16.S: Chemistry of Benzene - Electrophilic Aromatic Substitution (Summary)
- Page ID
- 217189
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Concepts & Vocabulary
- Aromatic compounds don't typically undergo addition reactions.
- Aromatic compounds typically undergo substitution reactions.
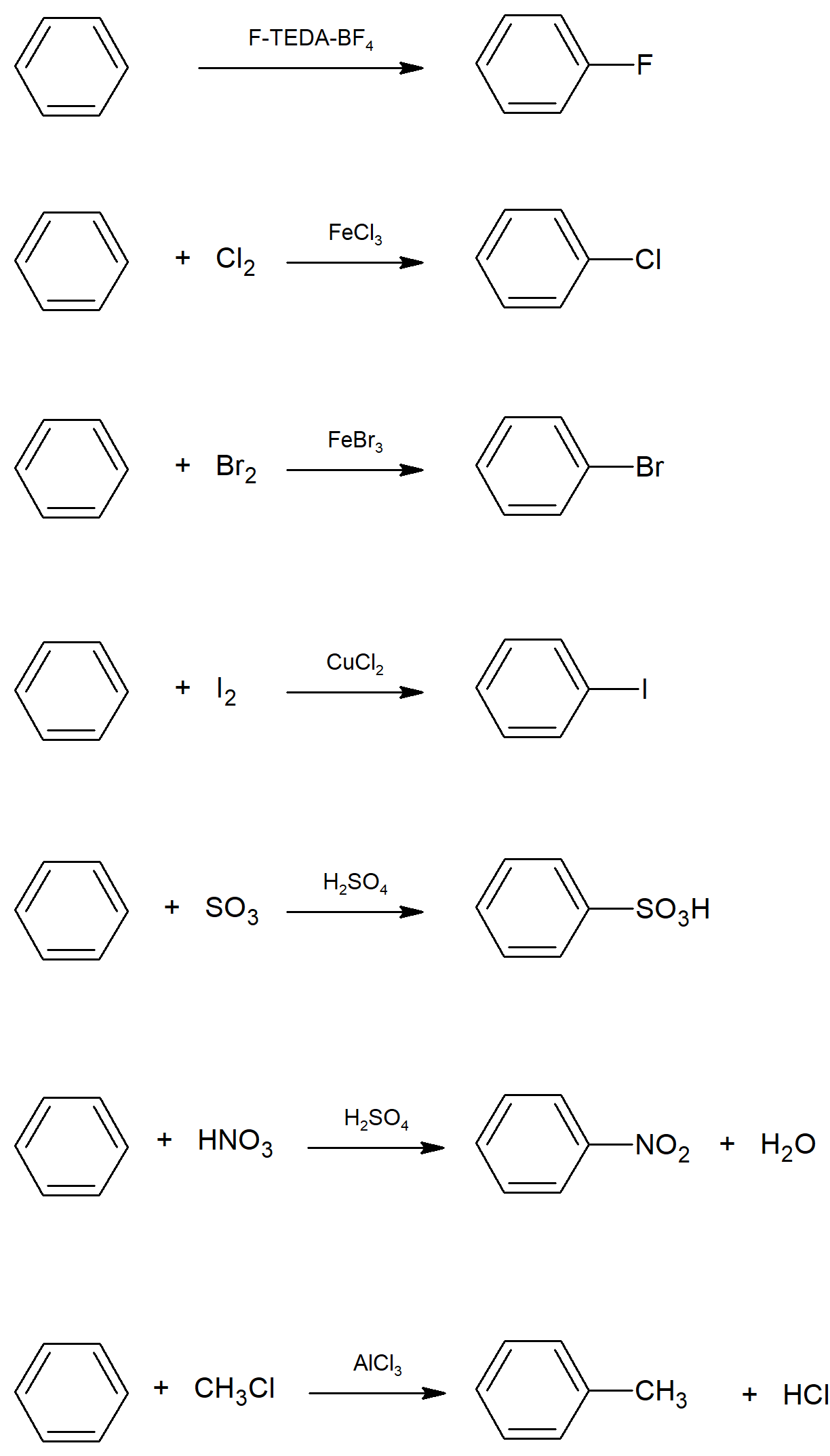
16.1 Electrophilic Aromatic Substitution Reactions: Bromination
- Aromatic molecules only react with strong electrophiles.
- The first step in many electrophilic aromatic substitution mechanisms is activation or formation of the electrophile.
- The electrophilic aromatic substitution mechanism occurs in two steps. The first is addition of the electrophile to the ring and the second is elimination of a hydrogen from the ring to re-form the pi bond and restore aromaticity.
- In bromination of an aromatic ring, molecular bromine (Br2) is reacted with iron tribromide (FeBr3) to form the strongly electrophilic bromine cation and FeBr4. Following this, the aromatic ring is reacted with the bromine cation and adds to the ring to form a benzenonium cation. This molecule then reacts with one of the bromine atoms from FeBr4 to lose a hydrogen forming the product and HBr as well as reforming the iron tribromide.
