20.2: The War against Pests
- Page ID
- 152303
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- List the different types of pesticides and their uses.
Pesticides are substances that are meant to control pests. The term pesticide includes all of the following: herbicide, insecticides (which may include insect growth regulators, termiticides, etc.) nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, antimicrobial, and fungicide. The most common of these are herbicides which account for approximately 80% of all pesticide use. Most pesticides are intended to serve as plant protection products (also known as crop protection products), which in general, protect plants from weeds, fungi, or insects. As an example, the fungus Alternaria solani is used to combat the aquatic weed Salvinia.
In general, a pesticide is a chemical (such as carbamate) or biological agent (such as a virus, bacterium, or fungus) that deters, incapacitates, kills, or otherwise discourages pests. Target pests can include insects, plant pathogens, weeds, molluscs, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, or spread disease, or are disease vectors. Along with these benefits, pesticides also have drawbacks, such as potential toxicity to humans and other species.
Pesticides can be classified by target organism (e.g., herbicides, insecticides, fungicides, rodenticides, and pediculicides – see table), chemical structure (e.g., organic, inorganic, synthetic, or biological (biopesticide), although the distinction can sometimes blur), and physical state (e.g. gaseous (fumigant)). Biopesticides include microbial pesticides and biochemical pesticides. Plant-derived pesticides, or "botanicals", have been developing quickly. These include the pyrethroids, rotenoids, nicotinoids, and a fourth group that includes strychnine and scilliroside.:15
Since before 2000 BC, humans have utilized pesticides to protect their crops. The first known pesticide was elemental sulfur dusting used in ancient Sumer about 4,500 years ago in ancient Mesopotamia. The Rig Veda, which is about 4,000 years old, mentions the use of poisonous plants for pest control.[13] By the 15th century, toxic chemicals such as arsenic, mercury, and lead were being applied to crops to kill pests. In the 17th century, nicotine sulfate was extracted from tobacco leaves for use as an insecticide. The 19th century saw the introduction of two more natural pesticides, pyrethrum, which is derived from chrysanthemums, and rotenone, which is derived from the roots of tropical vegetables.[14] Until the 1950s, arsenic-based pesticides were dominant.[15] Paul Müller discovered that DDT was a very effective insecticide. Organochlorines such as DDT were dominant, but they were replaced in the U.S. by organophosphates and carbamates by 1975. Since then, pyrethrin compounds have become the dominant insecticide.[15] Herbicides became common in the 1960s, led by "triazine and other nitrogen-based compounds, carboxylic acids such as 2,4-dichlorophenoxyacetic acid, and glyphosate".
The discussion in this section will be focused mainly on insecticides and the next section on herbicides and defoliants. Many pesticides can be grouped into chemical families. Prominent insecticide families include organochlorines, organophosphates, and carbamates.
Table \(\PageIndex{1}\) below gives the LD50 values for some insecticides. In each case, the chemical was fed to laboratory rats. Note that the lower the LD50, the more toxic the chemical.
| Chemical | Category | Oral LD50 in Rats (mg/kg) |
|---|---|---|
| Aldicarb ("Temik") | Carbamate | 1 |
| Carbaryl ("Sevin") | Carbamate | 307 |
| DDT | Chlorinated hydrocarbon | 87 |
| Dieldrin | Chlorinated hydrocarbon | 40 |
| Diflubenzuron ("Dimilin") | Chitin inhibitor | 10,000 |
| Malathion | Organophosphate | 885 |
| Methoprene | JH mimic | 34,600 |
| Methoxychlor | Chlorinated hydrocarbon | 5,000 |
| Parathion | Organophosphate | 3 |
| Piperonyl butoxide | Synergist | 7,500 |
| Pyrethrins | Plant extract | 200 |
| Rotenone | Plant extract | 60 |
Note: LD50 (abbreviation for "lethal dose, 50%") of a toxin, radiation, or pathogen is the dose required to kill half the members of a tested population after a specified test duration. LD50 figures are frequently used as a general indicator of a substance's acute toxicity. A lower LD50 is indicative of increased toxicity.
Benefits of Using Pesticides
Pesticides can save farmers' money by preventing crop losses to insects and other pests; in the U.S., farmers get an estimated fourfold return on money they spend on pesticides. One study found that not using pesticides reduced crop yields by about 10%. Another study, conducted in 1999, found that a ban on pesticides in the United States may result in a rise of food prices, loss of jobs, and an increase in world hunger.
