4.5: Formulas and Names of Binary Ionic Compounds
- Page ID
- 152158
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Use the rules for naming ionic compounds.
- Write the correct formula for an ionic compound.
Names of Binary ionic Compounds
After learning a few more details about the names of individual ions, you will be a step away from knowing how to name ionic compounds. This section begins the formal study of nomenclature, the systematic naming of chemical compounds.
Naming Ions
The name of a monatomic cation is simply the name of the element followed by the word ion. Thus, Na+ is the sodium ion, Al3+ is the aluminum ion, Ca2+ is the calcium ion, and so forth.
We have seen that some elements lose different numbers of electrons, producing ions of different charges (Figure 4.4.1) . Iron, for example, can form two cations, each of which, when combined with the same anion, makes a different compound with unique physical and chemical properties. Thus, we need a different name for each iron ion to distinguish Fe2+ from Fe3+. The same issue arises for other ions with more than one possible charge.
There are two ways to make this distinction. In the simpler, more modern approach, called the Stock system (Table \(\PageIndex{1}\)), an ion’s positive charge is indicated by a roman numeral in parentheses after the element name, followed by the word ion. Thus, Fe2+ is called the iron(II) ion, while Fe3+ is called the iron(III) ion. This system is used only for elements that form more than one common positive ion. We do not call the Na+ ion the sodium(I) ion because (I) is unnecessary. Sodium forms only a 1+ ion, so there is no ambiguity about the name sodium ion.
| Element | Stem | Charge |
Modern Name (Stock System) |
Common Name |
|---|---|---|---|---|
| iron | ferr- | 2+ | iron(II) ion | ferrous ion |
| 3+ | iron(III) ion | ferric ion | ||
| copper | cupr- | 1+ | copper(I) ion | cuprous ion |
| 2+ | copper(II) ion | cupric ion | ||
| tin | stann- | 2+ | tin(II) ion | stannous ion |
| 4+ | tin(IV) ion | stannic ion | ||
| lead | plumb- | 2+ | lead(II) ion | plumbous ion |
| 4+ | lead(IV) ion | plumbic ion | ||
| chromium | chrom- | 2+ | chromium(II) ion | chromous ion |
| 3+ | chromium(III) ion | chromic ion | ||
| gold | aur- | 1+ | gold(I) ion | aurous ion |
| 3+ | gold(III) ion | auric ion |
The second system, called the common system, is not conventional but is still prevalent and used in the health sciences. This system recognizes that many metals have two common cations. The common system uses two suffixes (-ic and -ous) that are appended to the stem of the element name. The -ic suffix represents the greater of the two cation charges, and the -ous suffix represents the lower one. In many cases, the stem of the element name comes from the Latin name of the element. Table \(\PageIndex{1}\) lists the elements that use the common system, along with their respective cation names.
| Ion | Name |
|---|---|
| F− | fluoride ion |
| Cl− | chloride ion |
| Br− | bromide ion |
| I− | iodide ion |
| O2− | oxide ion |
| S2− | sulfide ion |
| P3− | phosphide ion |
| N3− | nitride ion |
The name of a monatomic anion consists of the stem of the element name, the suffix -ide, and then the word ion. Thus, as we have already seen, Cl− is “chlor-” + “-ide ion,” or the chloride ion. Similarly, O2− is the oxide ion, Se2− is the selenide ion, and so forth. Table \(\PageIndex{2}\) lists the names of some common monatomic ions.
Name each ion.
- Ca2+
- S2−
- Cu+
Solution
- the calcium ion
- the sulfide ion
- the copper(I) ion or the cuprous ion
Name each ion.
- Fe2+
- Fe3+
- Ba2+
- Answer a:
- iron(II) ion
- Answer b:
- iron(III) ion
- Answer c:
- barium ion
Write the formula for each ion.
- the bromide ion
- the cupric ion
- the magnesium ion
Solution
- Br−
- Cu2+
- Mg2+
Write the formula for each ion.
- the fluoride ion
- the stannous ion
- the potassium ion
- Answer a:
- F-
- Answer b:
- Sn 2+
- Answer c:
- K+
Naming Binary Ionic Compounds with a Metal that Forms Only One Type of Cation
Now that we know how to name ions, we are ready to name ionic compounds. A binary ionic compound is a compound composed of a monatomic metal cation and a monatomic nonmetal anion. The metal cation is named first, followed by the nonmetal anion as illustrated in Figure \(\PageIndex{1}\) for the compound BaCl2. The word ion is dropped from both parts.
Subscripts in the formula do not affect the name.
Name each ionic compound.
- CaCl2
- AlF3
- KCl
Solution
- Using the names of the ions, this ionic compound is named calcium chloride.
