20.1: Farming and Chemicals- Fertilizers
- Page ID
- 152302
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Identify the three main macronutrients, the secondary macronutrients, and micronutrients that are essential for plant growth.
- Describe the different types of commercial fertilizers.
A fertilizer is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply one or more plant nutrients essential to the growth of plants. Many sources of fertilizer exist, both natural and industrially produced.[1]Commercial fertilizers, are applied to agricultural crops to increase crop yields, using equipment similar to what is shown in Figure \(\PageIndex{1}\). Before the 1950s, most farming occurred on small family farms with limited use of chemicals. The shift since then to larger corporate farms has coincided with the use of chemical fertilizers in modern agricultural practices. The three major types of commercial fertilizer used in the U.S. are nitrogen, phosphate, and potash.
Fertilizers enhance the growth of plants. This goal is met in two ways, the traditional one being additives that provide nutrients. The second mode by which some fertilizers act is to enhance the effectiveness of the soil by modifying its water retention and aeration. This article, like many on fertilizers, emphasizes the nutritional aspect. Fertilizers typically provide, in varying proportions:[15]
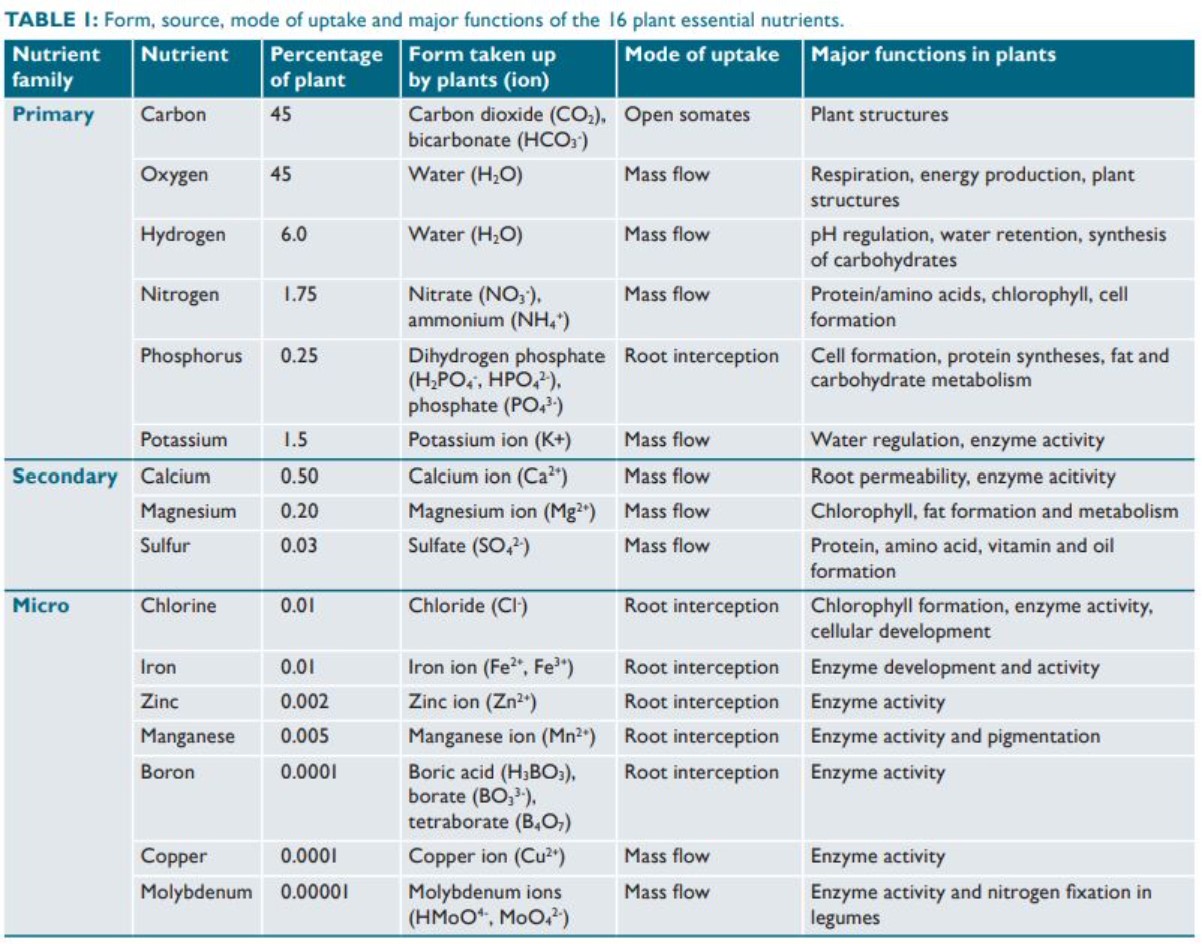
- three main macronutrients:
- Nitrogen (N): leaf growth
- Phosphorus (P): Development of roots, flowers, seeds, fruit;
- Potassium (K): Strong stem growth, movement of water in plants, promotion of flowering and fruiting;
