4.3: Uses of Extraction
- Page ID
- 93531
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)There are several reasons to use extraction in the chemistry lab. It is a principal method for isolating compounds from plant materials. Extraction moves compounds from one liquid to another, so that they can be more easily manipulated or concentrated. It also enables the selective removal of components in a mixture.
Extracting Natural Compounds
Fruit and plant leaves are primarily composed of cellulose and water, but also contain "essential oils." a greasy mixture of compounds that capture the "essence" of the plant material's smell and taste. Orange oil is roughly \(95\%\) limonene (Figure 4.3b), and due to its nonpolar structure, can be extracted from its rind into an organic solvent like hexanes or dichloromethane (Figure 4.3a). The oil can then be concentrated and used to flavor or scent foods, cleaning supplies, and candles.
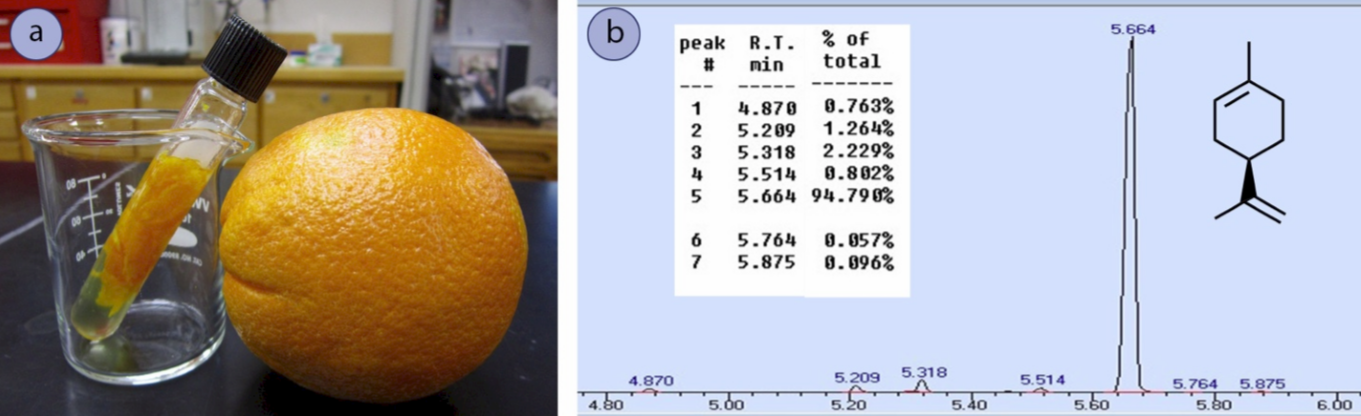
Figure 4.3: a) Orange rind extracted into dichloromethane, b) GC spectrum of orange oil.
In the chemistry lab, essential oils are often extracted from their source using solvents, and analyzed using gas chromatography or spectroscopy.
Transferring Compounds From Layers
Another method for extracting essential oils from fragrant plant materials is through steam distillation (Figure 4.4b). This process often results in the lovely smelling compounds suspended in the aqueous distillate (Figure 4.4c). In order to concentrate the oil, the aqueous suspension is often extracted with a low-boiling organic solvent (Figure 4.4d), which can then be easily removed from the oil.
.png?revision=1)
Figure 4.4: a) Whole cloves, b) Steam distillation of cloves, c) Milky distillate composed of oil and water, d) Using extraction to separate the oil from the water.
Selective Removal of Components
When conducting an experiment that synthesizes a chemical product, a reaction is often complete whenever stirring or heating is ceased. And yet, there are always more steps in the procedure! What commonly happens directly afterwards is to "work-up" the reaction in some way. A work-up refers to methods aimed at isolating the product from the reaction mixture, and often begins by using a separatory funnel and extractions.

For example, imagine that acetic acid and isopentanol have been heated in the presence of an acid catalyst for one hour (Figure 4.5) in order to make isopentyl acetate, an ester that smells of bananas (see reaction scheme in Figure 4.6). After the one-hour time period, there is unfortunately not just the banana-smelling ester in the flask. The flask will also contain byproducts (the water in this case), leftover starting materials if the reaction is incomplete, as well as any catalysts used (\(\ce{H_2SO_4}\) in this case). In this example, there could be five compounds in the reaction flask after heating is ceased (Figure 4.7)!
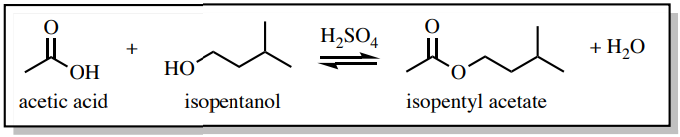
When "working up" this reaction, the resulting mixture is often poured into a separatory funnel along with some water and organic solvent. This produces two layers in the separatory funnel: an aqueous layer and an organic layer.
After shaking this heterogeneous mixture, the compounds distribute themselves based on their solubility. Compounds that have high water solubility favor the aqueous layer while less polar compounds favor the organic layer. In this example, the acid catalyst and residual carboxylic acid or alcohol would likely be drawn into the water layer. The ester would have a greater affinity for the organic layer than the aqueous layer, causing it to be isolated from the other components in the reaction mixture (Figure 4.7).
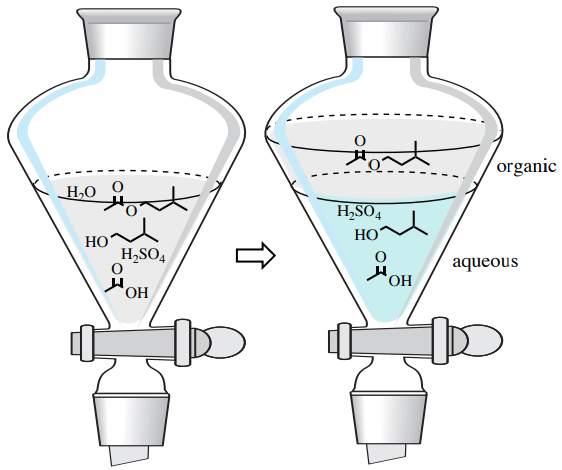
In this example, it is possible that small amounts of alcohol are also drawn into the organic layer, but they could likely be removed with a water "wash." In a wash, the desired compound (e.g. the isopentyl acetate), remains in its current layer of the separatory funnel (in this example the organic layer), and unwanted compounds are removed, or "washed away" into another layer (e.g. the aqueous layer). A wash is different than an extraction, because in an extraction the desired compound moves from its current location (i.e. moves from an aqueous layer to an organic layer), while in washing the desired compound stays in its current layer.


