1.5: Chiral Phosphoric Acids (PAs)
- Page ID
- 169465
Chiral phosphoric acids (PAs) derived from optically active BINOL carrying 3,3'-substituents have been utilized as effective chiral catalysts to various organic transformations. Phosphoric acids act as bifunctional catalysts bearing both Brønsted acidic site and a Lewis basic site and the 3,3'-substituents play a crucial role in attaining high stereoinduction by controlling the structural and electronic properties.
Asymmetric Counterion Directed Catalysis (ACDC)
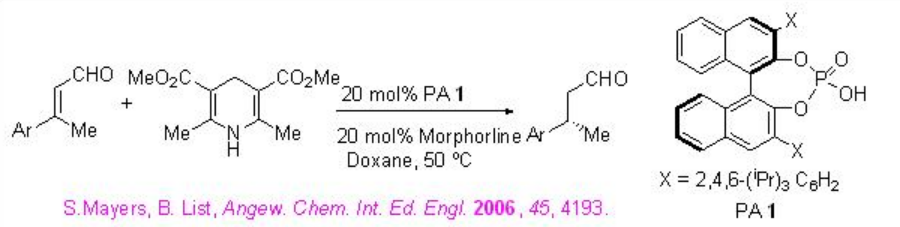
ACDC is a new concept of enantioselective synthesis. In 2006, List group first reported ACDC concept for the 1,4-hydrogenation of α,β -unsaturated aldehydes using the combination of morpholine and PA 1 at moderate temperature (Scheme \(\PageIndex{1}\)). In this reaction, PA 1 reacts with morpholine to give the morpholine salt of chiral anion PA 1 that catalyzes the reaction. The reaction takes place via the formation of iminium salt, wherein phosphate anion is believed to effectively shield one of the enantioface of the iminium salt.
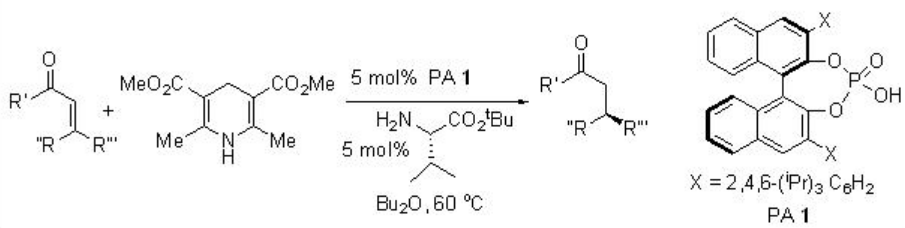
This method has been subsequently utilized for the reduction of α,β -unsaturated ketones (Scheme \(\PageIndex{2}\)). In which both cation and anion are chiral that catalyze with high enantioselectivity.
Examples:
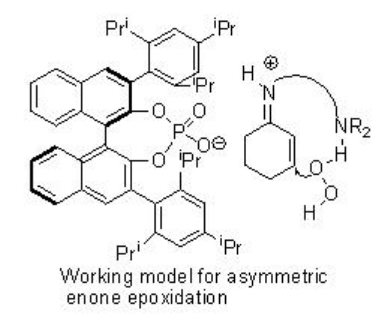
The PA 2 has been further utilized for the asymmetric epoxidation of α,β -unsaturated aldehydes in the presence of t-BuOOH (Scheme \(\PageIndex{3}\)). The proposed catalytic cycle is shown in Scheme 12. The initial addition product is achiral and the subsequent cyclization to iminium ion leads to the stereogenic center.
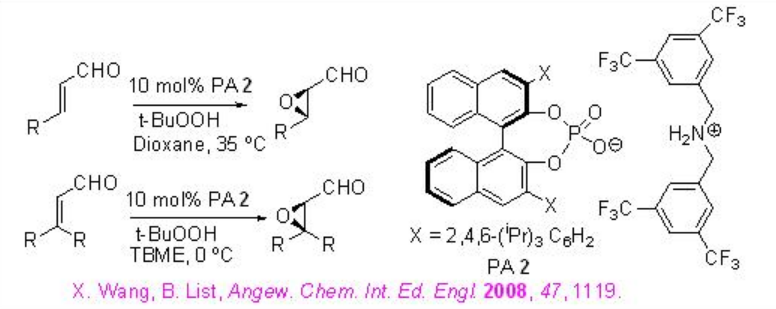
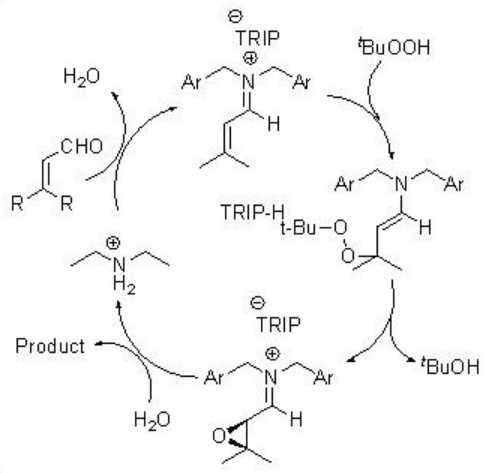
This methodology has been further extended for the epoxidation of α,β -unsaturated ketones (Scheme \(\PageIndex{5}\)). In this system, the diamine salt may serve as a bifunctional catalyst to possibly activate the enone substrate via iminium ion formation and hydrogen peroxide via general base catalysis as shown in Scheme \(\PageIndex{6}\).