There are two levels of benefits for pesticide use, primary and secondary. Primary benefits are direct gains from the use of pesticides and secondary benefits are effects that are more long-term.
Primary benefits
Controlling pests and plant disease vectors
- Improved crop yields
- Improved crop/livestock quality
- Invasive species controlled
Controlling human/livestock disease vectors and nuisance organisms
- Human lives saved and disease reduced. Diseases controlled include malaria, with millions of lives having been saved or enhanced with the use of DDT alone.
- Animal lives saved and disease reduced
Controlling organisms that harm other human activities and structures
- Drivers view unobstructed
- Tree/brush/leaf hazards prevented
- Wooden structures protected
Monetary
In one study, it was estimated that for every dollar ($1) that is spent on pesticides for crops can yield up to four dollars ($4) in crops saved. This means based that, on the amount of money spent per year on pesticides, $10 billion, there is an additional $40 billion savings in crop that would be lost due to damage by insects and weeds. In general, farmers benefit from having an increase in crop yield and from being able to grow a variety of crops throughout the year. Consumers of agricultural products also benefit from being able to afford the vast quantities of produce available year-round.
DDT: The Dream Insecticide - or Nightmare
DDT was the first of a long line of insecticides based on hydrocarbons with chlorine atoms replacing some of the hydrogen atoms. Its chemical name is dichloro, diphenyl, trichloroethane (see Figure \(\PageIndex{1a}\)).
a. 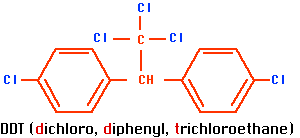 b.
b. 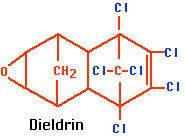
Figure \(\PageIndex{1}\) a. DDT and b. dieldrin
Some others:
- methoxychlor
- dieldrin (see Figure \(\PageIndex{1a}\)).
- dicofol (Kelthane®)
- endosulfan (Thiodan®, Phaser®)
DDT was introduced during World War II and, along with penicillin and the sulfa drugs, was responsible for the fact that this was the first war in history where trauma killed more people - combatants and noncombatants alike - than infectious disease.
DDT is effective against
- vectors of human diseases such as
- malaria and yellow fever (both transmitted by mosquitoes)
- plague (transmitted by fleas)
- many crop pests
Prior to the introduction of DDT, the number of cases of malaria in Ceylon (now Sri Lanka) was more than a million a year. By 1963 the disease had been practically eliminated from the island. However, growing concern about the hazards of DDT led to its abandonment there in the mid-1960s, and soon thereafter malaria became common once again.
DDT was especially effective against malarial mosquitoes because of its persistence and resistance to breakdown in the environment. One or two sprays a year on the walls of homes kept them free of mosquitoes. But DDT has several serious drawbacks.
Development of Insect Resistance
As early as 1946, Swedish workers discovered populations of houseflies resistant to DDT. This was quickly followed by many other reports of developing resistance. Other chlorinated hydrocarbons (like dieldrin and methoxychlor) were developed as substitutes, but in time insects developed resistance to these as well.
Biomagnification
Although no harmful effects from average exposures to DDT have been seen in humans, DDT and other chlorinated hydrocarbons have been shown to harm other species, such as fishes, earthworms, and robins. The hazard of DDT to nontarget animals is particularly acute for those species living at the top of food chains. Carnivores at the ends of long food chains (e.g., ospreys, pelicans, falcons, and eagles) once suffered serious declines in fecundity and hence in population size because of this. High levels of chlorinated hydrocarbons interfere with forming eggshells of normal thickness.
Organophosphates
The organophosphates, e.g., parathion (Figure \(\PageIndex{2}\) ), are related to the nerve gases developed during World War II. They react irreversibly with the enzyme acetylcholinesterase, which is responsible for inactivating acetylcholine (ACh) at neuromuscular junctions and at certain synapses in the central and peripheral nervous systems.
Some other examples:
- malathion
- diazinon
- phosmet (Imidan®)
- chlorpyrifos (Lorsban®)
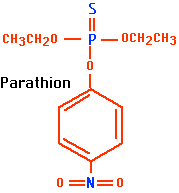
Some of the organophosphates are very toxic. Parathion, for example, is 30 times more toxic than DDT. Each year organophosphates poison thousands of humans throughout the world, causing hundreds of deaths. Medical personnel caring for poisoning victims are also at risk. They may be seriously poisoned by the excretions of, and even the vapors emanating from, their patients.
Unlike chlorinated hydrocarbons,
- Organophosphates break down quickly in the environment, and thus residues on crops are less likely to be a problem.
