Other Reactions of Amides
- Page ID
- 3944
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page explains the reason for the lack of basic character in amides, and describes their dehydration to give nitriles, reaction with bromine and sodium hydroxide solution to form primary amines with one less carbon atom (the Hofmann degradation), and their reduction using LiAlH4.
The lack of base character in amides
Unusually for compounds containing the -NH2 group, amides are neutral. This section explains why -NH2 groups are usually basic and why amides are different.
The usual basic character of the -NH2 group
Simple compounds containing an -NH2 group such as ammonia, NH3, or a primary amine like methylamine, CH3NH2, are weak bases. A primary amine is a compound where the -NH2 group is attached to a hydrocarbon group. The active lone pair of electrons on the nitrogen atom in ammonia can combine with a hydrogen ion (a proton) from some other source - in other words it acts as a base.
With a compound like methylamine, all that has happened is that one of the hydrogen atoms attached to the nitrogen has been replaced by a methyl group. It doesn't make a huge amount of difference to the lone pair and so ammonia and methylamine behave similarly.

For example, if these compounds are dissolved in water, the nitrogen lone pair takes a hydrogen ion from a water molecule - and equilibria like these are set up:
\[ NH_3 (aq) + H_2O (l) \rightleftharpoons NH_4^+(aq) + OH^- (aq)\]
\[ CH_3NH_2 (aq) + H_2O (l) \rightleftharpoons CH_3NH_3^+(aq) + OH^- (aq)\]
Notice that the reactions are reversible. In both cases the positions of equilibrium lie well to the left. These compounds are weak bases because they don't hang on to the incoming hydrogen ion very well. Both ammonia and the amines are alkaline in solution because of the presence of the hydroxide ions, and both of them turn red litmus blue.
Why doesn't something similar happen with amides?
Amides are neutral to litmus and have virtually no basic character at all - despite having the -NH2 group. Their tendency to attract hydrogen ions is so slight that it can be ignored for most purposes. We need to look at the bonding in the -CONH2 group.
Like any other double bond, a carbon-oxygen double bond is made up of two different parts. One electron pair is found on the line between the two nuclei - this is known as a sigma bond. The other electron pair is found above and below the plane of the molecule in a \(pi\) bond.
A \(pi\) bond is made by sideways overlap between p orbitals on the carbon and the oxygen. In an amide, the lone pair on the nitrogen atom ends up almost parallel to these p orbitals, and overlaps with them as they form the pi bond.
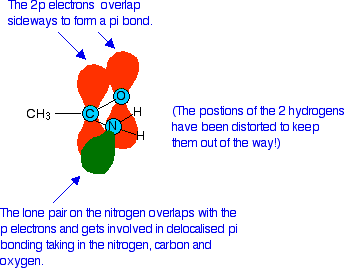
The result of this is that the nitrogen lone pair becomes delocalized - in other words it is no longer found located on the nitrogen atom, but the electrons from it are spread out over the whole of that part of the molecule. This has two effects which prevent the lone pair accepting hydrogen ions and acting as a base:
- Because the lone pair is no longer located on a single atom as an intensely negative region of space, it isn't anything like as attractive for a nearby hydrogen ion.
- Delocalization makes molecules more stable. For the nitrogen to reclaim its lone pair and join to a hydrogen ion, the delocalization would have to be broken, and that will cost energy.
The dehydration of amides
Amides are dehydrated by heating a solid mixture of the amide and phosphorus(V) oxide, P4O10. Water is removed from the amide group to leave a nitrile group, -CN. The liquid nitrile is collected by simple distillation. For example, with ethanamide, you will get ethanenitrile.

The Hofmann Degradation
The Hofmann degradation is a reaction between an amide and a mixture of bromine and sodium hydroxide solution. Heat is needed. The net effect of the reaction is a loss of the -CO- part of the amide group. You get a primary amine with one less carbon atom than the original amide had.
The general case would be (as a flow scheme):
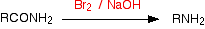
If you started with ethanamide, you would get methylamine. The full equation for the reaction is:
\[ CH_3CONH_2 + Br_2 + 4NaOH \rightarrow CH_3NH_2 + Na_2CO_3 + 2NaBr + 2H_2O\]
The Hofmann degradation is used as a way of cutting a single carbon atom out of a chain.
The reduction of amides
Amides can be reduced to primary amines by reaction with lithium tetrahydridoaluminate, LiAlH4, in dry ether (ethoxyethane) at room temperature. The initial reaction is followed by treatment with dilute acid, such as dilute sulphuric or hydrochloric acid.
For example, if you reduce ethanamide, you will get ethylamine.
\[ CH_3CONH_2 + 4[H] \rightarrow CH_3CH_2NH_2 + H2O\]
An overall equation with hydrogen in square brackets is fine for this level. You might notice that this is a slightly different reduction from the one that happens when LiAlH4 reduces the carbon-oxygen double bond in an aldehyde or ketone. In those cases, the oxygen remains in the final molecule, and you get an -OH group formed. Here, the oxygen is removed entirely.
Contributors
Jim Clark (Chemguide.co.uk)