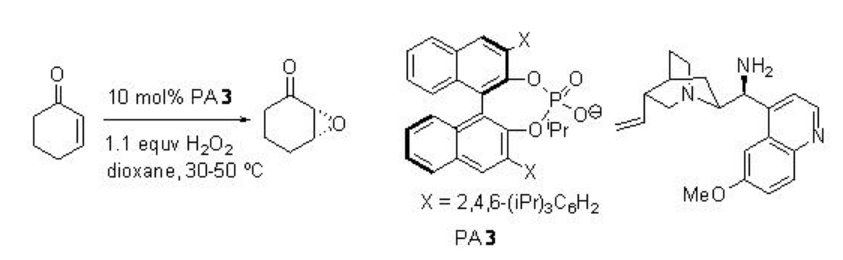
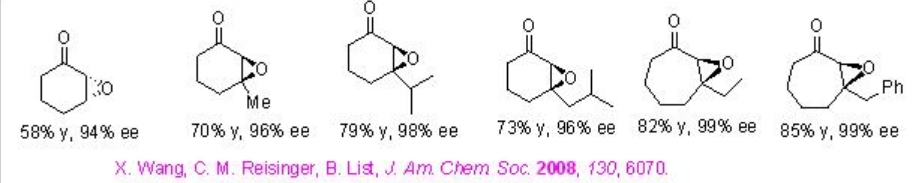
Examples:
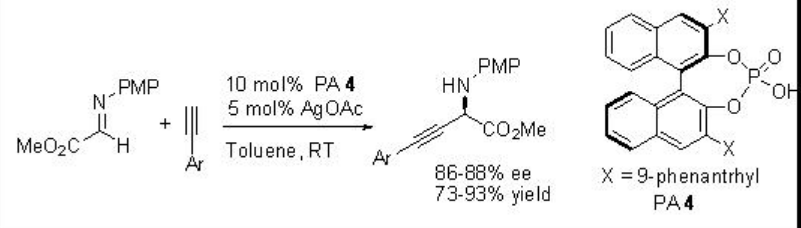
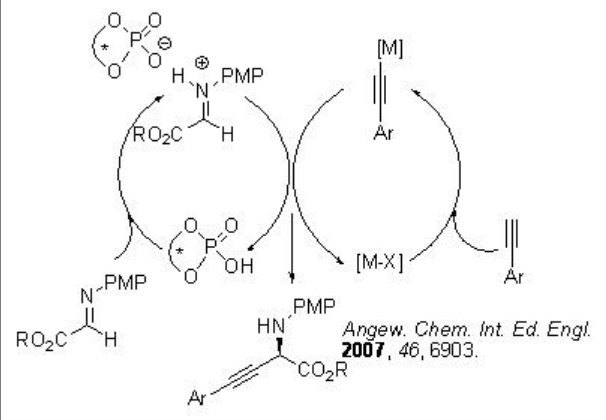
A dual catalytic procedure has been developed for the enantioselective activation of imines by a Brønsted acid combined with BINOL phosphate complex that results in a new metal catalyzed reaction in which the chiral counterion induces the enantioselectivity (Scheme 15).
Nucleophilic Additions of Aldimines
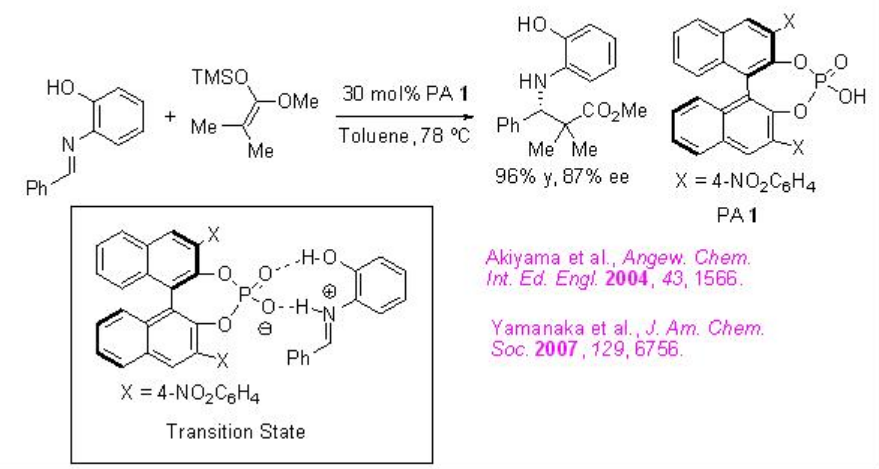
Chiral phosphoric acids (PAs) have been investigated as effective catalysts for Mannich type reactions. For examples, the reaction of imines with ketene silyl acetals has been studied using PA 1 in which introduction of 4-nitrophenyl substituents at 3,3'-positions has a beneficial effect on obtaining the high enantioselectivity (Scheme \(\PageIndex{8}\)). Based on DFT calculations a nine-membered zwitterionic transition state has been proposed to explain the stereoinduction.
The reaction of acetylacetone with N -boc-protected imines has been subsequently reported employing 2 mol% PA 2 with excellent yield and enantioselectivites (Scheme \(\PageIndex{9}\)). The procedure is compatible with a series of substrates to afford target products in high enantioselectivities.
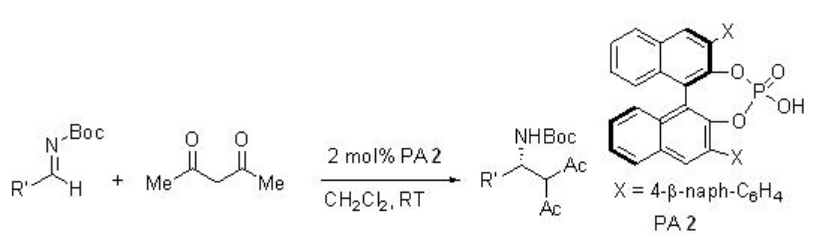
Examples:
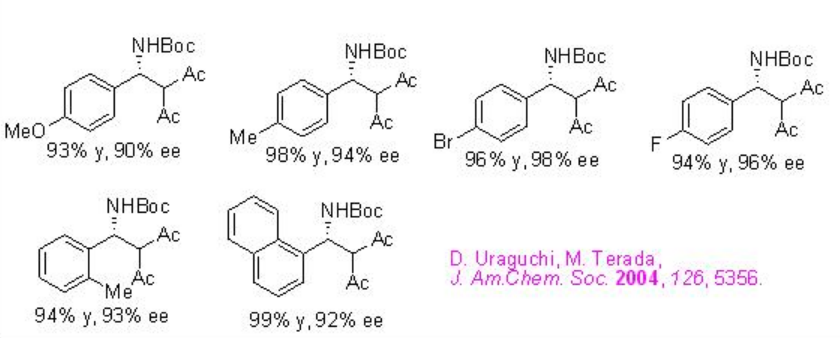
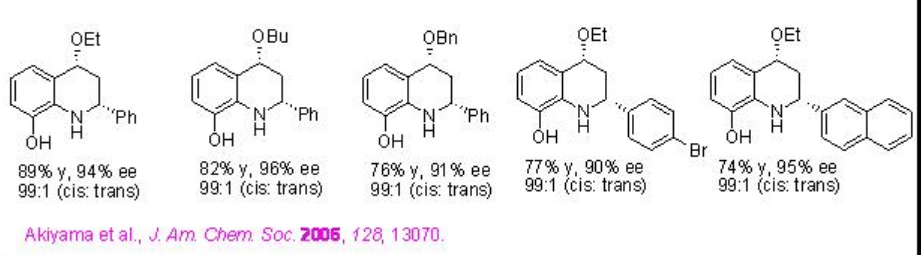
Phosphoric acid PA 3 derived from H8-BINOL derivative has been further studied for the direct Mannich reactions between in situ generated N -aryl imines and ketones (Scheme \(\PageIndex{10}\)). The authors have proposed TS-1 for the acid-promoted enolization of the ketone and its addition to the protonated aldimine.