- They are not stored in animal tissue, so biomagnification has not been a problem either.
For these reasons, their use has greatly reduced the hazard to non target species like ospreys and eagles (at the price of a much greater hazard to humans). Development of resistance is just as much a problem as it is with the chlorinated hydrocarbons. The carbamates were introduced in an attempt to keep ahead.
Carbamates
Carbamate insecticides are also inhibitors of acetylcholinesterase, but their action is reversible.
Some examples:
- carbaryl (Sevin®)
- aldicarb (Temik®)
- methomyl (Lannate®)
Features:
- These compounds are rapidly detoxified and excreted so their risk to warm-blooded animals is less than the other agents we have looked at.
- They are degraded rapidly in the environment so persistence is not a problem.
- They are, however, a danger to many useful insects, especially honeybees.

Growth Regulators
The members of this diverse group interfere in one way or another with insect development. Although most insect growth regulators do not affect adults, for many pests, it is the larval stages that are the most destructive.
Chitin Inhibitors
These substances, diflubenzuron (Dimilin®) is an example, interfere with the synthesis of chitin, the material that makes up the insect exoskeleton. It seems to have very low toxicity for vertebrates, but is harmful to crustaceans as well as insects. Its effect on fungi, which also synthesize chitin, needs to be studied.
Molting Disruptors
Insects must periodically shed their exoskeleton — called molting — in order to grow. Each molt is triggered by a steroid hormone called ecdysone.
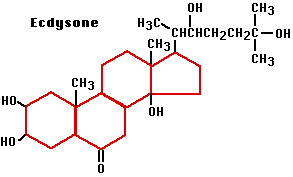
A few synthetic ecdysone
- agonists, e.g.,
- tebufenozide (Confirm®)
- methoxyfenozide (Intrepid®)
- inhibitors, e.g., azadirachtin (Neemix®)
are now used as insecticides.
Organic Pesticides
Generally speaking, pesticides derived from natural materials or living organisms are allowed in organic production as long as they do not contain synthetic additives or are not specifically prohibited on the National List under § 205.602. By contrast, most synthetic pesticides are not allowed; those few that are can be found on the National List under § 205.601.
Allowed inputs typically include but are not limited to the following:
Botanical pesticides. Botanicals are derived from plants. They include pyrethrum, rotenone, sabadilla, neem, ryania, and garlic. Strychnine and nicotine are also botanicals, but are expressly prohibited in organic production. Since botanical pesticides are relatively nonselective, they can affect natural predators and other nontarget organisms. Rotenone, for example, is highly toxic to fish. For this reason, many organic growers use botanical pesticides only as a last resort.
Spray oils. Vegetable- or animal-derived oils are generally allowed as suffocating (stylet) oils, summer oils, dormant oils, and surfactants. Some petroleum-derived oils, referred to as “narrowrange oils,” are allowed for the same purposes. Spray oils are commonly used to control scale and mite pests. There are two organizations that review products and publish lists of products allowed for organic production: the Organic Materials Review Institute (OMRI) and the Washington State Department of Agriculture (WSDA) Organic Food Program.
Insecticidal soaps. Fatty acid insecticidal soaps are synthetic pesticides specifically allowed in organic production. Safer® Brand Insect Killing Soap Concentrate II is a product that is commonly used by organic farmers. Insecticidal soaps can be hard on beneficial predatory mites, so they should be used with caution.
Minerals. Mineral-based pesticides include sulfur, copper products, diatomaceous earth, and kaolin clay. These must be used with caution, even though they are allowed. Sulfur can reduce the populations of some beneficial insects and may burn plants if used during hot weather. Diatomaceous earth can cause respiratory problems and itching in the farmworkers who apply it. Copper can accumulate in soils, so it is allowed with restrictions. The organic regulations state that “copper-based materials must be used in a manner that minimizes accumulation in the soil…” Certifiers may require soil testing to verify that copper is not accumulating in the soil. Certain highly toxic minerals, including arsenic and lead, are specifically prohibited.
Pheromones. Pheromones are chemicals released from insects that cause other insects of the same species to change their behavior. Pheromones are not considered pesticides because they do not kill the insects. The pheromones used for pest control are often called mating disrupters because they alter mating behavior. Being totally natural, the pheromones themselves are allowed in organic production. However, some of the inert ingredients in mating disrupter products are prohibited.
