22.5: Acidity of Alpha Hydrogen Atoms- Enolate Ion Formation
- Page ID
- 36416
After completing this section, you should be able to
- explain why the alpha hydrogens of carbonyl compounds are more acidic than the hydrogens in a typical hydrocarbon.
- list the properties that make lithium diisopropylamide a suitable reagent for converting a wide range of carbonyl compounds into their enolate anions.
- arrange a given list of carbonyl compounds in order of increasing or decreasing acidity.
- determine whether a given carbonyl‑containing compound is more or less acidic than selected other compounds, such as water, ammonia, alcohols, alkanes, alkenes, alkynes and amines.
- explain why dicarbonyl compounds, such as β‑diketones, are more acidic than compounds that contain only a single carbonyl group.
Make certain that you can define, and use in context, the key terms below.
- β‑diketone
- β‑keto ester
α-hydrogens are weakly acidic because their conjugate base, called an enolate, is stabilized though conjugation with the π orbitals of the adjacent carbonyl. Removal of an α-hydrogen creates an sp2 hybridized carbon anion. The negative charge and lone pair electrons of the carbon anion are contained in an unhybridized p orbital. Orbital overlap with p orbitals from the adjacent carbonyl pi bond allows for the lone pair electrons and negative charge to be shared with the electronegative Carbonyl oxygen, stabilizing the negative charge of the enolate ion.
Enolate conjugation can be represent by two major resonance forms represented by structures A and B which share the negative charge (Shown Below). In resonance form A, the negative charge is on a carbon while in resonance form B the negative change is on an oxygen.
Resonance form B is preferred because oxygen is highly electronegative and better able to stabilize the negative charge. Both resonance form contribute to an overall resonance hybrid but not equally. Due to its stability, resonance form A makes a greater contribution to the resonance hybrid structure. Form A’s increased contribution to the hybrid is seen when considering the electrostatic potential map of an acetone enolate. The red/yellow color shows there is a high electron density around the oxygen. The yellow/green color around the enolate carbon shows that there is a lower but still significant electron density present. Notice that the electron density on the enolate carbon is much larger than the non-enolate carbon (blue color).
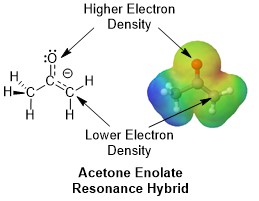
The effect of an adjacent carbonyl on the acidity of α-hydrogens is seen when comparing the pKa of aldehydes (~16-18) and ketones (~19-21) to the pKa of an alkane (~50). The ability of the electronegative oxygen to stabilize a negative charge can be seen by considering the pKa of an allyl C-H bond (~42). Having a C-H bond adjacent to a C=C double bond allows the conjugate base created by deprotonating an allyl C-H bond to be stabilized by conjugation. However, the stability gained only provides a 108 increase in acidity when compared to an alkane. In comparison, the presence of an electronegative oxygen allows the acidity of an α-hydrogen to be 1020 times greater than that of an alkane.
Although α-hydrogens are weakly acidic, typical strong bases such as a hydroxide or alkoxides are only capable of forming the enolate ion in very low concentrations. This can leave a significant concentration of the electrophilic carbonyl carbon remaining, which can react with the bases or the enolate. To achieve the complete deprotonation of aldehyde or ketone reactants to their enolate conjugate bases, a very strong base such as LDA (lithium diisopropylamide) must be used. Complete deprotonation removes the carbonyl groups of the starting material from the reaction mixture and prevents their ability to form unwanted products. Also, the large alkyl substituents sterically hinder the ability of LDA and its corresponding amine (diisopropyl amine) to undergo nucleophilic addition to the carbonyl. In addition to aldehydes and ketones, LDA readily deprotonates a wide variety of carbonyl containing functional groups including esters, amides and nitriles. Ether solvents like tetrahydrofuran (THF) are commonly used for enolate formation reactions using LDA. Certain other strong bases, such as alkyl lithium and Grignard reagents, cannot be used to make enolate anions because they rapidly and irreversibly add to the carbonyl groups. Nevertheless, these very strong bases are useful in the preparation of LDA. By reacting n-butyllithium with diisopropylamine (pKa 36), LDA can be easily formed.

Example

Hydrogen atoms with two or more adjacent carbonyl groups are more acidic than typical α-hydrogens.

The enolate ions of compound such as β-diketones, β-keto-esters, and β-diesters are stabilized through additional resonance forms which share the negative charge with multiple carbonyl oxygens. The acidity of these compounds is increased to the point where weaker bases, such as sodium ethoxide (NaOCH2CH3) can be used to form the enolate.
The pKa and acidic hydrogens of multiple functional groups are shown in the table below.
* = a functional group with two ketones.
| Functional Group | Structure | pKa |
|---|---|---|
| carboxylic acid | HO–(C=O)R | 5 |
| nitro | RCH2–NO2 | 9 |
| β-diketone * | R(O=C)–CH2–(C=O)R | 9 |
| β-ketoester * | R(O=C)–CH2–(C=O)OR | 11 |
| β-diester * | RO(O=C)–CH2–(C=O)OR | 13 |
| amide | RNH–(C=O)R | 15 |
| alcohol | RCH2–OH | 16 |
| aldehyde | RCH2–(C=O)H | 17 |
| ketone | RCH2–(C=O)R | 20 |
| thioester | RCH2–(C=O)SR | 21 |
| ester | RCH2–(C=O)OR | 25 |
| nitrile | RCH2–C≡N | 25 |
| sulfone | RCH2–SO2R | 25 |
| amide | RCH2–(C=O)N(CH3)2 | 30 |
| alkane | CH3–R | 50 |