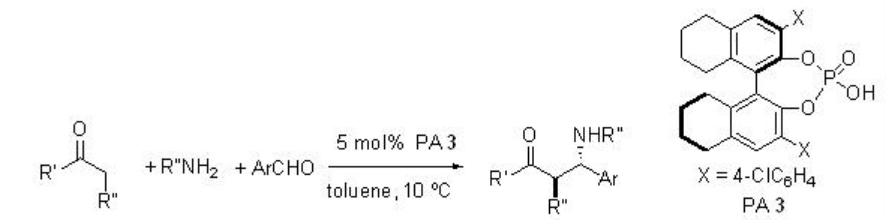
Examples:
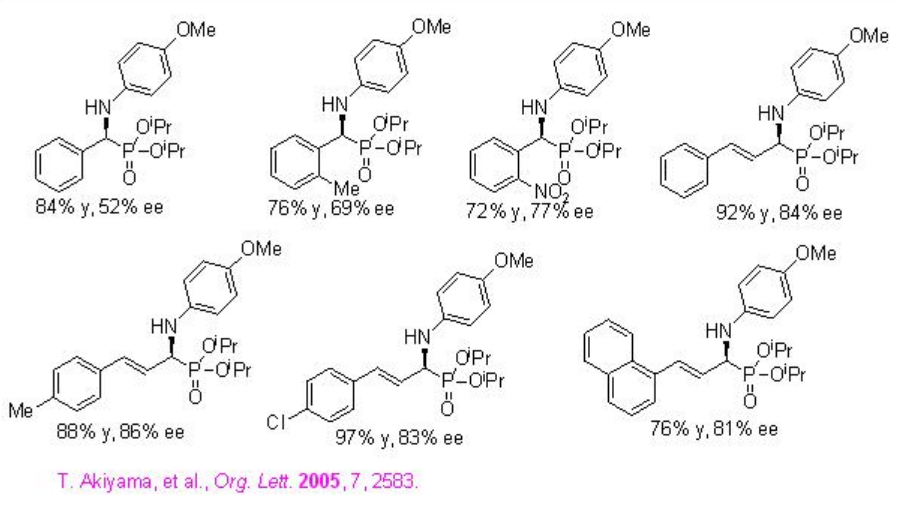
Hydrophosphorylation of aldimines with dialkyl phosphate has been studied using PA 4 to afford optically active α -amino phosphonates in good to high yields and enantioselectivities (Scheme \(\PageIndex{11}\)). The proposed transition state is shown in TS-2, where PA 4 acts as a bifunctional catalyst: the OH in phosphoric acid activates the aldimine as Brønsted acid and the phosphoryl oxygen activates the nucleophile as a Lewis base, thereby orienting both nucleophile and electrophile.
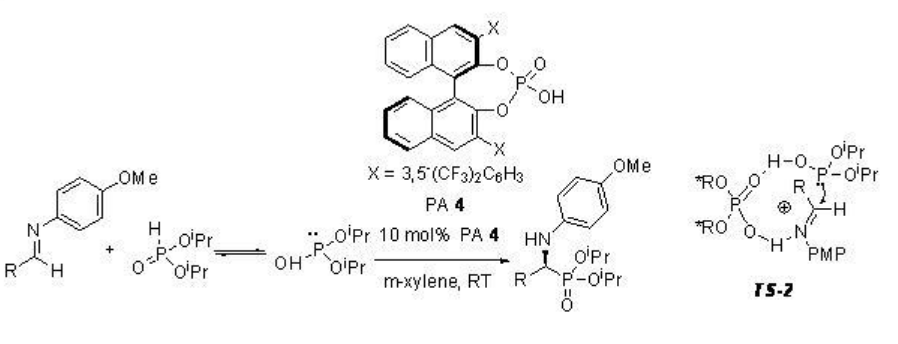
Examples:
Aza-Friedel-Crafts Reactions
The first organocatalytic aza-Friedel-Crafts reaction of aldimines has been accomplished using PA 5 (Scheme \(\PageIndex{12}\)). It is important to note that N-boc-protected aryl imines having electron-donating or –withdrawing groups at either the ortho -, meta -, or para - positions are compatible with the reaction condition.
