4.4: Which Layer is Which?
- Page ID
- 93532
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Density
It is essential that you know whether the aqueous layer is above or below the organic layer in the separatory funnel, as it dictates which layer is kept and which is eventually discarded. Two immiscible solvents will stack atop one another based on differences in density. The solution with the lower density will rest on top, and the denser solution will rest on the bottom.
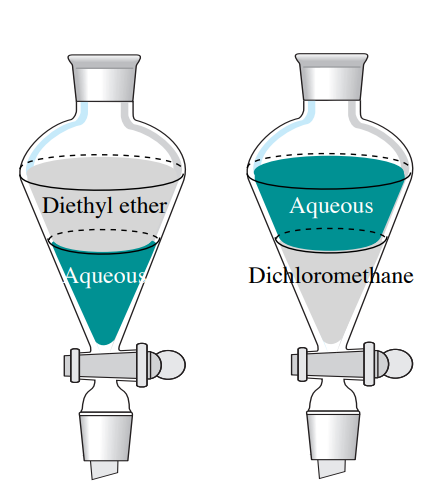
Most non-halogenated organic solvents have densities less than 1 g/mL, so will float atop an aqueous solution (if they are immiscible). A notable exception is that halogenated solvents are denser than water (have densities greater than 1 g/mL), and so will instead sink below aqueous solutions (Table 4.1 and Figure 4.8).
| Solvent | Density (g/mL) |
|---|---|
| Pentane | 0.626 |
| Petroleum Ether (mixture of C5 - C6 hydrocarbons) | 0.653 |
| Hexanes (mixture of 6 hydrocarbons) | 0.655 |
| Diethyl ether | 0.713 |
| Ethyl acetate | 0.902 |
| Water | 0.998 |
| Dicholoromethane (CH2Cl2) | 1.33 |
| Chloroform (CHCl3) | 1.49 |
Many solutions used in separatory funnels are fairly dilute, so the density of the solution is approximately the same as the density of the solvent. For example, if mixing diethyl ether and a \(10\% \: \ce{NaOH} \left( aq \right)\) solution in a separatory funnel, knowledge of the exact density of the \(10\% \: \ce{NaOH}\) solution is not necessary. A \(10\% \: \ce{NaOH} \left( aq \right)\) solution is \(90\%\) water (by mass), meaning the density should be fairly close to the density of water (approximately \(1 \: \text{g/mL}\)). The actual density of a \(10\% \: \ce{NaOH} \left( aq \right)\) solution is \(1.1089 \: \text{g/mL}\), a value only slightly greater than the density of water. The diethyl ether will be the top layer in this situation.
There are times, however, when so may solute particles are dissolved that a solution's density is much greater than the solvent density. For example, a saturated \(\ce{NaCl} \left( aq \right)\) solution has a density around \(1.2 \: \text{g/mL}\) (significantly greater than the density of water), and can cause separation problems with solvents of similar densities like dichloromethane.
How to Determine the Aqueous Layer
Solvent densities may be used to predict which layer is organic and which is aqueous in a separatory funnel, but there are other methods that can be useful in this determination. If unsure which layer is aqueous and which layer is organic, do one of the following things:
- Add a bit of water from a squirt bottle to the separatory funnel (Figure 4.9a) and watch where the water droplets go.
If the top layer is aqueous, the water droplets should mix with the top layer, and they will look as if they disappear. If the bottom layer is aqueous, the water droplets will fall through the top layer to mix with the bottom layer (as indicated by an arrow in Figure 4.9b+c). If it is difficult to track where the water droplets go, also keep track of the volume of the layers: whichever layer increases with the addition of water is the aqueous layer.

- Consider relative volumes of aqueous and organic solvents, based on quantities used in the experiment.
Figure 4.10a shows a \(125 \: \text{mL}\) separatory funnel containing \(10 \: \text{mL}\) hexane and \(100 \: \text{mL}\) water (tinted with blue dye). If these were the quantities used in an experiment, the aqueous layer would have to be the lower layer as it is so much larger. Although unequivocal in this case, it is important to know that the odd shape of the separatory funnel may cause you to misjudge volumes. A separatory funnel with equal volumes of aqueous and organic layers is shown in Figure 4.10b, although the layers rise to different heights in the funnel.

\(^1\)The solvents listed in Table 4.1 are pure compounds except for petroleum ether and hexanes. "Petroleum Ether" contains pentane, 2-methylbutane, 2,2-dimethylpropane, n-hexane, 2-methylpentane, 3-methylpentane, 2,2-dimethylbutane, and 2,3-dimethylbutane. "Hexanes" contains 2-methylpentane, 3-methylpentane, n-hexane, and methylcyclopentane.