Biological Pesticides
Biopesticides, a contraction of 'biological pesticides', include several types of pest management intervention: through predatory, parasitic, or chemical relationships. The term has been associated historically with biological pest control – and by implication, the manipulation of living organisms. Regulatory positions can be influenced by public perceptions, thus:
- in the EU, biopesticides have been defined as "a form of pesticide based on micro-organisms or natural products".
- the US EPA states that they "include naturally occurring substances that control pests (biochemical pesticides), microorganisms that control pests (microbial pesticides), and pesticidal substances produced by plants containing added genetic material (plant-incorporated protectants) or PIPs".
They are obtained from organisms including plants, bacteria and other microbes, fungi, nematodes, etc.[page needed] They are often important components of integrated pest management (IPM) programmes, and have received much practical attention as substitutes to synthetic chemical plant protection products (PPPs).
Biopesticides can be classified into these classes:
- Microbial pesticides which consist of bacteria, entomopathogenic fungi or viruses (and sometimes includes the metabolites that bacteria or fungi produce). Entomopathogenic nematodes are also often classed as microbial pesticides, even though they are multi-cellular.[page needed]
- Bio-derived chemicals. Four groups are in commercial use: pyrethrum, rotenone, neem oil, and various essential oils are naturally occurring substances that control (or monitor in the case of pheromones) pests and microbial diseases.
- Plant-incorporated protectants (PIPs) have genetic material from other species incorporated into their genetic material (i.e. GM crops). Their use is controversial, especially in many European countries.
- RNAi pesticides, some of which are topical and some of which are absorbed by the crop.
Biopesticides have usually no known function in photosynthesis, growth or other basic aspects of plant physiology. Instead, they are active against biological pests. Many chemical compounds have been identified that are produced by plants to protect them from pests so they are called antifeedants. These materials are biodegradable and renewable alternatives, which can be economical for practical use. Organic farming systems embraces this approach to pest control.
Examples
Bacillus thuringiensis, a bacterium capable of causing disease of Lepidoptera, Coleoptera and Diptera, is a well-known insecticide example. The toxin from B. thuringiensis (Bt toxin) has been incorporated directly into plants through the use of genetic engineering. The use of Bt Toxin is particularly controversial. Its manufacturers claim it has little effect on other organisms, and is more environmentally friendly than synthetic pesticides.
Other microbial control agents include products based on:
- entomopathogenic fungi (e.g. Beauveria bassiana, Isaria fumosorosea, Lecanicillium and Metarhizium spp.),
- plant disease control agents: include Trichoderma spp. and Ampelomyces quisqualis (a hyper-parasite of grape powdery mildew); Bacillus subtilis is also used to control plant pathogens.
- beneficial nematodes attacking insect (e.g. Steinernema feltiae) or slug (e.g. Phasmarhabditis hermaphrodita) pests
- entomopathogenic viruses (e.g.. Cydia pomonella granulovirus).
- weeds and rodents have also been controlled with microbial agents.
Various naturally occurring materials, including fungal and plant extracts, have been described as biopesticides. Products in this category include:
- Insect pheromones and other semiochemicals
- Fermentation products such as Spinosad (a macro-cyclic lactone)
- Chitosan: a plant in the presence of this product will naturally induce systemic resistance (ISR) to allow the plant to defend itself against disease, pathogens and pests.
- Biopesticides may include natural plant-derived products, which include alkaloids, terpenoids, phenolics and other secondary chemicals. Certain vegetable oils such as canola oil are known to have pesticidal properties[citation needed]. Products based on extracts of plants such as garlic have now been registered in the EU and elsewhere
Genetically Engineered Insect and Virus Resistance
Genetically modified crops (GM crops) are plants used in agriculture, the DNA of which has been modified using genetic engineering methods. Plant genomes can be engineered by physical methods or by use of Agrobacterium for the delivery of sequences hosted in T-DNA binary vectors. In most cases, the aim is to introduce a new trait to the plant which does not occur naturally in the species. Examples in food crops include resistance to certain pests, diseases, environmental conditions, reduction of spoilage, resistance to chemical treatments (e.g. resistance to a herbicide), or improving the nutrient profile of the crop.
Insects
Tobacco, corn, rice and some other crops have been engineered to express genes encoding for insecticidal proteins from Bacillus thuringiensis (Bt). The introduction of Bt crops during the period between 1996 and 2005 has been estimated to have reduced the total volume of insecticide active ingredient use in the United States by over 100 thousand tons. This represents a 19.4% reduction in insecticide use.
In the late 1990s, a genetically modified potato that was resistant to the Colorado potato beetle was withdrawn because major buyers rejected it, fearing consumer opposition.
