12.2: Recognizing Nucleophiles
- Page ID
- 216021
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
For the most part, the same criteria used for identifying Bronsted bases can also be used to identify nucleophiles. Here is a summary.
1. The most common type of nucleophiles are those containing atoms with unshared electron pairs, such as the following.

Notice that nonbonding electrons are frequently omitted from formulas. Always remember their presence on oxygen, nitrogen, and negatively charged carbon atoms.
2. A negatively charged species is usually a stronger nucleophile or base than its neutral analog. Thus, hydroxide ion is stronger, both as a base and as a nucleophile, than water.
3. Carbon bonded to a metal has strong negative character, revealed when writing resonance structures. The carbon atom in such molecules is considered a strong nucleophile. Recall the structures of n-butyllithium and Grignard (organomagnesium) reagents from previous notes.
4. The pi bond is a region of high electron density. Pi bonds are not as strong as sigma bonds, which means that pi electrons are more available for reactions because pi bonds ore more easily broken. Molecules containing pi bonds are considered weak nucleophiles or bases, because they can react with strong acids or electrophiles. See examples under Bronsted acid-base chemistry.
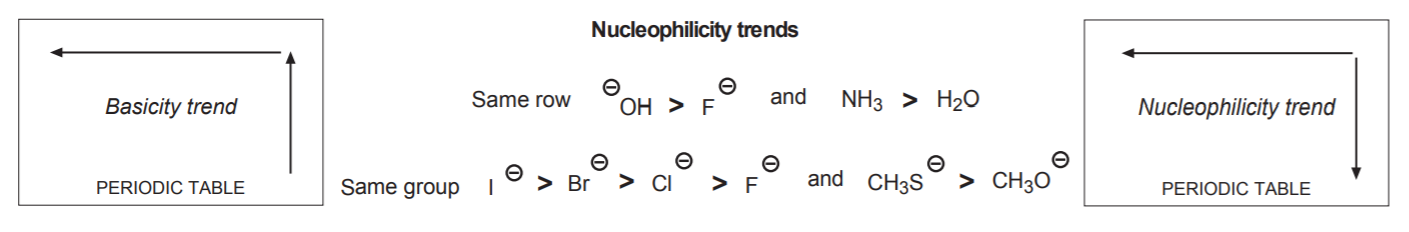
5. Periodic trends in nucleophilicity are such that, other factors being similar, nucleophilicity increases from right to left across the same row, and from top to bottom across the same period or group. Make a note that this trend is different from the basicity trend, which increases in the same way across a row, but from bottom to top within a period.
6. Basic vs. nucleophilic behavior. There are some differences between Bronsted bases and nucleophiles. First, the term base is reserved for substances seeking acidic protons, whereas the term nucleophile is used for substances seeking electron-deficient centers, be they protons or other atoms (most commonly carbon).
Second, in acid-base reactions the size (or bulk) of the base seldom matters. In nucleophilic reactions, the size of the nucleophile can be an important (steric) factor. Smaller is more effective. Thus, although the nucleophilic atom in the two species below is the same (oxygen) and they have similar structures, methoxide ion is a more effective nucleophile than t-butoxide ion, even though they are about equally effective as bases. The larger t-butoxide ion has more difficulty reaching an electrophilic center, which is typically a carbon buried at the core of the molecular structure. That is not a problem when it acts as a base because acidic protons usually lie in the periphery of the molecule and are easily accessible.

methoxide ion smaller ion, better nucleophile
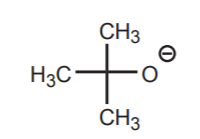
t-butoxide ion larger ion, poorer nucleophile


