4.7: Reaction Work-Ups
- Page ID
- 96881
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Purpose of a Work-Up
When the goal of an experiment is to conduct a reaction and isolate the product, the general sequence of events is shown in Table 4.4.
 |
 |
 |
 |
| a) Conduct the reaction. | b) Perform multiple extractions and/or washes to partially purify the desired product. | c) Remove trace water with a drying agent. d) Filter or decant the drying agent. |
e) Remove the solvent with a rotary evaporator f) Further purify if necessary. |
A key step in this sequence comes immediately after the reaction is complete, and is called the reaction "work-up" (step b) in Table 4.4). The work-up refers to methods aimed at purifying the material, and most commonly occur in a separatory funnel. Solutions are added to the funnel to either extract or wash the mixture, with the goal of isolating the product from excess reagents, catalysts, side products, solvents, or compounds formed from side reactions.
Common Washes
Water
The most common wash in separatory funnels is probably water. Water is cheap, non-hazardous, and works well to remove may impurities found alongside a desired product.
Water can potentially remove water-soluble impurities from an organic layer, as long as they are present in quantities that do not exceed their water solubility. The following are common materials that can be removed with a water wash: unconsumed acid or base, many ionic salts, and compounds that can hydrogen bond with water (have an oxygen or nitrogen atom) and are relatively small (e.g. \(\ce{CH_3CH_2OH}\) or \(\ce{CH_3COCH_3}\)).
To demonstrate the effectiveness of a water wash, a Fischer esterification reaction was conducted to produce isoamyl acetate (Figure 4.38). In this reaction, an excess of acetic acid is used to drive the reaction through Le Chatelier's principle, and the acetic acid had to be removed from the product during the purification process.
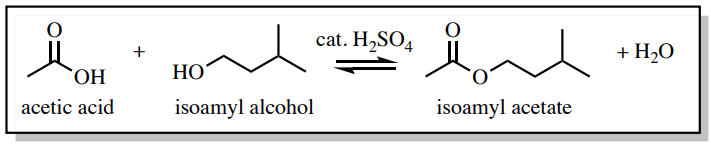
The \(\ce{^1H}\) NMR spectrum in Figure 4.39a was taken of the reaction mixture immediately after ceasing heating and before the work-up. As expected, a significant signal for acetic acid is seen at \(2.097 \: \text{ppm}\).
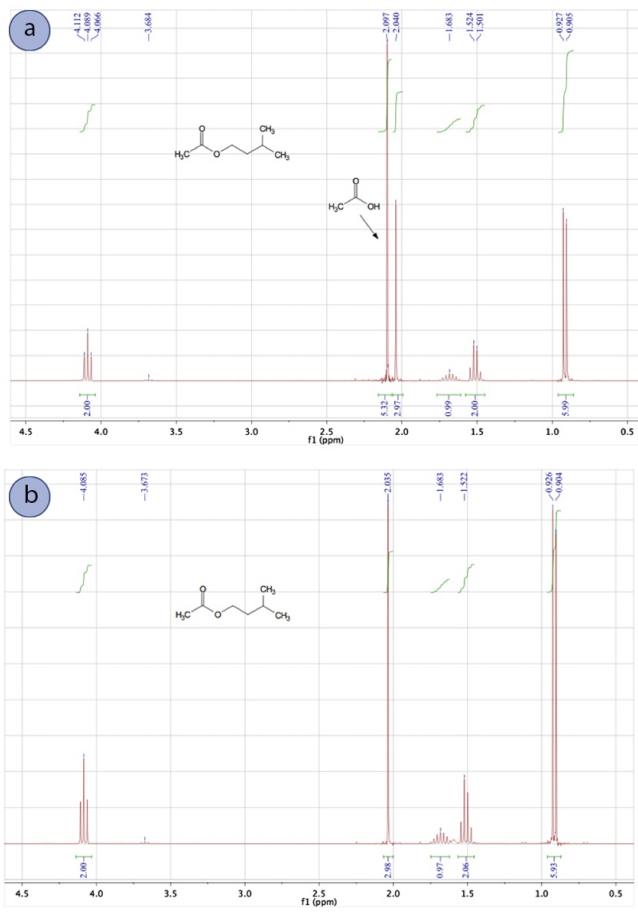
The reaction was then "worked up" by pouring the reaction mixture into a separatory funnel and washing the organic layer with water, sodium bicarbonate, and brine in succession. The main purpose of the water wash was to remove the majority of the catalytic sulfuric acid and the excess acetic acid, while the sodium bicarbonate wash neutralized the rest. The \(\ce{^1H}\) NMR spectrum of the final product (Figure 4.39b) showed the washes were effective as the acetic acid signal at \(2.097 \: \text{ppm}\) is absent.
The sodium bicarbonate wash in this example was necessary (and discussed in the next section) because a water wash alone may not fully remove the acetic acid. It's important to know that when a compound is "water soluble" it does not necessarily mean it is "organic insoluble", a common misconception that arises from the "like dissolves like" principle. For example, acetic acid has a \(K\) of 0.5 when partitioning between diethyl ether and water, meaning acetic acid favors the aqueous layer only twice as much as the organic layer.\(^4\) The ability of acetic acid and other polar compounds to dissolve in the organic layer of a separatory funnel should not be ignored.
Sodium Bicarbonate and Sodium Carbonate
How They Work
A normal part of many work-ups includes neutralization. It is important to neutralize any organic solvent that was exposed to an acidic or basic solution as trace acid or base may cause undesired reactions to occur when the solutions are concentrated. Also, samples intended for GC analysis must be neutral as acidic solutions degrade the polymeric coating of the GC column. In addition, it is preferable to manipulate neutral materials rather than acidic or basic ones, as spills are then less hazardous.
Aqueous solutions of saturated sodium bicarbonate \(\left( \ce{NaHCO_3} \right)\) and sodium carbonate \(\left( \ce{Na_2CO_3} \right)\) are basic, and the purpose of these washes is to neutralize an organic layer that may contain trace acidic components. Even if an organic layer should not in theory dissolve very polar components such as acid, acid sometimes "hitches a ride" on polar components that may dissolve in an organic layer, such as small amounts of alcohols or water.

