3.3: Molecules and Chemical Nomenclature
- Page ID
- 64018
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define molecule.
- Name simple molecules based on their formulas.
- Determine a formula of a molecule based on its name.
There are many substances that exist as two or more atoms connected together so strongly that they behave as a single particle. These multiatom combinations are called molecules. A molecule is the smallest part of a substance that has the physical and chemical properties of that substance. In some respects, a molecule is similar to an atom. A molecule, however, is composed of more than one atom.
| Hydrogen (\(\ce{H2}\)) | Oxygen (\(\ce{O2}\)) | Nitrogen (\(\ce{N2}\)) | Fluorine (\(\ce{F2}\)) |
|---|---|---|---|
| Chlorine (\(\ce{Cl2}\)) | Bromine (\(\ce{Br2}\)) | Iodine (\(\ce{I2}\)) |
Some elements exist naturally as molecules. For example, hydrogen and oxygen exist as two-atom molecules. Other elements also exist naturally as diatomic molecules (Table \(\PageIndex{1}\)). As with any molecule, these elements are labeled with a molecular formula, a formal listing of what and how many atoms are in a molecule. (Sometimes only the word formula is used, and its meaning is inferred from the context.) For example, the molecular formula for elemental hydrogen is H2, with H being the symbol for hydrogen and the subscript 2 implying that there are two atoms of this element in the molecule. Other diatomic elements have similar formulas: O2, N2, and so forth. Other elements exist as molecules—for example, sulfur normally exists as an eight-atom molecule, S8, while phosphorus exists as a four-atom molecule, P4 (Figure \(\PageIndex{1}\)). Otherwise, we will assume that elements exist as individual atoms, rather than molecules. It is assumed that there is only one atom in a formula if there is no numerical subscript on the right side of an element's symbol.
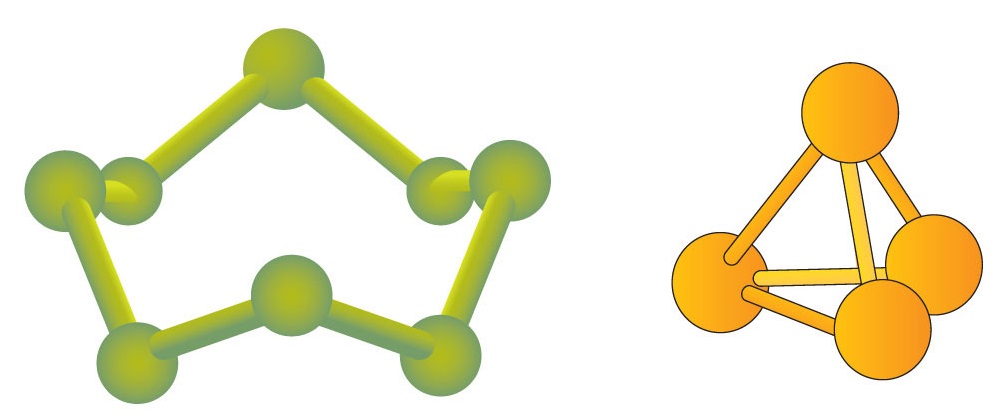
Figure \(\PageIndex{1}\) shows two examples of how we will be representing molecules in this text. An atom is represented by a small ball or sphere, which generally indicates where the nucleus is in the molecule. A cylindrical line connecting the balls represents the connection between the atoms that make this collection of atoms a molecule. This connection is called a chemical bond and is the connection between two atoms in a molecule.
Many compounds exist as molecules. In particular, when nonmetals connect with other nonmetals, the compounds typically exist as molecules. (Compounds between a metal and a nonmetal are different and will be considered in Section 3.4.) In some cases, there are many different kinds of molecules that can be formed between any given elements, with all the different molecules having different chemical and physical properties. How do we tell them apart?
The answer is a very specific system of naming compounds, called chemical nomenclature. By following the rules of nomenclature, each and every compound has its own unique name, and each name refers to one and only one compound. Here, we will start with relatively simple molecules that have only two elements in them, the so-called binary compounds:
- Identify the elements in the molecule from its formula.