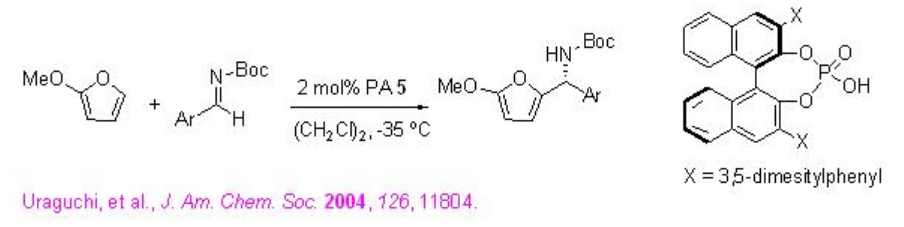
Examples:
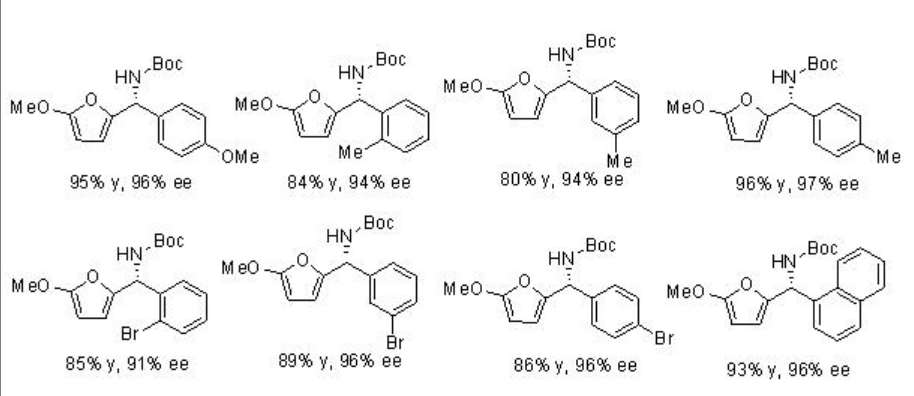
The reaction of indoles with enecarbamates has been successfully accomplished in the presence
Examples:
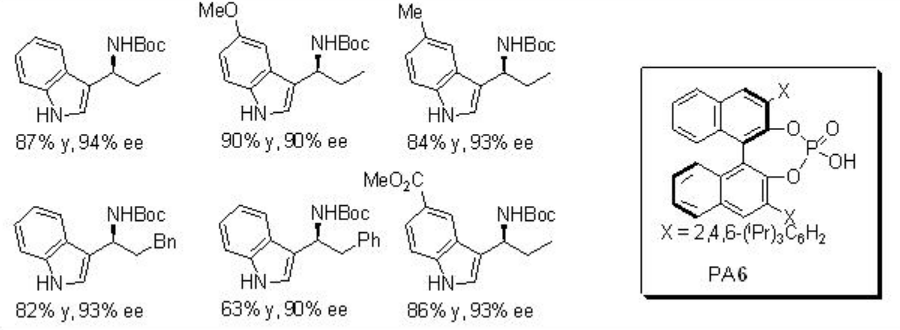
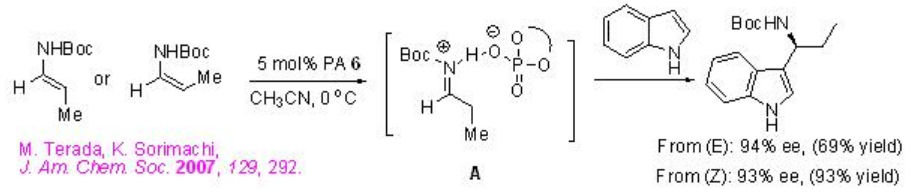
of PA 6 (Scheme \(\PageIndex{13}\)). Use of either pure regioisomers (E) or (Z)-enecarbamate gives the same product with similar enantioselectivities. Thus, the reaction is believed to takes place via a common intermediate A that could be generated by the protonation of the enecarbamates.
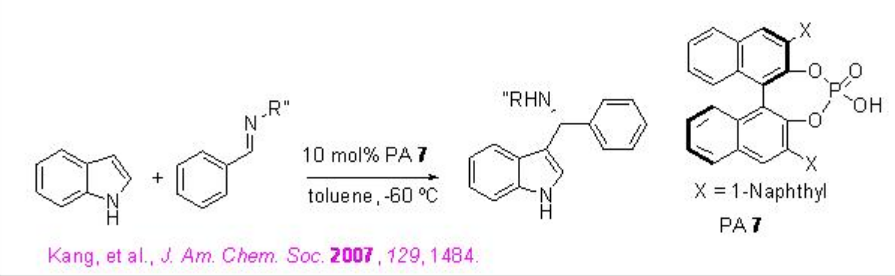
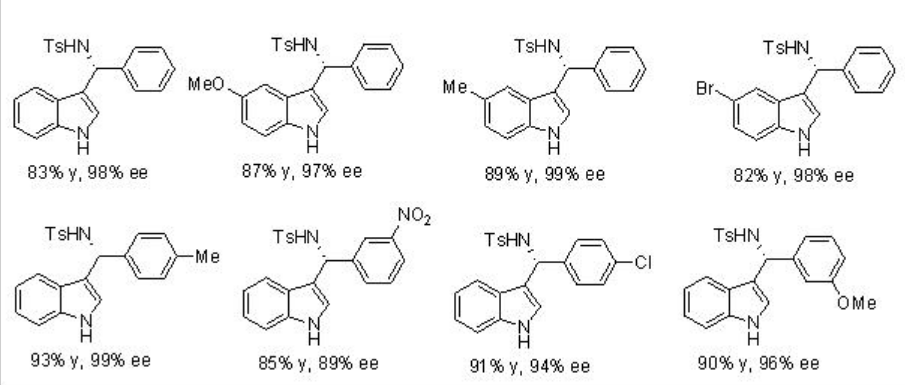
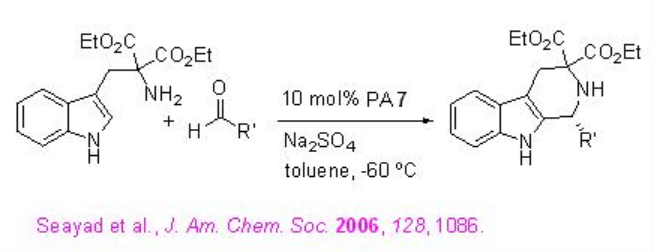
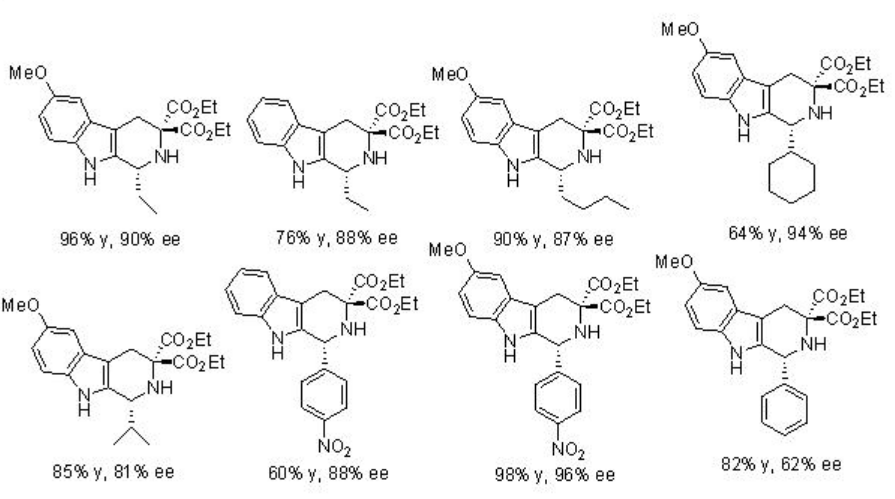
The reactions of indole with a wide range of imines, derived from aromatic aldehydes, have been demonstrated using PA 7 with excellent enantioselectivities (Scheme \(\PageIndex{14}\)).