The following reactions occur between bicarbonate ion (1), carbonate ion (2) and acid \(\left( \ce{H^+} \right)\) during a wash:
\[\ce{HCO_3^-} \left( aq \right) + \ce{H^+} \left( aq \right) \rightarrow \ce{H_2CO_3} \left( aq \right) \rightleftharpoons \ce{H_2O} \left( l \right) + \ce{CO_2} \left( g \right) \tag{1}\]
\[\ce{CO_3^{2-}} \left( aq \right) + \ce{H^+} \left( aq \right) \rightarrow \ce{HCO_3^-} \left( aq \right) \tag{2}\]
The initial product of reaction (1) is carbonic acid \(\left( \ce{H_2CO_3} \right)\), which is in equilibrium with water and carbon dioxide gas. This means that solutions of bicarbonate often bubble during a neutralization wash in a separatory funnel. The product of reaction (2) is the bicarbonate ion, which can subsequently undergo reaction (1). This means that solutions of carbonate ion also often bubble during neutralizations.
Safety note: To prevent excess pressure form being generated by the release of carbon dioxide gas into a separatory funnel during neutralization, the layers should be gently swirled together before placement of the stopper. They should be vented directly after inversion, and more frequently than usual. Figure 4.41 shows a strongly acidic organic layer (top) in contact with an aqueous solution of \(10\%\) sodium bicarbonate (bottom). A vigorous stream of bubbles is seen originating from a small portion of organic layer trapped on the bottom of the funnel. The bubbling was even more vigorous when the layers were mixed together.