- The name of this ionic compound is aluminum fluoride.
- The name of this ionic compound is potassium chloride
Name each ionic compound.
- AgI
- MgO
- Ca3P2
- Answer a:
- silver iodide
- Answer b:
- magnesium oxide
- Answer c:
- calcium phosphide
Naming Binary Ionic Compounds with a Metal That Forms More Than One Type of Cation
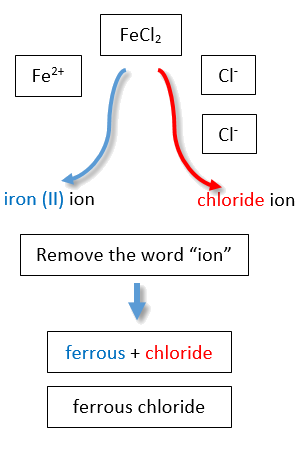
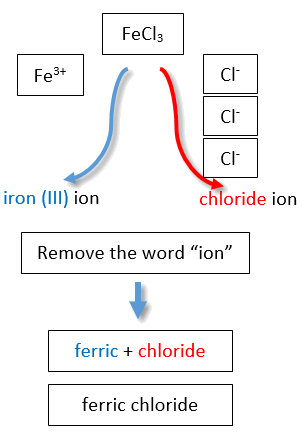
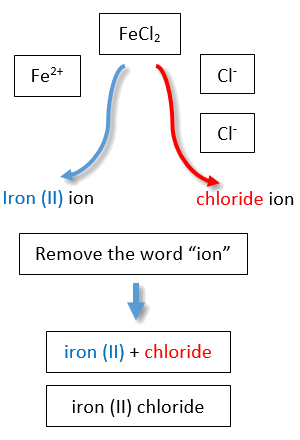
If you are given a formula for an ionic compound whose cation can have more than one possible charge, you must first determine the charge on the cation before identifying its correct name. For example, consider FeCl2 and FeCl3 . In the first compound, the iron ion has a 2+ charge because there are two Cl− ions in the formula (1− charge on each chloride ion). In the second compound, the iron ion has a 3+ charge, as indicated by the three Cl− ions in the formula. These are two different compounds that need two different names. By the Stock system, the names are iron(II) chloride and iron(III) chloride (Figure \(\PageIndex{2}\)).
| Name of cation (metal) + (Roman Numeral in parenthesis) + Base name of anion (nonmetal) and -ide | |
|---|---|
 |
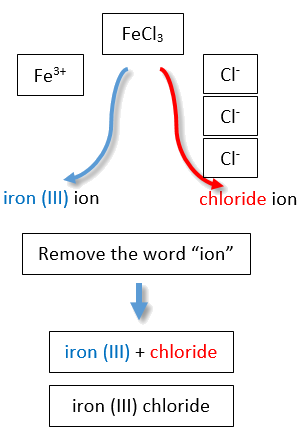 |
If we were to use the stems and suffixes of the common system, the names would be ferrous chloride and ferric chloride, respectively (Figure \(\PageIndex{3}\)) .
| "Old" base name of cation (metal) and -ic or -ous + Base name of anion (nonmetal) and -ide | |
|---|---|
|
-ous (for ions with lower charge)
|
-ic (for ions with higher charge)
|
Name each ionic compound.
- Co2O3
- FeCl2
Solution
| Ionic Compound | Explanation | Answer |
|---|---|---|
| Co2O3 |
We know that cobalt can have more than one possible charge; we just need to determine what it is.
|
cobalt(III) oxide |
| FeCl2 |
Iron can also have more than one possible charge.
|
iron(II) chloride |
Name each ionic compound.
- AuCl3
- PbO2
- CuO
- Answer a:
- gold(III) chloride
- Answer b:
- lead(IV) oxide
- Answer c:
- copper(II) oxide
Figure \(\PageIndex{4}\) is a synopsis of how to name simple ionic compounds.
