11.2: Nuclear Equations
- Page ID
- 152204
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Identify common particles and energies involved in nuclear reactions.
- Write and balance nuclear equations.
Changes of nuclei that result in changes in their atomic numbers, mass numbers, or energy states are nuclear reactions. To describe a nuclear reaction, we use an equation that identifies the nuclides involved in the reaction, their mass numbers and atomic numbers, and the other particles involved in the reaction.
Types of Particles in Nuclear Reactions
Many entities can be involved in nuclear reactions. The most common are protons, neutrons, alpha particles, beta particles, positrons, and gamma rays, as shown in Table \(\PageIndex{1}\).
Table \(\PageIndex{1}\) A Summary of the Names, Symbols, Representations, and Descriptions of the Most Common Particles in Nuclear Reactions.

Balancing Nuclear Reactions
A balanced chemical reaction equation reflects the fact that during a chemical reaction, bonds break and form, and atoms are rearranged, but the total numbers of atoms of each element are conserved and do not change. A balanced nuclear reaction equation indicates that there is a rearrangement during a nuclear reaction, but of subatomic particles rather than atoms. Nuclear reactions also follow conservation laws, and they are balanced in two ways:
- The sum of the mass numbers of the reactants equals the sum of the mass numbers of the products.
- The sum of the charges of the reactants equals the sum of the charges of the products.
If the atomic number and the mass number of all but one of the particles in a nuclear reaction are known, we can identify the particle by balancing the reaction. For instance, we could determine that \(\ce{^{17}_8O}\) is a product of the nuclear reaction of \(\ce{^{14}_7N}\) and \(\ce{^4_2He}\) if we knew that a proton, \(\ce{^1_1H}\), was one of the two products. Example \(\PageIndex{1}\) shows how we can identify a nuclide by balancing the nuclear reaction.
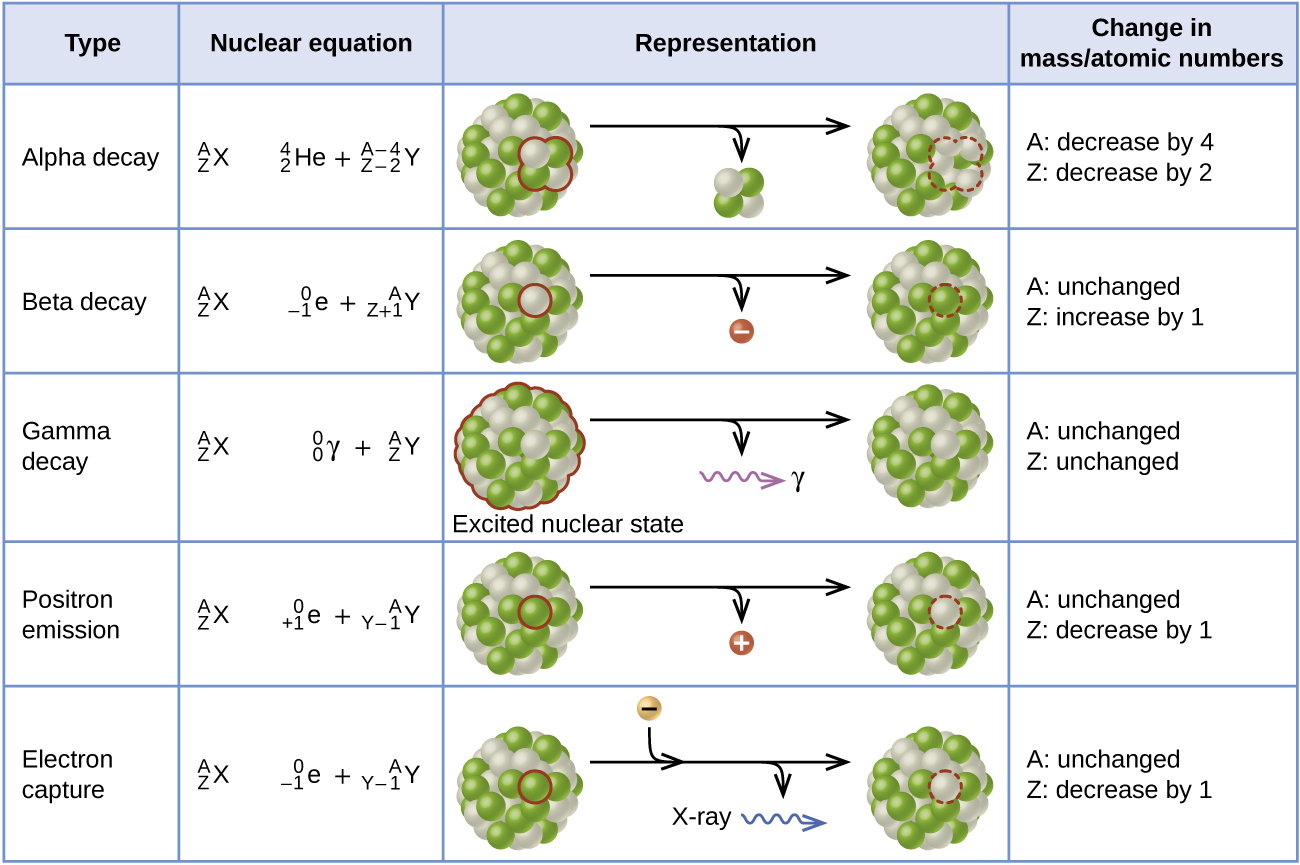
Nuclear Decay Processes