Testing the pH After a Wash
To test whether a base wash with \(\ce{NaHCO_3}\) or \(\ce{Na_2CO_3}\) was effective at removing all the acid from an organic layer, it is helpful to test the pH. It is not possible to test the pH of an organic solution directly, however it is possible to test the pH of an aqueous solution that the organic solution has been in contact with. If the aqueous layer is on the top of a separatory funnel, insert a glass stirring rod into the top layer and touch the wet rod to blue litmus paper. An acidic solution turns blue litmus paper pink (or red), while a neutral or basic solution gives blue litmus paper only a darkened "wet" appearance (Figure 4.42d). If the litmus paper turns pink at all\(^5\), the base wash has not fully neutralized the organic layer, and subsequent base washes are needed.
If the aqueous layer is on the bottom of the separatory funnel, test an "aliquot" of the aqueous layer (or tiny sample) on litmus paper through the following method:

- With a finger placed atop a glass pipette, insert the pipette into the separatory funnel so the tip is positioned in the bottom aqueous layer (Figure 4.42a).
- Remove the finger on the pipette to allow a sample of the aqueous layer to enter the pipette through capillary action (Figure 4.42b).
- With a finger placed atop the glass pipette again, remove the pipette from the separatory funnel. A bit of liquid should remain in the pipette tip, an aliquot of the bottom layer (Figure 4.42c).
- Touch the aliquot to blue litmus paper and observe the color (Figure 4.42d).
- If the litmus paper turns pink at all, the base wash has not fully neutralized the organic layer, and subsequent base washes are needed.
Brine (Saturated \(\ce{NaCl}\))
In some experiments, an organic layer may be washed with brine, which is a saturated solution of \(\ce{NaCl} \left( aq \right)\). The purpose of this wash is to remove large amounts of water than may be dissolved in the organic layer. Although the organic layer should always be later exposed to a drying agent (e.g. anhydrous sodium sulfate, magnesium sulfate, or calcium chloride), these reagents at best remove only small amounts of water.

The organic solvents that require a brine wash before exposure to a solid drying agent are diethyl ether and ethyl acetate. These solvents dissolve large quantities of water in comparison to other solvents (Table 4.5).
| Solvent | Grams water dissolved in 100 mL solvent |
|---|---|
| Diethyl ether | 1.24 g |
| Ethyl acetate | 2.92 g |
| Dichloromethane (\(CH_2Cl_2\)) | 0.32 g |
| Hexanes | 0.007 g |
Brine works to remove water from an organic layer because it is highly concentrated (since \(\ce{NaCl}\) is so highly water soluble). A saturated \(\ce{NaCl} \left( aq \right)\) solution is highly ordered, causing a large motivation for water to draw into the solution from the organic layer to increase the entropy of the salt solution (to dilute the solution).
Figure 4.44 shows a qualitative difference in the amount of water present in an organic layer with and without the use of a brine wash. Ethyl acetate was shaken with water (Figure 4.44a), then dried with a portion of anhydrous \(\ce{MgSO_4}\). The large clumps of drying agent in Figure 4.44b indicate that this ethyl acetate layer is still noticeably wet. Ethyl acetate was then shaken with brine (Figure 4.44c), and dried with the same quantity of anhydrous \(\ce{MgSO_4}\). There is little clumping of the drying agent in this ethyl acetate layer, and fine particles are seen (Figure 4.44d), signifying this layer contained very little water.

If drying agents are used to remove water, you might wonder "Why bother with brine; why not use lots of drying agent when the time comes?" The main reason to limit the amount of water present in an organic solution before the drying agent step is that the drying agent will often adsorb compound along with water. Using as little as possible will maximize the yield.
To demonstrate, Figure 4.45 shows an ethyl acetate solution that has a faint pink tint because it contains some dissolved red food dye. the solution was swirled with white anhydrous \(\ce{MgSO_4}\), and the drying agent turned pink as it adsorbed the red food dye compound (Figure 4.45a). Addition of more anhydrous \(\ce{MgSO_4}\) made the drying agent pinker (Figure 4.45b), as more dye was removed from solution. In this example, even after filter and rinsing the drying agent with additional solvent, the drying agent remained pink (Figure 4.45c). Thus, the more drying agent that is used, the more compound that may be irrecoverably lost.