- Begin the name with the element name of the first element. If there is more than one atom of this element in the molecular formula, use a numerical prefix to indicate the number of atoms, as listed in Table \(\PageIndex{2}\). Do not use the prefix mono- if there is only one atom of the first element.
- Name the second element by using three pieces:
- a numerical prefix indicating the number of atoms of the second element, plus
- the stem of the element name (e.g., ox for oxygen, chlor for chlorine, etc.), plus
- the suffix -ide
- Combine the two words, leaving a space between them.
| The Number of Atoms of an Element | Prefix |
|---|---|
| 1 | mono- |
| 2 | di- |
| 3 | tri- |
| 4 | tetra- |
| 5 | penta- |
| 6 | hexa- |
| 7 | hepta- |
| 8 | octa- |
| 9 | nona- |
| 10 | deca- |
Let us see how these steps work for a molecule whose molecular formula is SO2, which has one sulfur atom and two oxygen atoms—this completes step 1. According to step 2, we start with the name of the first element—sulfur. Remember, we do not use the mono- prefix for the first element. Now for step 3, we combine the numerical prefix di- (see Table \(\PageIndex{2}\)) with the stem ox- and the suffix -ide, to make dioxide. Bringing these two words together, we have the unique name for this compound—sulfur dioxide.
Why all this trouble? There is another common compound consisting of sulfur and oxygen whose molecular formula is SO3, so the compounds need to be distinguished. SO3 has three oxygen atoms in it, so it is a different compound with different chemical and physical properties. The system of chemical nomenclature is designed to give this compound its own unique name. Its name, if you go through all the steps, is sulfur trioxide. Different compounds have different names.
In some cases, when a prefix ends in a or o; and the element name begins with o, we drop the a or o on the prefix. So we see monoxide or pentoxide, rather than monooxide or pentaoxide in molecule names.
Name each molecule.
- PF3
- CO
- Se2Br2
Solution
- A molecule with a single phosphorus atom and three fluorine atoms is called phosphorus trifluoride.
- A compound with one carbon atom and one oxygen atom is properly called carbon monoxide, not carbon monooxide.
- There are two atoms of each element, selenium and bromine. According to the rules, the proper name here is diselenium dibromide.
Name each molecule.
- SF4
- P2S5
- Answer a
-
sulfur tetrafluoride
- Answer b
-
diphosphorus pentasulfide
One great thing about this system is that it works both ways. From the name of a compound, you should be able to determine its molecular formula. Simply list the element symbols, with a numerical subscript if there is more than one atom of that element, in the order of the name (we do not use the subscript 1 if there is only one atom of the element present; 1 is implied). From the name nitrogen trichloride, you should be able to get NCl3 as the formula for this molecule. From the name diphosphorus pentoxide, you should be able to get the formula P2O5 (note the numerical prefix on the first element, indicating there is more than one atom of phosphorus in the formula).
Give the formula for each molecule.
- carbon tetrachloride
- silicon dioxide
- trisilicon tetranitride
Solution
- The name carbon tetrachloride implies one carbon atom and four chlorine atoms, so the formula is CCl4.
- The name silicon dioxide implies one silicon atom and two oxygen atoms, so the formula is SiO2.
- We have a name that has numerical prefixes on both elements. Tri- means three, and tetra- means four, so the formula of this compound is Si3N4.
Give the formula for each molecule.
- disulfur difluoride
- iodine pentabromide
- Answer a
-
\(\ce{S2F2 }\)
- Answer b
-
\(\ce{IBr5}\)
Some simple molecules have common names that we use as part of the formal system of chemical nomenclature. For example, H2O is given the name water, not dihydrogen monoxide. NH3 is called ammonia, while CH4 is called methane. We will occasionally see other molecules that have common names; we will point them out as they occur.
Key Takeaways
- Molecules are groups of atoms that behave as a single unit.
- Some elements exist as molecules: hydrogen, oxygen, sulfur, and so forth.
- There are rules that can express a unique name for any given molecule, and a unique formula for any given name.