Examples:
The Pictet-Spengler reaction of N-tritylsulfenyl tryptamines with various alphatic and aromatic aldehydes has been accomplished using PA 7 (Scheme \(\PageIndex{15}\)). The sulfenyl substituent stabilizes the intermediate iminium ion and favours the Pictet-Spengler cyclization compared to the undesired enamine formation.
Examples:
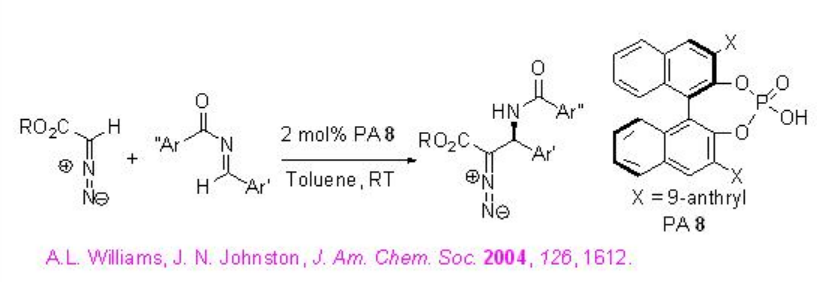
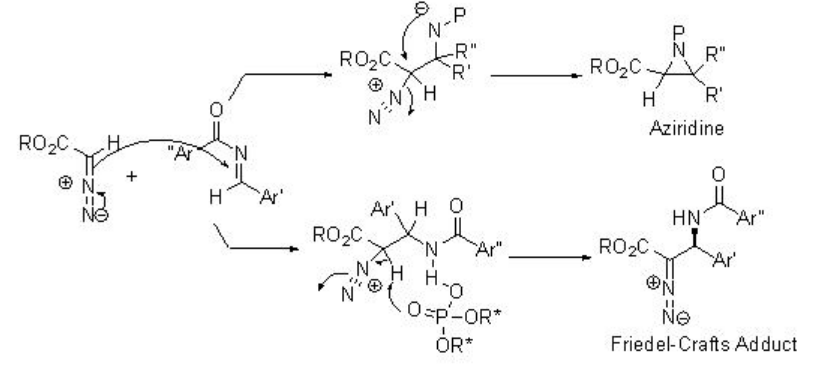
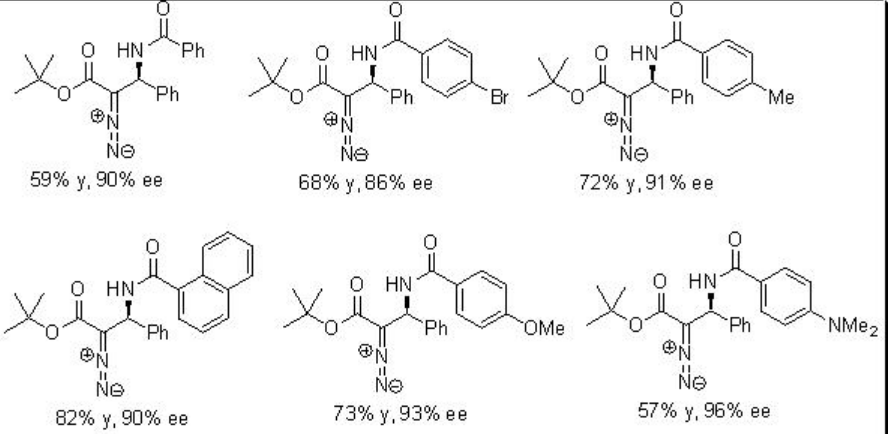
The quite interesting alkylation of α -diazoesters with N -acyl imines has been shown using PA 8 with high enantioselectivities (Scheme \(\PageIndex{16}\)). Diazoacetate is generally used in aziridine formation in the presence of Lewis acidic and Brønsted acidic conditions. Under these conditions, the competing aziridine formation has been eliminated by decreasing nucleophilicity of resulting amine intermediates and thus, the Friedel-Crafts adduct could be formed via C-H bond cleavage by the phosphoryl oxygen of phosphoric acid.
Examples:
1.4.4: Diels-Alder Reaction
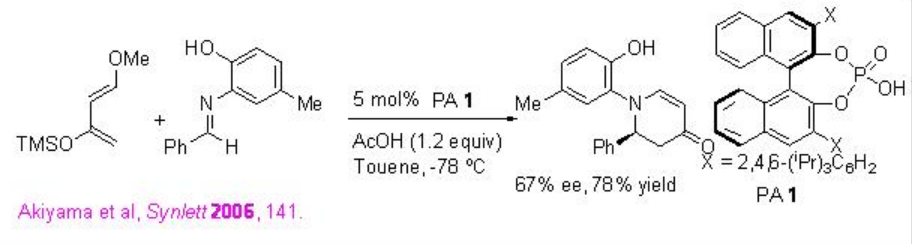
Chiral phosphoric acids (PAs) are excellent catalysts for the Diels-Alder reaction. For examples, the aza- Diels Alder reaction of Danishefsky's diene with aldimines is effective using PA 1 with good enantioselectivities (Scheme \(\PageIndex{17}\)). The addition of acetic acid leads to increase significantly the yield and enantioselectivities.