- three secondary macronutrients: calcium (Ca), magnesium (Mg), and sulfur (S);
- micronutrients: copper (Cu), iron (Fe), manganese (Mn), molybdenum (Mo), zinc (Zn), boron (B). Of occasional significance are silicon (Si), cobalt (Co), and vanadium (V).
The nutrients required for healthy plant life are classified according to the elements, but the elements are not used as fertilizers. Instead compounds containing these elements are the basis of fertilizers. The macro-nutrients are consumed in larger quantities and are present in plant tissue in quantities from 0.15% to 6.0% on a dry matter (DM) (0% moisture) basis. Plants are made up of four main elements: hydrogen, oxygen, carbon, and nitrogen. Carbon, hydrogen and oxygen are widely available as water and carbon dioxide. Although nitrogen makes up most of the atmosphere, it is in a form that is unavailable to plants. Nitrogen is the most important fertilizer since nitrogen is present in proteins, DNA and other components (e.g., chlorophyll). To be nutritious to plants, nitrogen must be made available in a "fixed" form. Only some bacteria and their host plants (notably legumes) can fix atmospheric nitrogen (N2) by converting it to ammonia. Phosphate is required for the production of DNA and ATP, the main energy carrier in cells, as well as certain lipids. Table \(\PageIndex{1}\) lists features of various plant essential nutrients.

Figure \(\PageIndex{2}\) A large, modern fertilizer spreader (right). Source: WIkipedia
Nitrogen Fertilizers
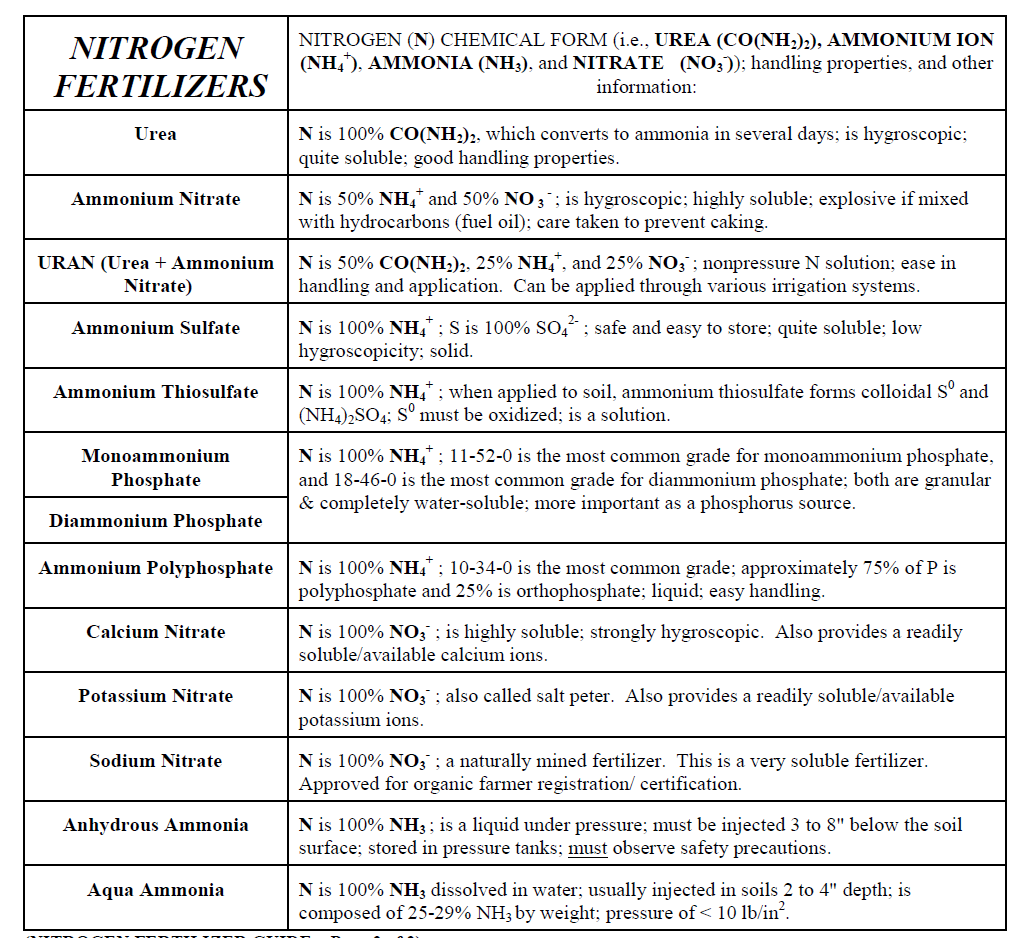