Decreasing Water Solubility of Organic Compounds ("Salting Out")
Saturated ionic solutions may be used to decrease the solubility of organic compounds in the aqueous layer, allowing more of a compound to dissolve in the organic layer. If a desired product can hydrogen bond with water and is relatively small, it may be difficult to keep it in the organic layer when partitioning with an aqueous phase (\(K\) will be <1). However, the equilibrium can favor the organic layer if all aqueous washes contain high concentrations of ions (e.g. saturated \(\ce{NaHCO_3}\), \(\ce{NaCl}\), or \(\ce{NH_4Cl}\)). With water being so tightly "occupied" in dissolving the ions in these solutions, they are less capable of dissolving organic compounds. Additionally, ionic solutions have high dielectric constants, making them less compatible with organic compounds.
Figure 4.47 shows how brine affects the partitioning of red food dye in ethyl acetate and aqueous solutions. Figure 4.47a shows addition of one drop of red food dye to a layer of water in a separatory funnel, and the dye dissolves easily even without swirling. Figure 4.47b shows the water layer containing the dye after shaking with a portion of ethyl acetate. The organic layer has only a very faint pink color, signifying that little dye has dissolved. The dye has obviously partitioned toward the aqueous layer, which is consistent with its very polar structure (Figure 4.46).

Figure 4.47c shows addition of one drop of red food dye to a brine solution, and the dye does not appear to mix with the brine at all. Figure 4.47d shows the brine layer containing the dye after shaking with a portion of ethyl acetate. The organic layer is pinker, signifying that more dye has now partitioned toward the organic layer. the polar dye molecules are much less soluble in the brine solution than in pure water (they have been "salted out"). In fact, some of the dye precipitated in the funnel (Figure 4.47d) as it had such low solubility in both brine and ethyl acetate.

Drying Agents
Why They are Used
An organic layer is always treated with a drying agent after having been exposed to water in a separatory funnel (step c) in Table 4.4). Drying agents are anhydrous inorganic materials that favorably form "hydrates", which incorporate water molecules into their solid lattice structure (for example, \(\ce{Na_2SO_4} \cdot 7 \ce{H_2O}\)). A drying agent is swirled with an organic solution to remove trace amounts of water.
Many organic solvents dissolve a significant portion of water (Table 4.6) that must be removed before rotary evaporation, or else water will be found in the concentrated product. After solvent removal using a rotary evaporator, it occasionally happens that so much water is present that droplets or a second layer is seen amongst the oily liquid in a round-bottomed flask. The presence of water with the product makes the yield inaccurate, and water also must be removed before GC-MS analysis, as water is incompatible with mass-spectrometer detectors.
| Solvent | Grams water dissolved in 100 mL solvent |
|---|---|
| Diethyl ether | 1.24 g |
| Ethyl acetate | 2.92 g |
| Dichloromethane (\(CH_2Cl_2\)) | 0.32 g |
| Hexanes | 0.007 g |
Drying agents must be used with even relatively nonpolar organic solvents that do not theoretically dissolve much water, as water may cling to the sides of the separatory funnel and inadvertently travel with the organic layer while draining. Additionally, solutes dissolved in an organic layer with polar functional groups (e.g. alcohols, carboxylic acids) can hydrogen-bond with water and increase the likelihood of water dissolving in the organic layer.
Types of Drying Agents

Drying agents (Figure 4.48) remove trace amounts of water from organic solutions by forming hydrates. The most useful drying agents indicate when they have completely absorbed all of the water from the solution. Anhydrous magnesium sulfate \(\left( \ce{MgSO_4} \right)\) is a fine, loose powder (Figure 4.49a), but its hydrate is clumpy and often clings to the glass (Figure 4.49b). A typical drying procedure is to add anhydrous \(\ce{MgSO_4}\) to an organic solution until it stops clumping and fine particles are seen, which indicate that there is no longer water available to form the clumpy hydrates.

