes are described in detail as they are encountered.
In the following sections and chapters of the book, you will learn important properties of the different functional groups that allow you to deduce the appropriate choices for the individual steps in reaction mechanisms. To help you accomplish this, as new mechanisms are introduced throughout the rest of the book, we will label each mechanistic step as one of the four mentioned here when appropriate, emphasizing the common features between even complex mechanisms. When you are able to predict which of the above choices is(are) the most appropriate for a given step in a mechanism, you will then be able to push electrons correctly without relying on memorization. At that point, you will have taken a major step toward mastering organic chemistry!
Common Mistakes in Arrow Pushing
Throughout this book arrow pushing is used to indicate the flow of electrons in the various organic reaction mechanisms that are discussed. A few simple rules for properly performing arrow pushing were introduced in Section 6.2. In this Appendix we examine some of the most common mistakes that students make when first learning arrow-pushing methods and tell you how to avoid them. The mistakes given below are the ones seen most often by the authors during their cumulative dozens of year of experience in teaching Introductory Organic Chemistry.
Backwards Arrows
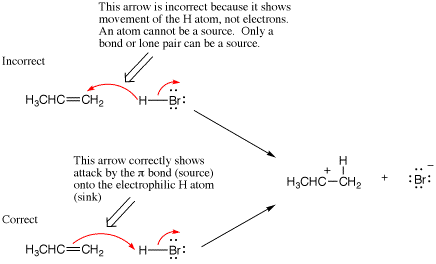
Reversing the direction of one or more arrows during a chemical step is the most common mistake made by students when writing organic reaction mechanisms. Backwards arrow pushing usually derives from a student thinking about the movement of atoms, not the movement of electrons. Hence, to avoid this mistake it is important to remember that arrows depict how electrons move, not where atoms move, within or between chemical structures. Further, one can avoid this mistake by remembering that every arrow must start at an electron source (a bond or lone pair) and terminate at an electron sink (an atom that can accept a new bond or lone pair).
Not Enough Arrows
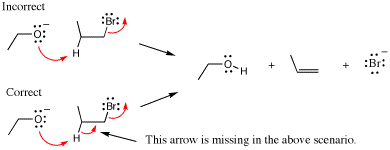
A second common mistake in writing arrow-pushing schemes is to not use enough arrows. This usually results from not keeping track of all lone pairs, bonds made, or bonds broken in a mechanism step. In other words, if you analyze exactly the new position of electrons resulting from each arrow, missing arrows will become evident. In the following example we compare two arrow-pushing scenarios, one of which is missing an arrow. In the incorrect scheme there is no arrow that indicates breaking of the C-H bond of the reactant and formation of the p-bond in the alkene product. Note that when an arrow is missing, the result is commonly too many bonds and/or lone pairs on one atom (see the next section on hypervalency) and not enough bonds or lone pairs on another.
Hypervalency
Another frequent mistake when writing arrow-pushing schemes is to expand the valency of an atom to more electrons than an atom can accommodate, a situation referred to as hypervalency. An overarching principle of organic chemistry is that carbon has eight electrons in its valence shell when present in stable organic molecules (the Octet Rule, Section 1.2). Analogously, many of the other most common elements in organic molecules, such as nitrogen, oxygen, and chlorine, also obey the Octet Rule. There are three common ways in which students incorrectly draw hypervalent atoms: 1) Too many bonds to an atom, 2) Forgetting the presence of hydrogens, and 3) Forgetting the presence of lone pairs.
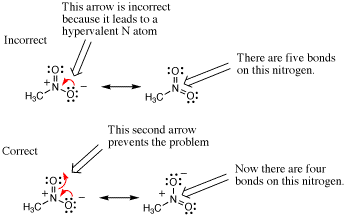
In the following case an arrow is used to depict a potential resonance structure of nitromethane. However, the result is a nitrogen atoms with 10 electrons in its valence shell because there are too many bonds to N. Such mistakes can be avoided by remembering to draw all bonds and lone pairs on an atom so that the total number of electrons in each atoms valence shell is apparent.