Nitrogen fertilizers are made from ammonia (NH3) produced by the Haber-Bosch process. In this energy-intensive process, natural gas (CH4) usually supplies the hydrogen, and the nitrogen (N2) is derived from the air. This ammonia is used as a feedstock for all other nitrogen fertilizers, such as anhydrous ammonium nitrate (NH4NO3) and urea (CO(NH2)2).
Deposits of sodium nitrate (NaNO3) (Chilean saltpeter) are also found in the Atacama desert in Chile and was one of the original (1830) nitrogen-rich fertilizers used. It is still mined for fertilizer. Nitrates are also produced from ammonia by the Ostwald process. Detailed information on different nitrogen fertilizers is given in (Table \(\PageIndex{2}\)).
Table \(\PageIndex{2}\) USDA Nitrogen Fertilizer Guide.
https://www.nrcs.usda.gov/Internet/F...4p2_068185.pdf
Phosphorus Fertilizers
Phosphate fertilizers are obtained by extraction from phosphate rock, which contains two principal phosphorus-containing minerals, fluorapatite Ca5(PO4)3F (CFA) and hydroxyapatite Ca5(PO4)3OH. These minerals are converted into water-soluble phosphate salts by treatment with sulfuric (H2SO4) or phosphoric acids (H3PO4). The large production of sulfuric acid is primarily motivated by this application. In the nitrophosphate process or Odda process (invented in 1927), phosphate rock with up to a 20% phosphorus (P) content is dissolved with nitric acid (HNO3) to produce a mixture of phosphoric acid (H3PO4) and calcium nitrate (Ca(NO3)2). This mixture can be combined with a potassium fertilizer to produce a compound fertilizer with the three macronutrients N, P and K in easily dissolved form.
