4.5: Oxidation Reactions
- Page ID
- 169794
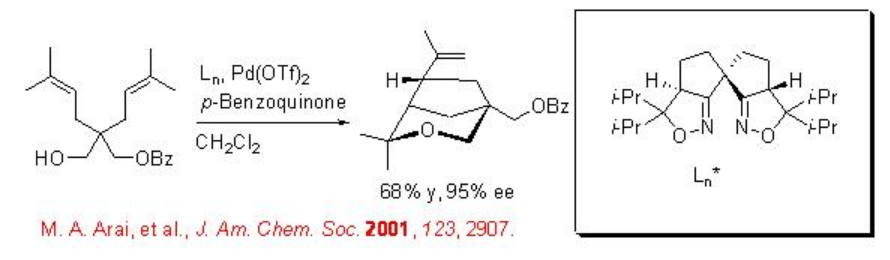
Wacker-type tandem cyclization reaction of alkenyl alcohol is reported using chiral palladium(II)-spirobis(isoxazoline) with excellent enantioselectivity (Scheme \(\PageIndex{1}\)). In this reaction, benzoquinone reoxidizes the reduced palladium(0) to palladium(II) species to complete the catalytic cycle.
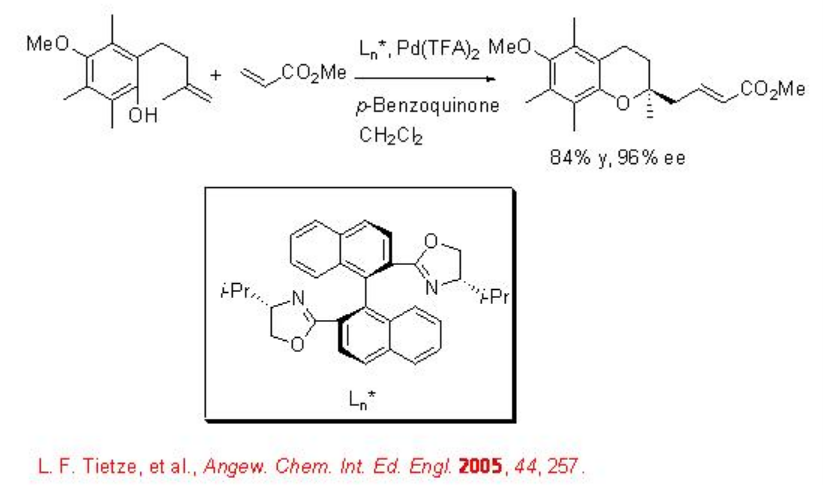
Palladium complex derived from Pd(TFA)2 and (S,S)-BOXAX has been found to be effective for the synthesis of chiral chroman framework in the presence of benzoquinone (Scheme \(\PageIndex{2}\)).
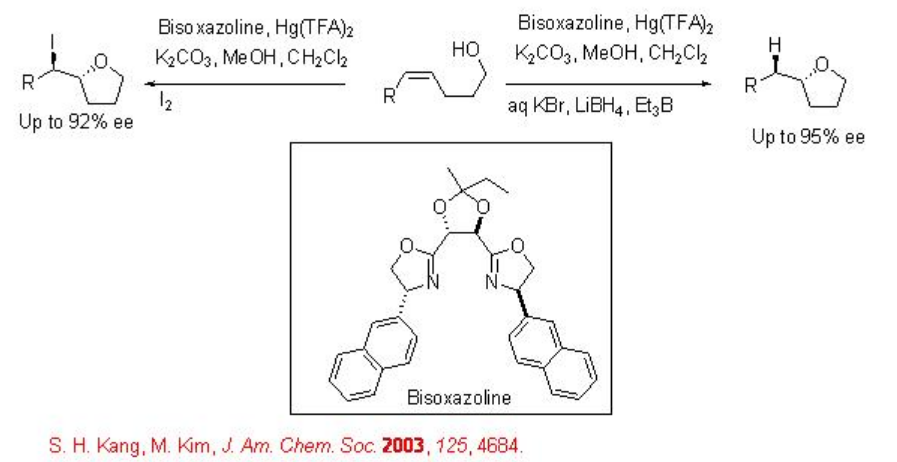
The mercury(II) complex derived from Hg(TFA)2 and bisoxazoline has been used for the mercuriocyclization with high enantioselectivity (Scheme \(\PageIndex{3}\)).
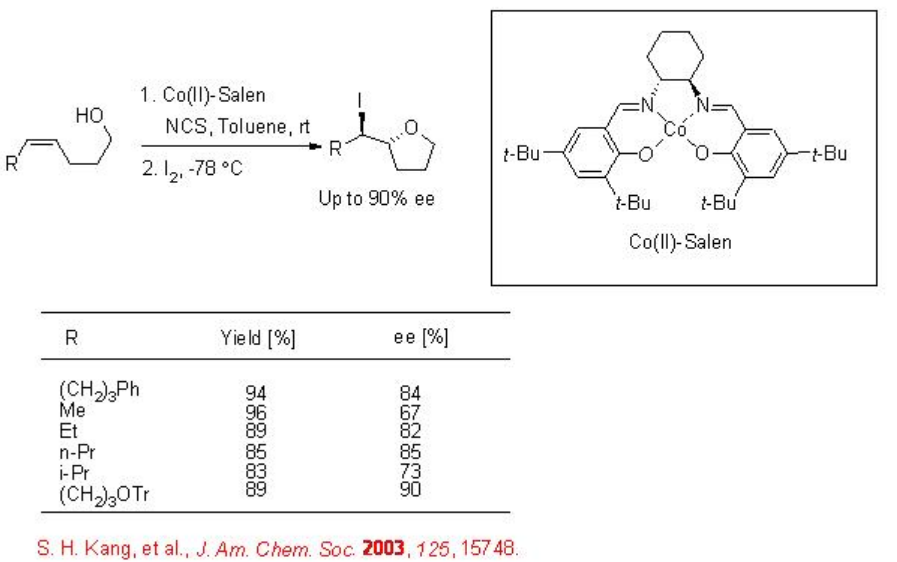
Chiral cobalt(II)-salen has been used for the enantioselective intramolecular iodoetherification to procure 2-substituted tetrahydrofurans with up to 90% ee (Scheme \(\PageIndex{4}\)).