Anhydrous calcium sulfate \(\left( \ce{CaSO_4} \right)\), can be purchased containing a cobalt compound that is blue when dry and pink when wet (this is then sold under the name Drierite, Figure 4.49c+d). In this way, blue Drierite can be used as a visual indicator for the presence of water.\(^8\)
The most common drying agents used to remove water from organic solutions are anhydrous sodium sulfate \(\left( \ce{Na_2SO_4} \right)\) and anhydrous magnesium sulfate \(\left( \ce{MgSO_4} \right)\). Many chemists consider \(\ce{MgSO_4}\) the "go-to" drying agent as it works quickly, holds a lot of water for its mass, and the hydrates are noticeably chunkier compared to the anhydrous form, making it easy to see when you've added enough. A drawback to using \(\ce{MgSO_4}\) is that it is a fine powder, and so the solutions must be subsequently filtered to remove the drying agent. Another drawback to \(\ce{MgSO_4}\) is that all fine powders heavily adsorb product on their surface (which is why they must be rinsed with solvent after filtration), and sometimes more granular drying agents are used to minimize the loss of product by adsorption.
In some procedures \(\ce{Na_2SO_4}\) or \(\ce{CaCl_2}\) are used if they seem to work just as well as \(\ce{MgSO_4}\), or if the solution is incompatible with \(\ce{MgSO_4}\) (see Table 4.8). A procedural advantage to these drying agents is that their granules are not easily dispersed, allowing for the solutions to be easily decanted (poured). In many situations drying agents are interchangeable (see Table 4.8 for a survey of drying agents). However, it is most common for desiccators and drying tubes to use \(\ce{CaSO_4}\) or \(\ce{CaCl_2}\) (Figure 4.50), as they can be easily manipulated in their pellet or rock forms.

| Drying Agent | Hydrate formula(s) | Practical Comments | Other Comments |
|---|---|---|---|
| Magnesium sulfate | \(\ce{MgSO_4} \cdot 7 \ce{H_2O}\) | Quickly removes most water, and can hold a lot for its mass (\(0.15\)-\(0.75 \: \text{g}\) water per \(\text{g}\) desiccant).\(^9\) Is a fine powder, so must be gravity filtered. Its high surface area means it will somewhat adsorb compound: be sure to rinse after filtering. | \(\ce{Mg(H_2O)_4^{2+}}\) is somewhat acidic, so is incompatible with highly acid-sensitive groups. |
| Sodium Sulfate |
\(\ce{Na_2SO_4} \cdot 7 \ce{H_2O}\) \(\ce{Na_2SO_4} \cdot 10 \ce{H_2O}\) |
Removes water at a moderate rate, so the solution should be allowed to sit with the drying agent for some time. Can hold a lot of water for its mass (\(1.25 \: \text{g}\) water per \(\text{g}\) desiccant), but may leave small amounts of water remaining. Solutions with \(\ce{Na_2SO_4}\) can usually be decanted. | Cannot dry diethyl ether well unless a brine wash was used. |
| Calcium chloride |
\(\ce{CaCl_2} \cdot 2 \ce{H_2O}\) \(\ce{CaCl_2} \cdot 6 \ce{H_2O}\) |
Quickly removes water well, although larger quantities are needed than other drying agents (holds \(0.30 \: \text{g}\) water per \(\text{g}\) desiccant). If using a fine powder, the solution must be gravity filtered and drying agent rinsed. If using pellets, the solution should be allowed to sit for a few minutes, then decanted. | Absorbs water as well as methanol and ethanol. |
| Calcium sulfate (Drierite) |
\(\ce{CaSO_4} \cdot \frac{1}{2} \ce{H_2O}\) \(\ce{CaSO_4} \cdot 2 \ce{H_2O}\) |
Quickly removes water, but needs large quantities as it holds little water per gram. Are most often used in desiccators and drying tubes, not with solutions. |
Drying Agents Procedure

