Introduction to Aldehydes and Ketones
- Page ID
- 748
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Introduction
A comparison of the properties and reactivity of aldehydes and ketones with those of the alkenes is warranted, since both have a double bond functional group. Because of the greater electronegativity of oxygen, the carbonyl group is polar, and aldehydes and ketones have larger molecular dipole moments (D) than do alkenes. The resonance structures in Figure 1 illustrate this polarity, and the relative dipole moments of formaldehyde, other aldehydes and ketones confirm the stabilizing influence that alkyl substituents have on carbocations (the larger the dipole moment the greater the polar character of the carbonyl group). We expect, therefore, that aldehydes and ketones will have higher boiling points than similar sized alkenes. Furthermore, the presence of oxygen with its non-bonding electron pairs makes aldehydes and ketones hydrogen-bond acceptors, and should increase their water solubility relative to hydrocarbons. Specific examples of these relationships are provided in the following table.
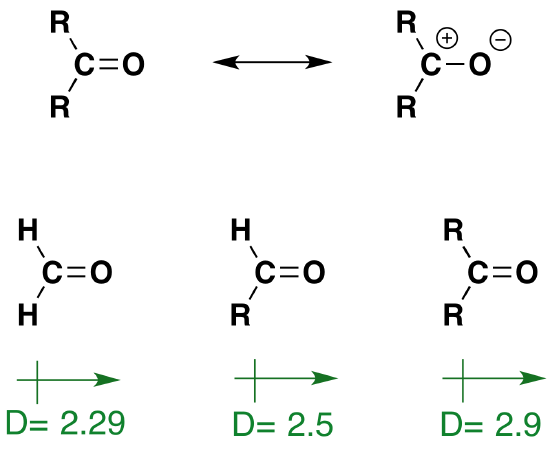
Figure 1: Resonance structures
|
Compound |
Mol. Wt. |
Boiling Point |
Water |
|
(CH3)2C=CH2 |
56 |
-7.0 ºC |
0.04 g/100 |
|
(CH3)2C=O |
58 |
56.5 ºC |
infinite |
|
CH3CH2CH2CH=CH2 |
70 |
30.0 ºC |
0.03 g/100 |
|
CH3CH2CH2CH=O |
72 |
76.0 ºC |
7 g/100 |
|
|
96 |
103.0 ºC |
insoluble |
|
|
98 |
155.6 ºC |
5 g/100 |
The polarity of the carbonyl group also has a profound effect on its chemical reactivity, compared with the non-polar double bonds of alkenes. Thus, reversible addition of water to the carbonyl function is fast, whereas water addition to alkenes is immeasurably slow in the absence of a strong acid catalyst. Curiously, relative bond energies influence the thermodynamics of such addition reactions in the opposite sense.
The C=C of alkenes has an average bond energy of 146 kcal/mole. Since a C–C σ-bond has a bond energy of 83 kcal/mole, the π-bond energy may be estimated at 63 kcal/mole (i.e. less than the energy of the sigma bond). The C=O bond energy of a carbonyl group, on the other hand, varies with its location, as follows:
| H2C=O | 170 kcal/mole |
| RCH=O | 175 kcal/mole |
| R2C=O | 180 kcal/mole |
The C–O σ-bond is found to have an average bond energy of 86 kcal/mole. Consequently, with the exception of formaldehyde, the carbonyl function of aldehydes and ketones has a π-bond energy greater than that of the sigma-bond, in contrast to the pi-sigma relationship in C=C. This suggests that addition reactions to carbonyl groups should be thermodynamically disfavored, as is the case for the addition of water. All of this is summarized in the following diagram (ΔHº values are for the addition reaction).
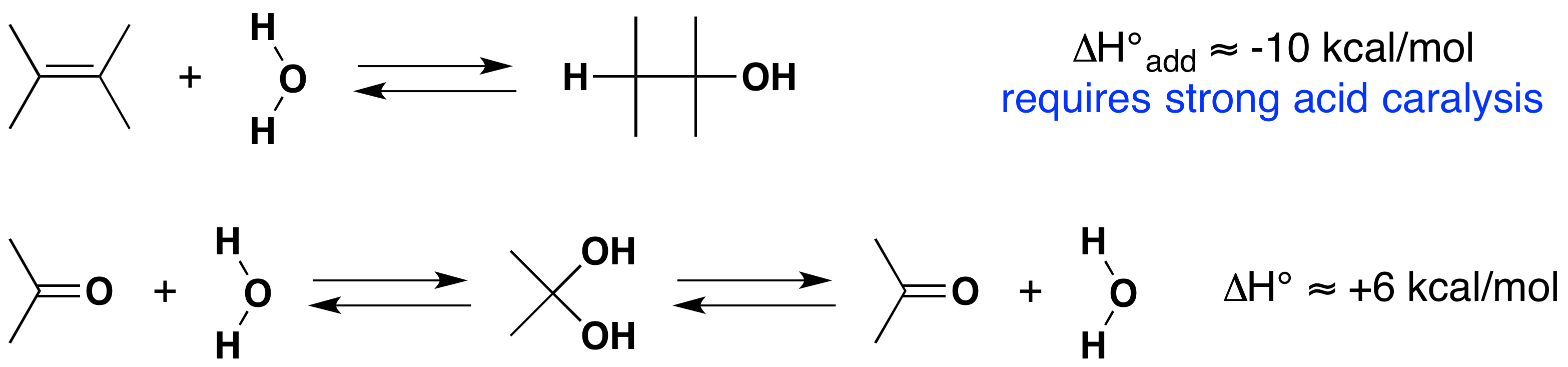
Although the addition of water to an alkene is exothermic and gives a stable product (an alcohol), the uncatalyzed reaction is extremely slow due to a high activation energy. The reverse reaction (dehydration of an alcohol) is even slower, and because of the kinetic barrier, both reactions are practical only in the presence of a strong acid.
In contrast, both the endothermic addition of water to a carbonyl function, and the exothermic elimination of water from the resulting geminal-diol are fast. The inherent polarity of the carbonyl group, together with its increased basicity (compared with alkenes), lowers the transition state energy for both reactions, with a resulting increase in rate. Acids and bases catalyze both the addition and elimination of water. Proof that rapid and reversible addition of water to carbonyl compounds occurs is provided by experiments using isotopically labeled water. If a carbonyl reactant composed of 16O (colored blue above) is treated with water incorporating the 18O isotope (colored red above), a rapid exchange of the oxygen isotope occurs. This can only be explained by the addition-elimination mechanism shown here.
Contributors
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry