7.2: Alcohols and Phenols- ROH, ArOH
- Page ID
- 22189
1. By the IUPAC system, the suffix -ol for \(OH\) is added to the name of the parent hydrocarbon. Notice that alkane \(+\) -ol becomes alkanol, with the e omitted:
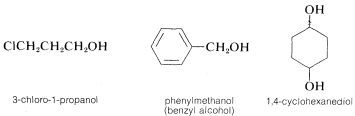
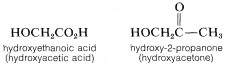
2. The substituent name for the \(OH\) group is hydroxy and should be used whenever the \(OH\) group is not the parent function (see Table 7-1). Notice how the precedence rules apply - hydroxy below carboxylic acid and hydroxy below ketone:
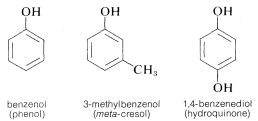
3. Many trivial names persist, particularly for aromatic, or arene alcohols (phenols):
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