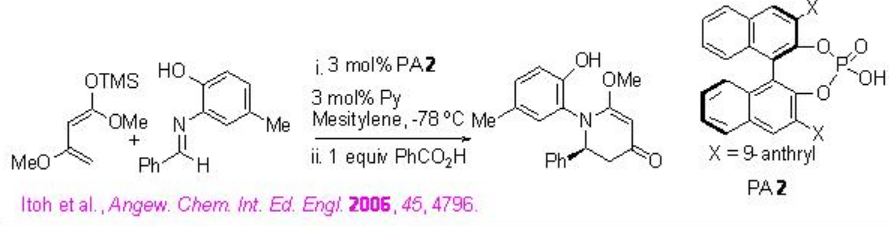
Although the aza- Diels Alder reaction of Brassard's diene using a Brønsted acid is rare due to the labilitiy of the diene in the presence of a strong Brønsted acid, PA 2 has been found to be an effective catalyst for the aza- Diels Alder reaction of Brassard's diene (Scheme \(\PageIndex{18}\)). The yield of the product could be improved using the pyridinium salt of the phosphoric acid as catalyst.
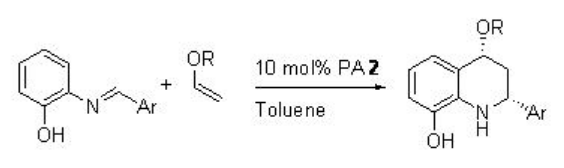
The PA 2 has also been found to effective for the inverse electron-demand aza -Diels Alder reaction of electron-rich alkenes with 2-aza dienes with excellent enantioselectivities (Scheme \(\PageIndex{19}\)). The presence of OH group is crucial for the cis selectivity in the products.
Examples:
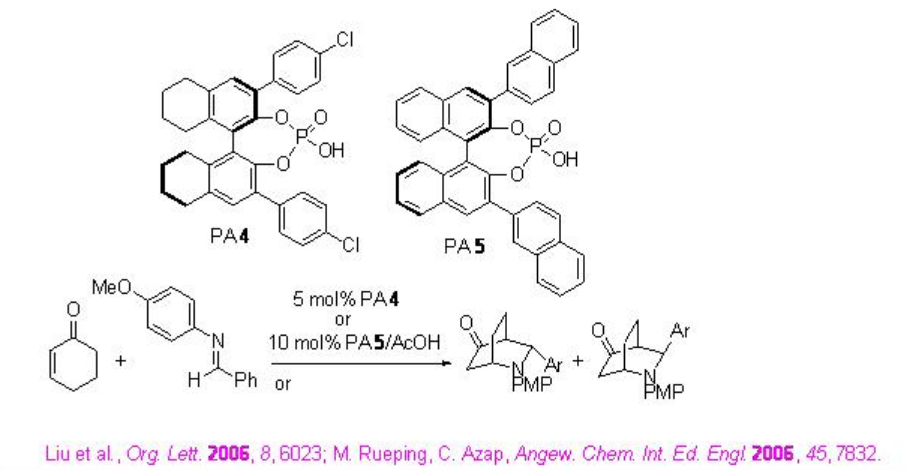
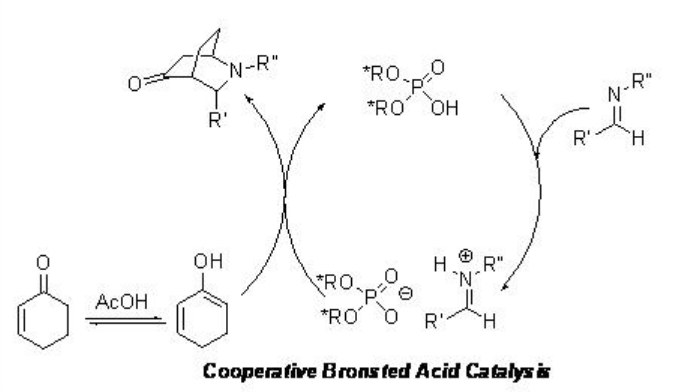
The aza- Diels Alder reaction of aldimines with cyclohexenone has been accomplished using either PA 4 or PA 5 /AcOH (Scheme \(\PageIndex{20}\)). A cooperative catalytic is proposed for the reaction using PA 5 /AcOH, where both the activation of an electrophile and a nucleophile takes place cooperatively (Scheme \(\PageIndex{21}\)).
Transfer Hydrogenation
Chiral phosphoric acids (PAs) are effective catalysts for the biomimetic hydrogenation using Hantzsch ester as a hydride source. For examples, the reduction of ketimines using Hantzsch ester can be accomplished using PA 6 with good yield and enantioselectivities (Scheme \(\PageIndex{22}\)). PA 1 bearing bulky 2,4,6-(i-Pr)3 C6 H3 at the 3,3'-positions of BINOL is found to superior to PA 6 for this purpose.

Examples:
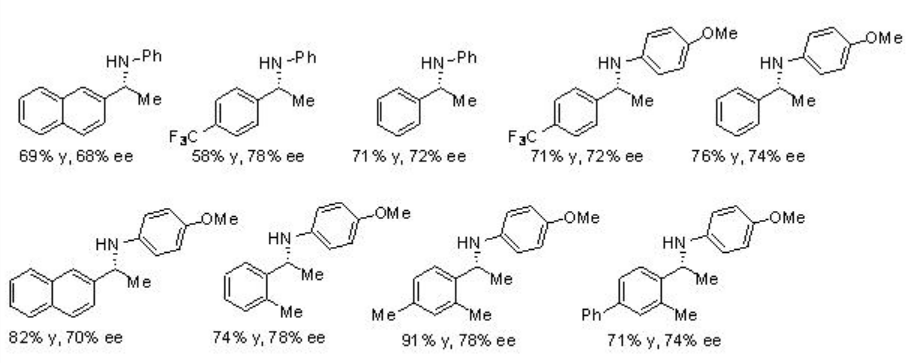
A three-component reductive amination reactions starting from ketones, amines and Hantzsch ester can be accomplished using PA 7 with excellent yield and enantioselectivities (Scheme \(\PageIndex{23}\)). This method is also compatible for the reactions of methyl phenyl ketones as well as methyl alkyl ketones.