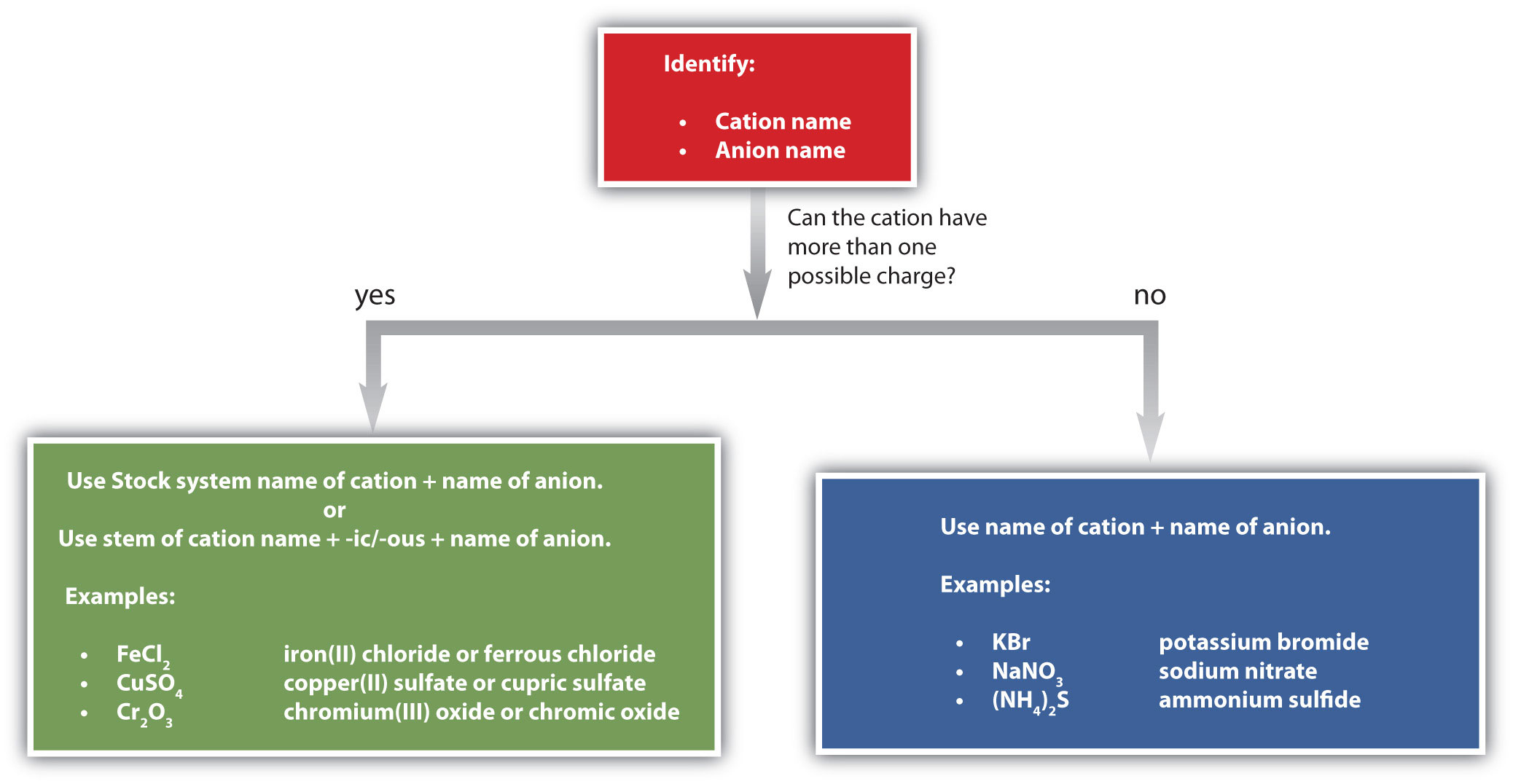
Name each ionic compound.
- ZnBr2
- Al2O3
- AuF3
- AgF
- Answer a:
- zinc bromide
- Answer b:
- aluminum oxide
- Answer c:
- gold(III) fluoride or auric fluoride
- Answer d:
- silver fluoride
Writing Formulas of Ionic Compounds
Ionic compounds do not exist as molecules. In the solid state, ionic compounds are in crystal lattice containing many ions each of the cation and anion. An ionic formula, like \(\ce{NaCl}\), is an empirical formula. This formula merely indicates that sodium chloride is made of an equal number of sodium and chloride ions. Sodium sulfide, another ionic compound, has the formula \(\ce{Na_2S}\). This formula indicates that this compound is made up of twice as many sodium ions as sulfide ions. This section will teach you how to find the correct ratio of ions, so that you can write a correct formula.
If you know the name of a binary ionic compound, you can write its chemical formula. Start by writing the metal ion with its charge, followed by the nonmetal ion with its charge. Because the overall compound must be electrically neutral, decide how many of each ion is needed in order for the positive and negative charge to cancel each other out.
Write the formula for aluminum nitride and lithium oxide.