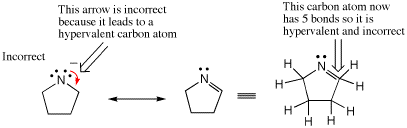
Another common way students mistakenly end up with a hypervalent atom is to forget the presence of hydrogens that are not explicitly written. When using stick diagrams to write organic chemical structures not all the hydrogens are drawn, and hence it is common to forget them during an arrow pushing exercise. The following example shows two proposed resonance contributing structures of an amide anion. The arrow drawn on the molecule to the left is incorrect because it depicts the formation of a new bond to a carbon that already has four bonds. When both bonds to hydrogen are drawn explicitly as on the structure farthest to the right, it is clear there are now five bonds around the indicated carbon atom. Notice also that the negative charge was lost upon drawing the contributing structures on the right, providing another clear signal that something was wrong because overall charge is always conserved when arrows are drawn correctly.
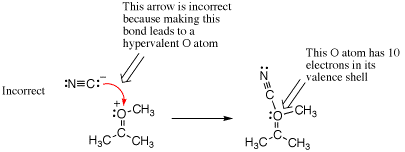
Another common way to make a hypervalency mistake is by forgetting to count all lone pairs of electrons. The following example shows a negatively charged nucleophile incorrectly adding to the formal positive charge on an alkylated ketone. This may look correct because atoms with positive and negative charges are being directly combined, but when counting bonds and lone pairs of electrons, it is found that the oxygen ends up with 10 electrons overall. Hence, this is a mistake.
Mixed Media Errors
Acids and bases are catalysts, reactants, products, and intermediates in many organic chemistry transformations. When writing mechanisms for reactions involving acids and bases, there are three general rules that will help guide you in depicting the correct mechanism.
Do not show the creation of a strong acid for a mechanism of a reaction that is performed in strongly basic media.
Do not show the creation of a strong base for a mechanism of a reaction that is performed in strongly acidic media.
In strongly acidic media, all the intermediates and products will be either neutral or positively charged, while in strongly basic media, all the products and intermediates will be neutral or negatively charged.
The reason for these rules is that significant extents of strong acids and bases cannot co-exist simultaneously in the same medium because they would rapidly undergo a proton transfer reaction before anything else would happen in the solution.
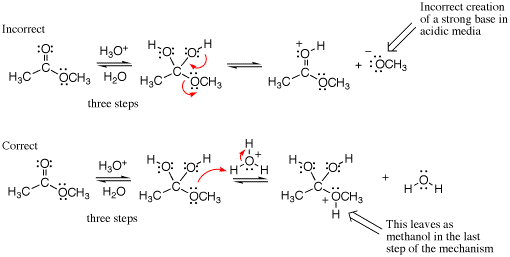
An example of a mixed media error is given below. The first example shows a strong base being created although the reaction is performed under acidic conditions (see conditions over the first equilibrium arrows). Not shown are the three steps that lead to the intermediate drawn. A mistake is made in the arrow pushing because a strong base (methoxide) is generated as the leaving group even though the reaction is run in strong acid. In the correct mechanism, the next step would be protonation of the ether oxygen atom followed by loss of methanol in the last step (not shown) to give a carboxylic acid product.
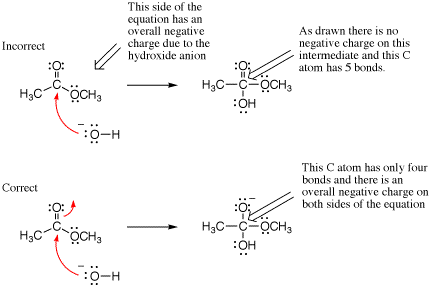
Failing to conserve charge
Overall charge must be conserved in all mechanism steps. Failure to conserve overall charge could be caused by some of the preceding errors (hypervalency, failure to draw arrows, mixed media errors), but we mention it by itself because it is always helpful to check that your arrow pushing is consistent by confirming that overall charge conservation is obeyed. In the example shown below, an arrow is missing leading to a neutral intermediate even thought the overall charge on the left side of the equation was minus one. Notice there are five bonds to carbon on the intermediate (hypervalency), providing another obvious indication that something was incorrect in the mechanism step as drawn.