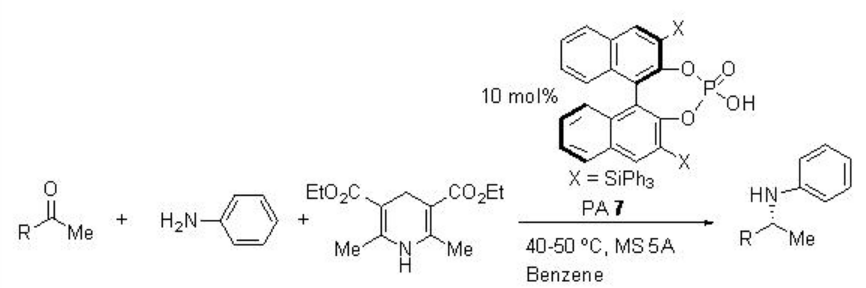
Examples:
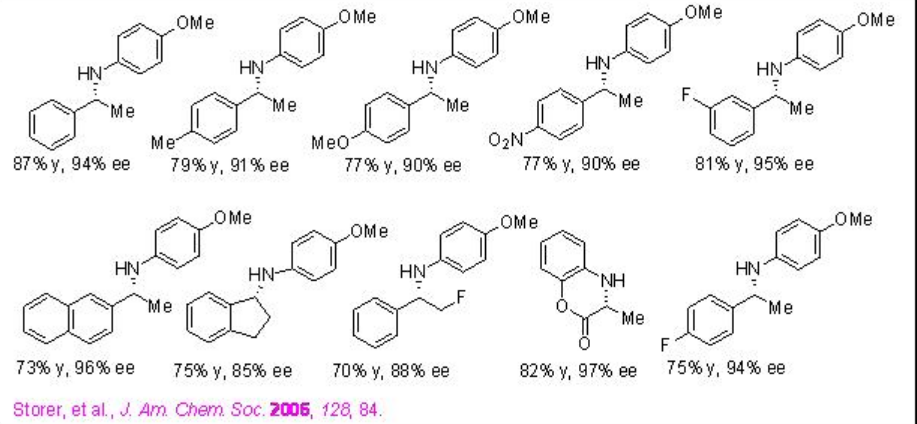
Following these initial studies, the reduction of wide of range of heterocycles has been explored. For examples, the reduction of a series of substituted quinonlines, benzoxazines, benzothiazines and benzoxazinones can be accomplished using PA 8 with excellent enantioselectivities (Scheme \(\PageIndex{24}\)).
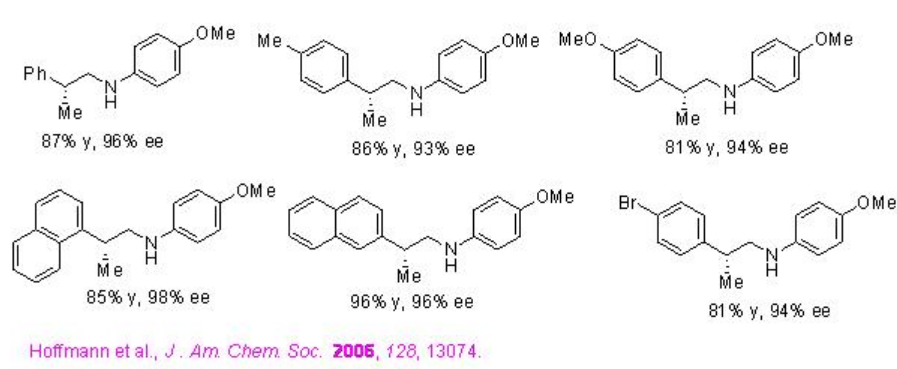
Asymmetric reductive amination of α -branched aldehydes and p -anisidine with Hantzch ester can be performed employing PA 1 with high enantioselectivities (Scheme \(\PageIndex{25}\)). The observed results suggest that the reaction proceeds via a dynamic kinetic resolution (Scheme \(\PageIndex{26}\)).
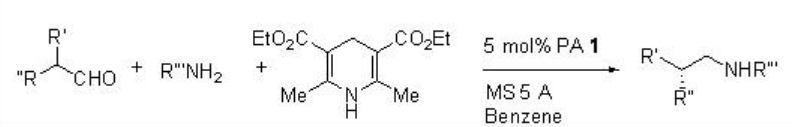
Examples:
Proposed Mechanism
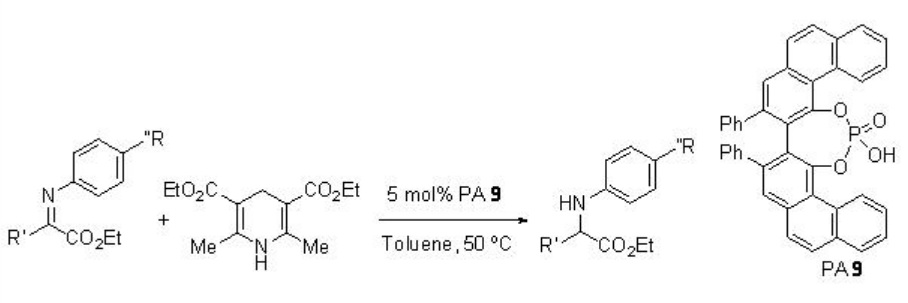
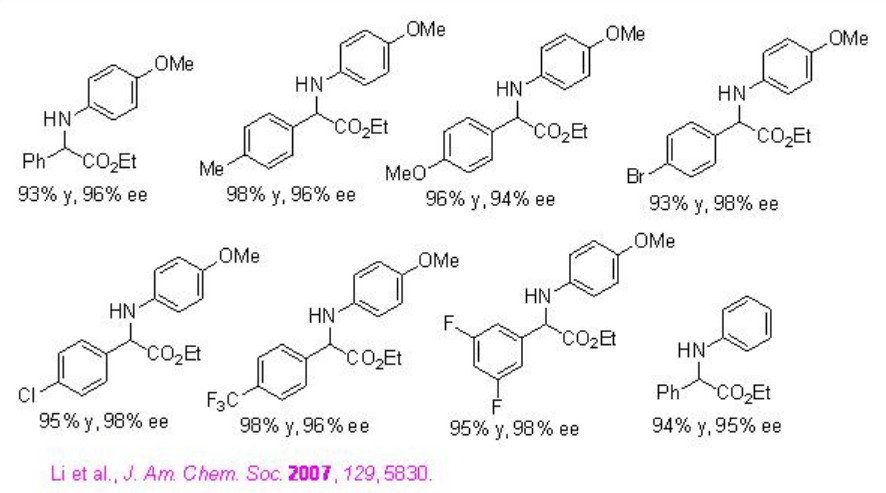
Chiral phosphoric acid PA 9 derived from ( S )-VAPOL is found to superior to PAs derived from BINOL for the reduction of α -imino esters using Hantzsh ester to afford α -amino esters with higher enantioselectivities (Scheme \(\PageIndex{27}\)).