Solution
| Steps for Problem Solving | Write the formula for aluminum nitride | Write the formula for lithium oxide |
|---|---|---|
| 1. Write the symbol and charge of the cation (metal) first and the anion (nonmetal) second. | \(\ce{Al^{3+}} \: \: \: \: \: \ce{N^{3-}}\) | \(\ce{Li^+} \: \: \: \: \: \ce{O^{2-}}\) |
| 2. Use a multiplier to make the total charge of the cations and anions equal to each other. |
total charge of cations = total charge of anions 1(3+) = 1(3-) +3 = -3 |
total charge of cations = total charge of anions 2(1+) = 1(2-) +2 = -2 |
| 3. Use the multipliers as subscript for each ion. | \(\ce{Al_1N_1}\) | \(\ce{Li_2O_1}\) |
| 4. Write the final formula. Leave out all charges and all subscripts that are 1. | \(\ce{AlN}\) | \(\ce{Li_2O}\) |
An alternative way to writing a correct formula for an ionic compound is to use the crisscross method. In this method, the numerical value of each of the ion charges is crossed over to become the subscript of the other ion. Signs of the charges are dropped.
Write the formula for lead (IV) oxide.
Solution
| Crisscross Method | Example: Write the formula for lead (IV) oxide |
|---|---|
| 1. Write the symbol and charge of the cation (metal) first and the anion (nonmetal) second. | \(\ce{Pb^{4+}} \: \: \: \: \: \ce{O^{2-}}\) |
| 2. Transpose only the number of the positive charge to become the subscript of the anion and the number only of the negative charge to become the subscript of the cation. | 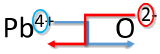 |
| 3. Reduce to the lowest ratio. | \(\ce{Pb_2O_4}\) |
| 4. Write the final formula. Leave out all subscripts that are 1. | \(\ce{PbO_2}\) |
Write the chemical formula for an ionic compound composed of each pair of ions.
- the calcium ion and the oxygen ion
- the 2+ copper ion and the sulfur ion
- the 1+ copper ion and the sulfur ion
- Answer a:
- CaO
- Answer b:
- CuS
- Answer c:
- Cu2S
Be aware that ionic compounds are empirical formulas and so must be written as the lowest ratio of the ions.
Write the formula for sodium combined with sulfur.
Solution
| Crisscross Method | Write the formula for sodium combined with sulfur |
|---|---|
| 1. Write the symbol and charge of the cation (metal) first and the anion (nonmetal) second. | \(\ce{Na^{+}} \: \: \: \: \: \ce{S^{2-}}\) |
| 2. Transpose only the number of the positive charge to become the subscript of the anion and the number only of the negative charge to become the subscript of the cation. | 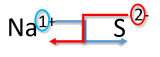 |
| 3. Reduce to the lowest ratio. | This step is not necessary. |
| 4. Write the final formula. Leave out all all subscripts that are 1. | \(\ce{Na_2S}\) |
Write the formula for each ionic compound.
- sodium bromide
- lithium chloride
- magnesium oxide
- Answer a:
- NaBr
- Answer b:
- LiCl
- Answer c:
- MgO
Recognizing Ionic Compounds
There are two ways to recognize ionic compounds. First, compounds between metal and nonmetal elements are usually ionic. For example, CaBr2 contains a metallic element (calcium, a group 2 (or 2A) metal) and a nonmetallic element (bromine, a group 17 (or 7A) nonmetal). Therefore, it is most likely an ionic compound. (In fact, it is ionic.) In contrast, the compound NO2 contains two elements that are both nonmetals (nitrogen, from group 15 (or 5A), and oxygen, from group 16 (or 6A). It is not an ionic compound; it belongs to the category of covalent compounds discussed elsewhere. Also note that this combination of nitrogen and oxygen has no electric charge specified, so it is not the nitrite ion.
Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. For example, if you see the formula Ba(NO3)2, you may recognize the “NO3” part as the nitrate ion, NO3−. (Remember that the convention for writing formulas for ionic compounds is not to include the ionic charge.) This is a clue that the other part of the formula, Ba, is actually the Ba2+ ion, with the 2+ charge balancing the overall 2− charge from the two nitrate ions. Thus, this compound is also ionic.
Identify each compound as ionic or not ionic.
- Na2O
- PCl3
- OF2
Solution
| Explanation | Answer |
|---|---|
| a. Sodium is a metal, and oxygen is a nonmetal. Therefore, Na2O is expected to be ionic. | \(Na_2O\), ionic |
| b. Both phosphorus and chlorine are nonmetals. Therefore, PCl3 is not ionic. | \(PCl_3\), not ionic |
| c. Both oxygen and fluorine are nonmetals. Therefore, OF2 is not ionic. | \(OF_2\), ionic |
Identify each compound as ionic or not ionic.
- N2O
- FeCl3
- Answer a:
- not ionic
- Answer b:
- ionic
Summary
- Ionic compounds are named by stating the cation first, followed by the anion.
- Positive and negative charges must balance.
- Some anions have multiple forms and are named accordingly with the use of roman numerals in parenthesis.
- Formulas for ionic compounds contain the symbols and number of each atom present in a compound in the lowest whole number ratio.
Contributors and Attributions
Henry Agnew (UC Davis)