Radioactive decay involves the emission of a particle and/or energy as one atom changes into another. In most instances, the atom changes its identity to become a new element. There are four different types of emissions that occur.
Alpha Emission
Alpha \(\left( \alpha \right)\) decay involves the release of helium ions from the nucleus of an atom. This ion consists of two protons and two neutrons and has a \(2+\) charge. Release of an \(\alpha\)-particle produces a new atom that has an atomic number two less than the original atom and an atomic weight that is four less. A typical alpha decay reaction is the conversion of uranium-238 to thorium:
\[\ce{^{238}_{92}U} \rightarrow \ce{^{234}_{90}Th} + \ce{^4_2 \alpha}^+ \nonumber \]
We see a decrease of two in the atomic number (uranium to thorium) and a decrease of four in the atomic weight (238 to 234). Usually the emission is not written with atomic number and weight indicated since it is a common particle whose properties should be memorized. Quite often the alpha emission is accompanied by gamma \(\left( \gamma \right)\) radiation, a form of energy release. Many of the largest elements in the periodic table are alpha-emitters.
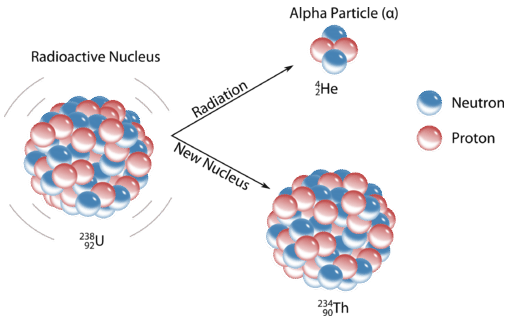
Chemists often use the names parent isotope and daughter isotope to represent the original atom and the product other than the alpha particle. In the previous example, \[_{92}^{238}\textrm{U} \nonumber \] is the parent isotope, and \[_{90}^{234}\textrm{Th} \nonumber \] is the daughter isotope. When one element changes into another in this manner, it undergoes radioactive decay.
Write the nuclear equation that represents the radioactive decay of radon-222 by alpha particle emission and identify the daughter isotope.
Solution
Radon has an atomic number of 86, so the parent isotope is represented as \[_{86}^{222}\textrm{Rn} \nonumber \]
We represent the alpha particle as
\[_{2}^{4}\textrm{He} \nonumber \]
Use subtraction (222 − 4 = 218 and 86 − 2 = 84) to identify the daughter isotope as polonium:
\[_{86}^{222}\textrm{Rn}\rightarrow \; _{2}^{4}\textrm{He}+\: _{84}^{218}\textrm{Th} \nonumber \]
Write the nuclear equation that represents radioactive decay of polonium-208 by alpha particle emission and identify the daughter isotope.