- The organic solution to be dried must be in an Erlenmeyer flask, as solutions can easily splash out of beakers when swirled.
- First inspect the solution to see if it's homogenous, or if there is a second layer of liquid (typically a puddle on the bottom). If a second layer is noticed, this is probably water and the majority of it should be pipetted out before continuing on (Figure 4.51a). It can be difficult to completely remove a water layer by pipette, so leaving a tiny bit is acceptable.
- Add a small portion of drying agent to the flask,the size of one pea for macroscale work (Figure 4.51b), and swirl the solution (Figure 4.51c). Be sure to close the jar of drying agent when not in use, as the reagents are hygroscopic. After a short period of time, inspect the mixture closely.
- If the entire drying agent clumps into pieces that are much larger than the original size (Figure 4.52b+c), there is still water remaining in the flask. Add another portion of drying agent and swirl.
- A solution is nearing dryness when fine particles are noticed that don't cling to other particles (Figure 4.52a+c) or to the glass when swirled (Figure 4.53a). A wet organic solution can be cloudy, and a dry one is always clear.
- If using anhydrous \(\ce{Na_2SO_4}\), allow the solution to sit for at least 5 minutes before declaring the solution dry, as this reagent takes time to work.


- When the solution is dry, separate the drying agent from the solution:
- If using \(\ce{Na_2SO_4}\), \(\ce{CaCl_2}\) pellets, or \(\ce{CaSO_4}\) rocks, carefully decant the solution into an appropriately sized round-bottomed flask (Figure 4.53b), being sure to fill the flask no more than halfway. Reminder: a mass of the empty flask should be obtained if the solvent will be evaporated on the rotary evaporator.
- If using \(\ce{MgSO_4}\), gravity filter the solution into an appropriately sized round-bottomed flask (Figure 4.53c). When pouring, leave the solid behind as long as possible (essentially decant the solution, but into the funnel lined with filter paper). Solid can slow drainage in the filter paper.
- With all drying agents, rinse the drying agent (in the flask and in the filter funnel) with a few \(\text{mL}\) of fresh organic solvent, and add the rinsing to the round-bottomed flask (Figure 4.53d). Remove the solvent using a rotary evaporator.
\(^4\)A. Seidell, Solubilities of Inorganic and Organic Substances, D. Van Nostrand Company, 1907.
\(^5\)When assessing the result of a litmus paper test, look at the center of the drop. The center is the most concentrated spot, and it's possible a color change may not be seen on the outside where the solution has spread and diluted. Any pink seen on blue litmus paper means the solution is acidic.
\(^6\)From: Fessenden, Fessenden, Feist, Organic Laboratory Techniques, 3\(^\text{th}\) ed., Brooks-Cole, 2001.
\(^7\)From: Fessenden, Fessenden, Feist, Organic Laboratory Techniques, 3\(^\text{th}\) ed., Brooks-Cole, 2001.
\(^8\)Blue Drierite is expensive, so is commonly used by mixing it together with white Drierite (\(\ce{CaSO_4}\) without the cobalt indicator). Pink (wet) Drierite can be dried by spreading it on a watch glass and drying in a \(110^\text{o} \text{C}\) oven overnight.
\(^9\)Grams water per gram of desiccant values are from: J. A. Dean, Lange's Handbook of Chemistry, 15\(^\text{th}\) ed., McGraw-Hill, 1999, Sect. 11.2. \(\ce{CaCl_2}\) value is quoted for the formation of \(\ce{CaCl_2} \cdot 2 \ce{H_2O}\).


