18.0: Introduction
- Page ID
- 36364
After completing this section, you should be able to use the terms “ether,” “diethyl ether ” and “ethyl ether ” appropriately in context.
Make certain that you can define, and use in context, the key terms below.
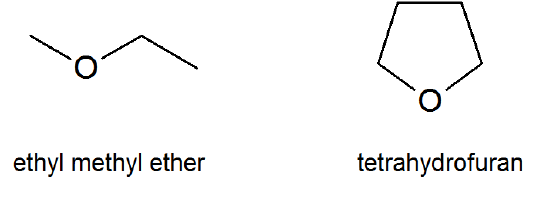
- ether (\(\ce{R-O-R′}\))
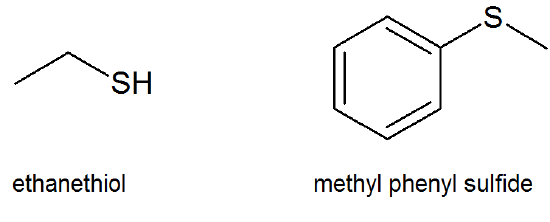
- sulfide (\(\ce{R-S-R′}\))
- thiol (\(\ce{R-S-H}\))
As defined in the textbook, an “ether” is a substance with the general formula (\(\ce{R-O-R′}\)) where \(\ce{R}\) and \(\ce{R′}\) are alkyl, aryl, vinyl or allyl groups. However, the word “ether” is also commonly used to refer to the specific compound, \(\ce{CH3-CH2-O-CH2-CH3}\), which is correctly called “diethyl ether.” Further confusion can arise because some chemists refer to “diethyl ether ” as “ethyl ether.” In this course, “ether ” will be used to refer to the class of compounds with the structure (\(\ce{R-O-R′}\)); diethyl ether will refer to the compound, \(\ce{CH3-CH2-O-CH2-CH3}\); and “ethyl ether ” will not be used.
Ethers and Epoxides
While the general formula for ethers is R-O-R′, keep in mind that there also cyclic ethers like tetrahydrofuran (a common organic solvent) or even epoxides which you first encounter in Section 8.7 in synthesizing diols from alkenes.
Thiols and Sulfides
Thiols (thio alcohols or mercaptans) and sulfides (thioethers) are the sulfur analogues of alcohols and ethers and have the general formulas of R-S-H and R-S-R′, respectively.