- Answer
-
\[_{80}^{208}\textrm{Po}\rightarrow \; _{2}^{4}\textrm{He}+\: _{82}^{204}\textrm{Pb} \nonumber \]
\[_{82}^{204}\textrm{Pb} \nonumber \]
Beta Emission
Beta \(\left( \beta \right)\) decay is a more complicated process. Unlike the \(\alpha\)-emission, which simply expels a particle, the \(\beta\)-emission involves the transformation of a neutron in the nucleus to a proton and an electron. The electron is then ejected from the nucleus. In the process,the atomic number increases by one while the atomic weight stays the same. As is the case with \(\alpha\)-emissions, \(\beta\)-emissions are often accompanied by \(\gamma\)-radiation.
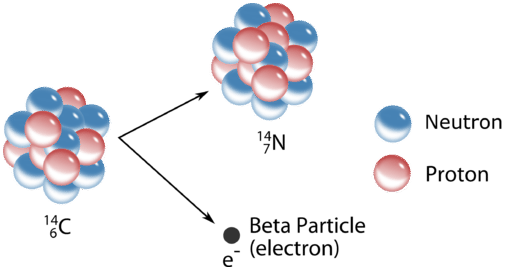
A typical beta decay process involves carbon-14, often used in radioactive dating techniques. The reaction forms nitrogen-14 and an electron:
\[\ce{^{14}_6C} \rightarrow \ce{^{14}_7N} + \ce{^0_{-1}e} \nonumber \]
Again, the beta emission is usually simply indicated by the Greek letter \(\beta\); memorization of the process is necessary in order to follow nuclear calculations in which the Greek letter \(\beta\) appears without further notation.
Write the nuclear equation that represents the radioactive decay of boron-12 by beta particle emission and identify the daughter isotope. A gamma ray is emitted simultaneously with the beta particle.
Solution
The parent isotope is \(\ce{_{2}^{4}He} \nonumber \)
while one of the products is \(\ce{_{-1}^{0}e} \nonumber \)
So that the mass and atomic numbers have the same value on both sides, the mass number of the daughter isotope must be 12, and its atomic number must be 6. The element having an atomic number of 6 is carbon. Thus the complete nuclear equation is as follows:
\[_{5}^{12}\textrm{B}\rightarrow \;_{6}^{12}\textrm{C}+_{-1}^{0}\textrm{e}+\gamma \nonumber \]
The daughter isotope is carbon-12.
Write the nuclear equation that represents the radioactive decay of rubidium-87 by beta particle emission and identify the daughter isotope.
- Answer
-
\[_{37}^{87}\textrm{Rb}\rightarrow \;_{38}^{87}\textrm{Sr}+_{-1}^{0}\textrm{e} \nonumber \]
\[_{38}^{87}\textrm{Sr} \nonumber \]
Gamma Emission
Gamma \(\left( \gamma \right)\) radiation is simply energy. It may be released by itself or more commonly in association with other radiation events. There is no change of atomic number of atomic weight in a simple \(\gamma\)-emission. Often, an isotope may produce \(\gamma\)-radiation as a result of a transition in a metastable isotope. This type of isotope may just "settle", with a shifting of particles in the nucleus. The composition of the atom is not altered, but the nucleus could be considered more "comfortable" after the shift. This shift increases the stability of the isotope from the energetically unstable (or "metastable") isotope to a more stable form of the nucleus. Gamma (\(\gamma\)) emission can occur virtually instantaneously, as it does in the alpha decay of uranium-238 to thorium-234, where the asterisk denotes an excited state:
\[^{238}_{92}\textrm{U}\rightarrow \, \underset{\textrm{excited} \\ \textrm{nuclear} \\ \textrm{state}}{^{234}_{90}\textrm{Th*}} + ^{4}_{2}\alpha\xrightarrow {\textrm{relaxation}\,}\,^{234}_{90}\textrm{Th}+^{0}_{0}\gamma\label{Eq13} \]
If we disregard the decay event that created the excited nucleus, then
\[^{234}_{88}\textrm{Th*} \rightarrow\, ^{234}_{88}\textrm{Th}+^{0}_{0}\gamma\label{Eq14} \]
or more generally,
\[^{A}_{Z}\textrm{X*} \rightarrow\, ^{A}_{Z}\textrm{X}+^{0}_{0}\gamma\label{Eq15} \]
Gamma emission can also occur after a significant delay. For example, technetium-99m has a half-life of about 6 hours before emitting a γ ray to form technetium-99 (the m is for metastable). Because γ rays are energy, their emission does not affect either the mass number or the atomic number of the daughter nuclide. Gamma-ray emission is therefore the only kind of radiation that does not necessarily involve the conversion of one element to another, although it is almost always observed in conjunction with some other nuclear decay reaction.