Viruses
Papaya, potatoes, and squash have been engineered to resist viral pathogens such as cucumber mosaic virus which, despite its name, infects a wide variety of plants. Virus resistant papaya were developed in response to a papaya ringspot virus (PRV) outbreak in Hawaii in the late 1990s. They incorporate PRV DNA. By 2010, 80% of Hawaiian papaya plants were genetically modified.
Potatoes were engineered for resistance to potato leaf roll virus and Potato virus Y in 1998. Poor sales led to their market withdrawal after three years.
Yellow squash that were resistant to at first two, then three viruses were developed, beginning in the 1990s. The viruses are watermelon, cucumber and zucchini/courgette yellow mosaic. Squash was the second GM crop to be approved by US regulators. The trait was later added to zucchini.
Many strains of corn have been developed in recent years to combat the spread of Maize dwarf mosaic virus, a costly virus that causes stunted growth which is carried in Johnson grass and spread by aphid insect vectors. These strands are commercially available although the resistance is not standard among GM corn variants.
Sterile Insect Technique
The sterile insect technique (SIT)[1][2] is a method of biological insect control, whereby overwhelming numbers of sterile insects are released into the wild. The released insects are preferably male, as this is more cost-effective and the females may in some situations cause damage by laying eggs in the crop, or, in the case of mosquitoes, taking blood from humans. The sterile males compete with wild males to mate with the females. Females that mate with a sterile male produce no offspring, thus reducing the next generation's population. Sterile insects are not self-replicating and, therefore, cannot become established in the environment. Repeated release of sterile males over low population densities can further reduce and in cases of isolation eliminate pest populations, although cost-effective control with dense target populations is subjected to population suppression prior to the release of the sterile males.
The technique has successfully been used to eradicate the screw-worm fly (Cochliomyia hominivorax) from North and Central America. Many successes have been achieved for control of fruit fly pests, most particularly the Mediterranean fruit fly (Ceratitis capitata) and the Mexican fruit fly (Anastrepha ludens). Active research is being conducted to determine this technique's effectiveness in combatting the Queensland Fruit Fly (Bactrocera tyroni).
Sterilization is induced through the effects of irradiation on the reproductive cells of the insects. SIT does not involve the release of insects modified through transgenic (genetic engineering) processes.[3] Moreover, SIT does not introduce non-native species into an ecosystem.
Pheromones: The Sex Trap
A pheromone trap is a type of insect trap that uses pheromones to lure insects. Sex pheromones and aggregating pheromones are the most common types used. A pheromone-impregnated lure, as the red rubber septa in the picture, is encased in a conventional trap such as a bottle trap, Delta trap, water-pan trap, or funnel trap. Pheromone traps are used both to count insect populations by sampling, and to trap pests such as clothes moths to destroy them.
Juvenile Hormone (JH)
In some insects, the final molt produces an adult that looks pretty much like the earlier larval stages. In others, like moths and butterflies, the final molt of the larva (caterpillar) produces a pupa. After a period of metamorphosis, the adult moth emerges.
Ecdysone (a steroidal hormone) triggers larva-to-larva molts as long as another hormone, called juvenile hormone (JH) see (Figure \(\PageIndex{3}\) ), is present. In its absence, ecdysone promotes the pupa-to-adult molt. Thus normal metamorphosis seems to occur when the output of JH diminishes spontaneously in the mature caterpillar.
When solutions of JH are sprayed on mature caterpillars, or on the foliage upon which they are feeding, their normal development is upset. This raises the possibility of using JH as an insecticide (one that might avoid the problem of developing resistance).
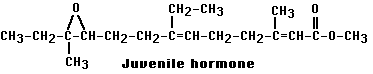
As it turns out, JH is too unstable to be practical, but some synthetic JH mimics, e.g.,
- methoprene (Altosid®)
- pyriproxyfen (Esteem®, Knack®, Distance®)
- diofenolan
are now being used.
Summary
- The term pesticide includes the substances used to control pests (which includes insects, mites, nematodes, plant pathogens, weeds, and vertebrates).
- Many pesticides can be grouped into chemical families which mainly include the insecticides organochlorines (e.g. DDT and dieldrin), organophosphates (e.g. parathion and malathion), and carbamates (e.g. carbaryl and aldicarb).
- Use of organic and biological pesticides are alternative means of pest control that is gaining popularity.
Contributor
- Wikipedia
- USDA
- Libretext: Biology (Kimball)