16.2 Other Aromatic Substitutions
- Aluminum bromide (AlBr3) can be used in place of FeBr3 to create the bromine cation. Also the chlorides of alumnium and iron can also be used to create a chlorine cation which will also undergo electrophilic aromatic substitution.
- Reacting nitric acid and sulfuric acid forms nitronium (NO2+), which will react with aromatics for form nitro compounds.
- Sulfonation of aromatics can be accomplished by reacting with sulfur trioxide and sulfuric acid to yield sulfonic acids.
16.3 Alkylation and Acylation of Aromatic Rings - The Friedel-Crafts Reaction
- Friedel-Crafts reactions incorporate activation of alkyl and acyl halides by reacting them with a Lewis Acid catalyst, AlCl3.
- Friedel-Crafts alkylations allow for adding alkyl chains to aromatic rings.
- After activation with aluminum chloride, alkyl carbocations can undergo rearrangement if it leads to a more stable intermediate.
- Friedel-Crafts acylations add alkyl ketones to aromatic rings.
16.4 Substituent Effects in Substituted Aromatic Rings
- Aromatic inductive effects are caused by differences in electronegativity between atoms bonded to the ring and the ring carbons.
- Most common heteroatoms (N, O, halogens) donate electron density toward the ring inductively.
- Aromatic resonance effects are caused by conjugation of substituents with the pi bonds of the ring.
- Substituents that increase the electron density of the ring activate the ring (make more reactive) toward electrophilic substitution.
- Substituents that decrease the electron density of the ring deactivate the ring (make less reactive) toward electrophilic substitution.
16.4b An Explanation of Substituent Effects
- Steric effects can increase para substitution as ortho/para directors become larger.
- Activating groups are ortho/para (o, p) directors.
- Deactivating groups are meta directors.
- Alkyl groups inductively donate electron density to the ring making them o, p directors.
- Groups with an O or N attached to the aromatic ring are activators and o, p directors due to resonance.
- Groups with a pi bond attached to the aromatic ring are deactivators and m directors due to resonance.
- Halogens are o, p directors, but are deactivators.
16.5 Trisubstituted Benzenes: Additivity of Effects
- When there is more than one group attached to an aromatic ring, these groups may reinforce directing effects (cooperative) or have opposing directing effects (non-cooperative).
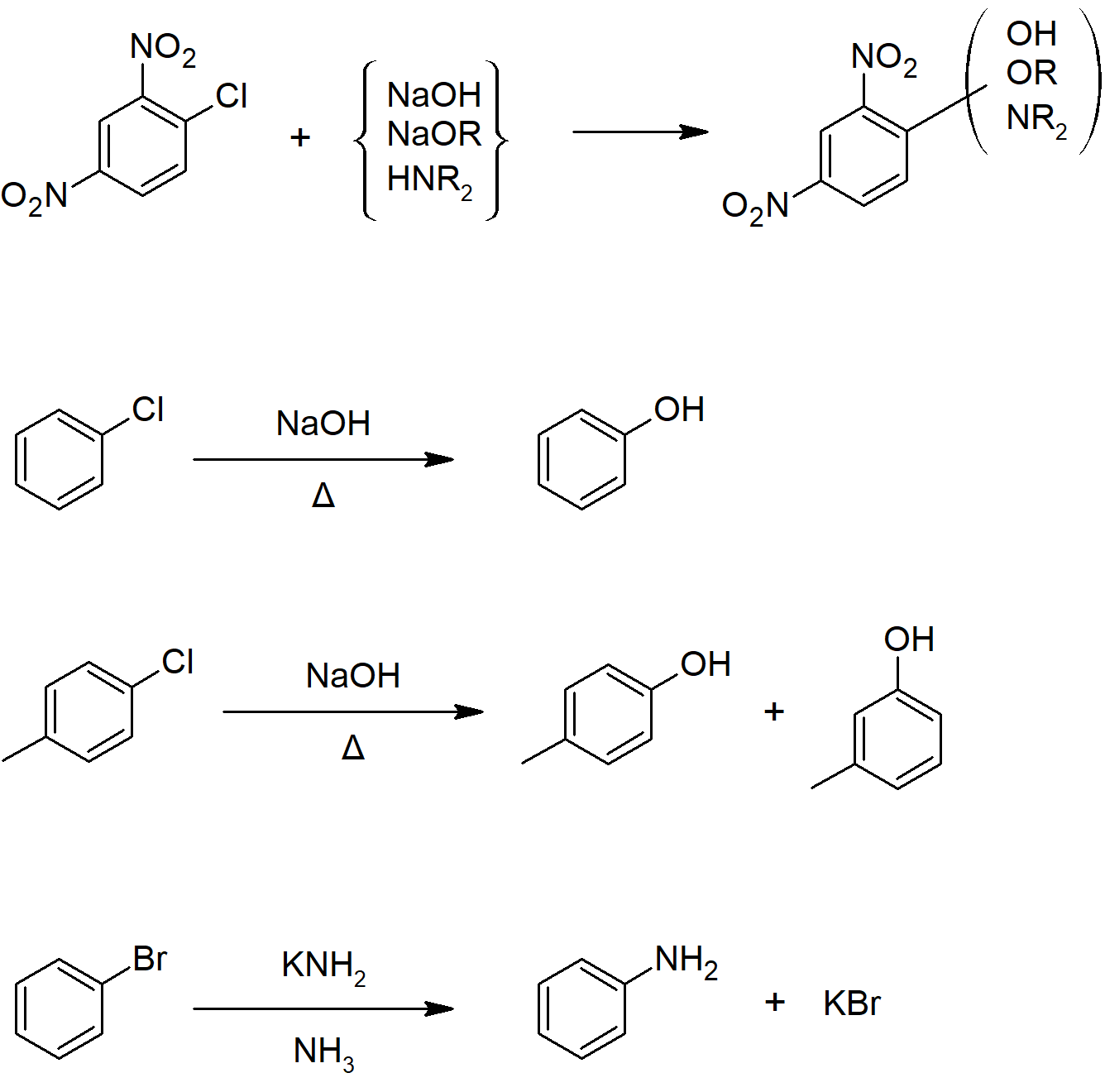
16.6 Nucleophilic Aromatic Substitution
- Highly activated aromatic rings can react with strong nucleophiles through a substitution mechanism.