Potassium Fertilizers
Potash is a mixture of potassium minerals used to make potassium (K) fertilizers. Potash is soluble in water, so the main effort in producing this nutrient from the ore involves some purification steps; e.g., to remove sodium chloride (NaCl) (common salt). Sometimes potash is referred to as K2O, as a matter of convenience to those describing the potassium content. In fact, potash fertilizers are usually potassium chloride, potassium sulfate, potassium carbonate, or potassium nitrate.[29]
Other Essential Elements
Calcium, magnesium, and sulfur are essential to plant growth but are needed in lesser amounts than nitrogen, phosphorus, and potassium but in larger amounts than the micronutrients. Calcium, magnesium, and phosphorus alter pH when added to the soil. Calcium and magnesium results to an elevation of pH levels while sulfur does the opposite.
Calcium provides structural support in growing plants. Magnesium is the central atom found in the chlorophyll molecule and thus has an important role in photosynthesis. Sulfur is needed in chlorophyll development and protein synthesis.
Micronutrients are consumed in smaller quantities and are present in plant tissue on the order of parts-per-million (ppm), ranging from 0.15 to 400 ppm or less than 0.04% dry matter.[16][17] These elements are often present at the active sites of enzymes that carry out the plant's metabolism. Because these elements enable catalysts (enzymes) their impact far exceeds their weight percentage. The main micronutrients are molybdenum, zinc, boron, and copper. These elements are provided as water-soluble salts. Iron presents special problems because it converts to insoluble (bio-unavailable) compounds at moderate soil pH and phosphate concentrations. For this reason, iron is often administered as a chelate complex, e.g., the EDTA derivative. The micronutrient needs depend on the plant and the environment. For example, sugar beets appear to require boron, and legumes require cobalt,[1] while environmental conditions such as heat or drought make boron less available for plants.[23]
Fertilizers a Mixed Bag
Multi nutrient fertilizers are common. They consist of two or more nutrient components.
Binary (NP, NK, PK) fertilizers. Major two-component fertilizers provide both nitrogen and phosphorus to the plants. These are called NP fertilizers. The main NP fertilizers are monoammonium phosphate (MAP) and diammonium phosphate (DAP). The active ingredient in MAP is NH4H2PO4. The active ingredient in DAP is (NH4)2HPO4. About 85% of MAP and DAP fertilizers are soluble in water.
NPK fertilizers are three-component fertilizers providing nitrogen, phosphorus, and potassium.
NPK rating is a rating system describing the amount of nitrogen, phosphorus, and potassium in a fertilizer. NPK ratings consist of three numbers separated by dashes (e.g., 10-10-10 or 16-4-8) describing the chemical content of fertilizers.[20][21] The first number represents the percentage of nitrogen in the product; the second number, P2O5; the third, K2O. Fertilizers do not actually contain P2O5 or K2O, but the system is a conventional shorthand for the amount of the phosphorus (P) or potassium (K) in a fertilizer. A 50-pound (23 kg) bag of fertilizer labeled 16-4-8 contains 8 lb (3.6 kg) of nitrogen (16% of the 50 pounds), an amount of phosphorus equivalent to that in 2 pounds of P2O5 (4% of 50 pounds), and 4 pounds of K2O (8% of 50 pounds). Most fertilizers are labeled according to this N-P-K convention, although Australian convention, following an N-P-K-S system, adds a fourth number for sulfur, and uses elemental values for all values including P and K.[22]
Organic Fertilizers
“Organic fertilizers” can be described as those fertilizers with an organic — biologic — origin—that is, fertilizers derived from living or formerly living materials. Organic fertilizers can also describe commercially available and frequently packaged products that strive to follow the expectations and restrictions adopted by “organic agriculture” and ”environmentally friendly" gardening — related systems of food and plant production that significantly limit or strictly avoid the use of synthetic fertilizers and pesticides. The “organic fertilizer” products typically contain both some organic materials as well as acceptable additives such as nutritive rock powders, ground sea shells (crab, oyster, etc.), other prepared products such as seed meal or kelp, and cultivated microorganisms and derivatives.