Positron Emission
A positron is a positive electron (a form of antimatter). This rare type of emission occurs when a proton is converted to a neutron and a positron in the nucleus, with ejection of the positron. The atomic number will decrease by one while the atomic weight does not change. A positron is often designated by \(\beta^+\).
Carbon-11 emits a positron to become boron-11:
\[\ce{^{11}_6C} \rightarrow \ce{^{11}_5B} + \ce{^0_{+1} \beta} \nonumber \]
Electron Capture
An alternate way for a nuclide to increase its neutron to proton ratio is by a phenomenon called electron capture. In electron capture, an electron from an inner orbital is captured by the nucleus of the atom and combined with a proton to form a neutron. For example, silver-106 undergoes electron capture to become palladium-106.
\[\ce{^{106}_{47}Ag} + \ce{^0_{-1}e} \rightarrow \ce{^{106}_{46}Pd} \nonumber \]
Note that the overall result of electron capture is identical to positron emission. The atomic number decreases by one while the mass number remains the same.
Table \(\PageIndex{2}\) Different Types of Decay and Changes in Atomic and Mass Numbers.
Following are the equations of several nuclear reactions that have important roles in the history of nuclear chemistry:
- The first naturally occurring unstable element that was isolated, polonium, was discovered by the Polish scientist Marie Curie and her husband Pierre in 1898. It decays, emitting α particles: \[\ce{^{212}_{84}Po⟶ ^{208}_{82}Pb + ^4_2He} \nonumber \]
- The first nuclide to be prepared by artificial means was an isotope of oxygen, 17O. It was made by Ernest Rutherford in 1919 by bombarding nitrogen atoms with α particles: \[\ce{^{14}_7N + ^4_2α⟶ ^{17}_8O + ^1_1H} \nonumber \]
- James Chadwick discovered the neutron in 1932, as a previously unknown neutral particle produced along with 12C by the nuclear reaction between 9Be and 4He: \[\ce{^9_4Be + ^4_2He⟶ ^{12}_6C + ^1_0n} \nonumber \]
- The first element to be prepared that does not occur naturally on the earth, technetium, was created by bombardment of molybdenum by deuterons (heavy hydrogen, \(\ce{^2_1H}\)), by Emilio Segre and Carlo Perrier in 1937: \[ \ce{^2_1H + ^{97}_{42}Mo⟶2^1_0n + ^{97}_{43}Tc} \nonumber \]
- The first controlled nuclear chain reaction was carried out in a reactor at the University of Chicago in 1942. One of the many reactions involved was: \[ \ce{^{235}_{92}U + ^1_0n⟶ ^{87}_{35}Br + ^{146}_{57}La + 3^1_0n} \nonumber \]
Summary
- Nuclei can undergo reactions that change their number of protons, number of neutrons, or energy state.
- Many different particles can be involved in nuclear reactions. The most common are protons, neutrons, positrons (which are positively charged electrons), electrons, alpha (α) particles (which are high-energy helium nuclei), beta (β) particles (which are high-energy electrons), and gamma (γ) rays (which compose high-energy electromagnetic radiation).
- As with chemical reactions, nuclear reactions are always balanced. When a nuclear reaction occurs, the total mass (number) and the total charge remain unchanged.
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).