- The mechanism typically begins with addition of a nucleophile followed by elimination of a leaving group.
- Under highly reactive conditions, a mechanism that begins with elimination for form a benzyne molecule intermediate followed by addition of a nucleophile resulting in nucleophilic aromatic substitution.
16.8 Oxidation of Aromatic Compounds
- Alkyl side-chains can be oxidized to benzoic acid (or a benzoic acid derivative if there are other groups present on the ring) by potassium permanganate (KMnO4) as long as the benzylic carbon has at least one hydrogen attached.
- Radical halogenation will occur at the benzylic carbon, due to stabilization of radical intermediates.
16.9 Reduction of Aromatic Compounds
- Catalytic hydrogenation (H2 and a catalyst) can be used to reduce many aromatic side-chains.
- Nitro groups can be selectively reduced to amines with SnCl2 and HCl or with Fe and HCl.
- Carbonyls adjacent to the ring can be reduced by either Clemmensen reduction (Zn(Hg) and HCl.
- Birch reductions can reduce aromatic rings.
16.10 Synthesis of Polysubstituted Benzenes
- Multistep synthesis requires a combination of forward (from the starting material) and backward (from the target compound) thinking.
Skills to Master
- Skill 16.1 Write detailed electrophilic aromatic substitution mechanisms (halogentation, nitration, sulfonation, Friedel-Crafts alkyation and acylation).
- Skill 16.2 Write detailed mechanisms for formation of reactive electrophiles.
- Skill 16.3 Predict and explain rearrangements that can occur during Friedel-Crafts alkylation.
- Skill 16.4 Explain activation and deactivation of aromatic rings toward electrophilic aromatic substitution.
- Skill 16.5 Explain ortho, para vs. meta directing during electrophilic aromatic substitution reactions.
- Skill 16.6 Combine activation and deactivation and directing effects to predict products of reactions of substituted aromatic molecules.
- Skill 16.7 Write detailed nucleophilic aromatic substitution mechanisms through addition-elimination.
- Skill 16.8 Write detailed nucleophilic aromatic substitution mechanisms through benzyne elimination-addition.
- Skill 16.9 Draw products of oxidation of aromatic molecules.
- Skill 16.10 Draw products of reduction of aromatic side-chains.
- Skill 16.11 Draw mechanisms for reduction of aromatic rings.
- Skill 16.12 Solve multistep synthesis problems incorporating directing effects and side-chain reactions.
Summary of Reactions
Reduction of Nitro Group
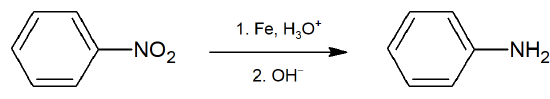
Oxidation of Alkylbenzene
Benzylic Bromination of Alkylbenzene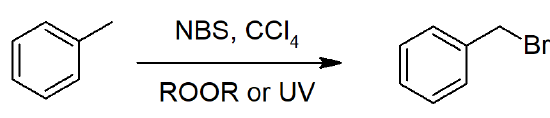
Reduction of Aromatic Compounds
Electrophilic Aromatic Substitution
Nucleophilic Aromatic Substitution