Fertilizers of an organic origin (the first definition) include animal wastes, plant wastes from agriculture, compost, and treated sewage sludge (biosolids). Beyond manures, animal sources can include products from the slaughter of animals — bloodmeal, bone meal, feather meal, hides, hoofs, and horns all are typical components.[15] Organically derived materials available to industry such as sewage sludge may not be acceptable components of organic farming and gardening, because of factors ranging from residual contaminants to public perception. On the other hand, marketed “organic fertilizers” may include, and promote, processed organics because the materials have consumer appeal. No matter the definition nor composition, most of these products contain less-concentrated nutrients, and the nutrients are not as easily quantified. They can offer soil-building advantages as well as be appealing to those who are trying to farm / garden more “naturally”.[30]
In terms of volume, peat is the most widely used packaged organic soil amendment. It is an immature form of coal and improves the soil by aeration and absorbing water but confers no nutritional value to the plants. It is therefore not a fertilizer as defined in the beginning of the article, but rather an amendment. Coir, (derived from coconut husks), bark, and sawdust when added to soil all act similarly (but not identically) to peat and are also considered organic soil amendments - or texturizers - because of their limited nutritive inputs. Some organic additives can have a reverse effect on nutrients — fresh sawdust can consume soil nutrients as it breaks down, and may lower soil pH — but these same organic texturizers (as well as compost, etc.) may increase the availability of nutrients through improved cation exchange, or through increased growth of microorganisms that in turn increase availability of certain plant nutrients. Organic fertilizers such as composts and manures may be distributed locally without going into industry production, making actual consumption more difficult to quantify.
Effects of Fertilizer Runoffs
Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways. Agricultural run-off is a major contributor to the eutrophication of fresh water bodies. For example, in the US, about half of all the lakes are eutrophic. The main contributor to eutrophication is phosphate, which is normally a limiting nutrient; high concentrations promote the growth of cyanobacteria and algae, the demise of which consumes oxygen. Cyanobacteria blooms ('algal blooms') can also produce harmful toxins that can accumulate in the food chain, and can be harmful to humans.
The nitrogen-rich compounds found in fertilizer runoff are the primary cause of serious oxygen depletion in many parts of oceans, especially in coastal zones, lakes and rivers. The resulting lack of dissolved oxygen greatly reduces the ability of these areas to sustain oceanic fauna. The number of oceanic dead zones near inhabited coastlines are increasing. As of 2006, the application of nitrogen fertilizer is being increasingly controlled in northwestern Europe and the United States. If eutrophication can be reversed, it may take decades[citation needed] before the accumulated nitrates in groundwater can be broken down by natural processes.
Summary
- A fertilizer is any material (of natural or synthetic origin) that is applied to soil or to plant tissues that provides one or more plant nutrients essential to the growth of plants.
- Nitrogen, phosphorus, and potassium are the main macronutrients and calcium, magnesium, and sulfur are the secondary macronutrients in fertilizers.
- Copper (Cu), iron (Fe), manganese (Mn), molybdenum (Mo), zinc (Zn), boron (B) are essential micronutrients that are needed by plants in relatively smaller quantities than the macronutrients.
- Phosphorus and nitrogen fertilizers when commonly used have major environmental effects. This is due to high rainfalls causing the fertilizers to be washed into waterways. Agricultural run-off is a major contributor to the eutrophication of fresh water bodies.
Contributors and Attributions
- Wikipedia
US Department of Agriculture (USDA)
“Secondary Plant Nutrients: Calcium, Magnesium, and Sulfur.” Mississippi State University Extension Service, extension.msstate.edu/publications/secondary-plant-nutrients-calcium-magnesium-and-sulfur.
“Essential Nutrients for Plants - How Do Nutrients Affect Plant Growth?” Texas A&M AgriLife Extension Service, 4 Mar. 2019, agrilifeextension.tamu.edu/library/gardening/essential-nutrients-for-plants/.