Examples:
Mannich-type Reaction
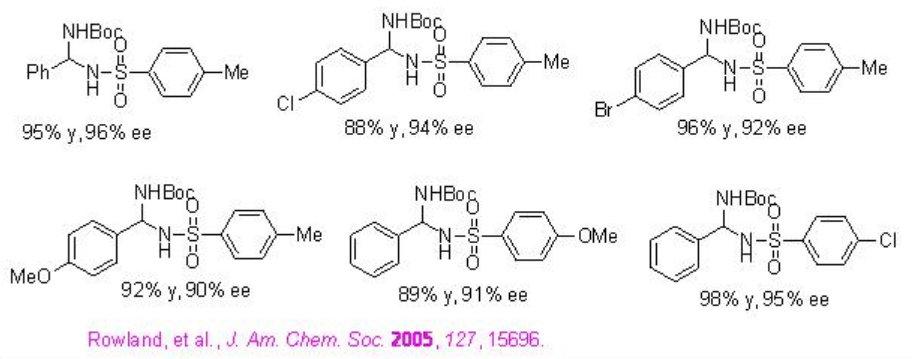
The utility of PA 9 has been further extended as excellent catalyst for the addition of nitrogen nucleophiles such as sulfonamides and imides to imines to give protected aminals (Scheme \(\PageIndex{28}\)). The procedure has wide substrate scope to give the target products in 73-99% ee and 80-99% yield.
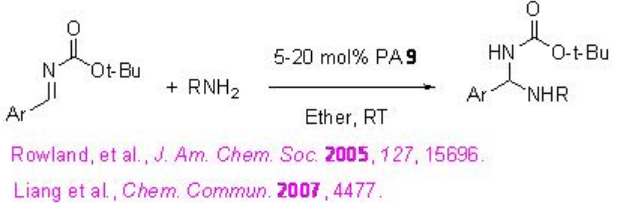
Examples:
Asymmetric Desymmetrization of meso-Aziridines
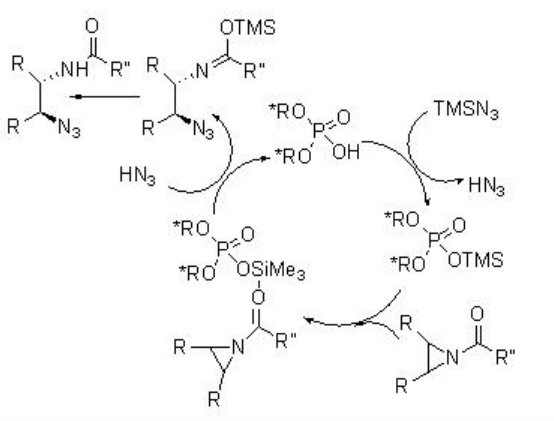
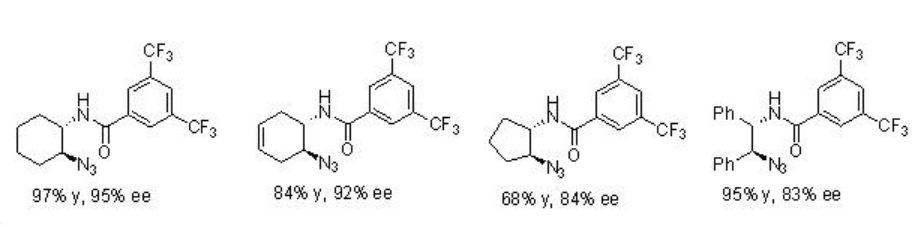
The application of PA 9 has been further extended to ring opening of meso -pyridines. This is the first example of organocatalyic desymmetrization of meso -aziridines. The substrates having electron-withdrawing protecting groups on the nitrogen proceed reaction with enhanced yields and enantioselectivity of the products (Scheme \(\PageIndex{29}\)).
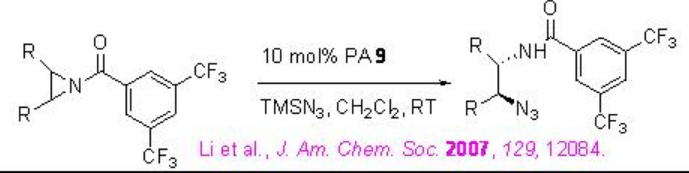
Examples:
Proposed Mechanism
The phosphoric acid first reacts with TMSN 3 to give silylated phosphoric acid as the active catalyst (Scheme \(\PageIndex{30}\)). The latter activates the aziridine by coordination of its carbonyl group, and subsequent attack of azide affords the precursor of the product and regeneration of the phosphoric acid.