Group 1 Compounds
- Page ID
- 3672
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page discusses a few compounds of the Group 1 elements (lithium, sodium, potassium, rubidium and cesium), including some information about the nitrates, carbonates, hydrogen carbonates and hydrides of the metals.
The effect of heat on Group 1 compounds
Group 1 compounds are more resistant to heat than the corresponding compounds in Group 2. Lithium compounds often behave similarly to Group 2 compounds, but the rest of Group 1 act differently in various ways.
Heating the nitrates
Most nitrates tend to decompose on heating to the metal oxide, brown fumes of nitrogen dioxide, and oxygen. For example, a typical Group 2 nitrate like magnesium nitrate decomposes this way:
\[ 2Mg(NO_3)_2 (s) \rightarrow 2MgO(s) + 4NO_2 (g) + O_2 (g) \nonumber \]
In Group 1, lithium nitrate behaves in the same way, producing lithium oxide, nitrogen dioxide, and oxygen as shown:
\[ 4LiNO_3 (s) \rightarrow 2Li_2O (s) + 4NO_2 (g) + O_2 (g) \nonumber \]
The other Group 1 nitrates, however, do not decompose completely at regular laboratory temperatures. They produce the metal nitrite and oxygen, but no nitrogen dioxide:
\[ 2XNO_3 (s) \rightarrow 2XNO_2(s) + O_2 (g) \nonumber \]
Each of the nitrates from sodium to cesium decomposes in this way; the only difference is in the temperature required for the reaction to proceed. For larger metals, the decomposition is more difficult and requires higher temperatures.
Heating the carbonates
Most carbonates decompose on heating to the metal oxide and carbon dioxide. For example, a typical Group 2 carbonate like calcium carbonate decomposes like this:
\[ CaCO_3 (s) \rightarrow CaO(s) + CO_2 \nonumber \]
In Group 1, lithium carbonate behaves in the same way, producing lithium oxide and carbon dioxide:
\[ Li_2CO_3 (s) \rightarrow Li_2O(s) + CO_2 \nonumber \]
The rest of the Group 1 carbonates do not decompose at laboratory temperatures, although at higher temperatures this becomes possible. The decomposition temperatures again increase down the Group.
The thermal stability of the hydrogen carbonates
The Group 2 hydrogen carbonates such as calcium hydrogen carbonate are so unstable to heat that they only exist in solution. Any attempt to extract them from solution causes them to decompose to the carbonate, carbon dioxide and water as shown:
\[ Ca(HCO_3)_2 (aq) \rightarrow CaCO_3 (s) + CO_2 (g) + H_2O (l) \nonumber \]
By contrast, the Group 1 hydrogen carbonates are stable enough to exist as solids, although they do decompose easily on heating. For example, this is the reaction for sodium hydrogen carbonate:
\[ 2NaHCO_3 (s) \rightarrow Na_2CO_3 (s) + CO_2 (g) + H_2O (l) \nonumber \]
Explaining the trends in thermal stability
Detailed explanations are given for the carbonates because the diagrams are easier to draw. Exactly the same arguments apply to the nitrates or hydrogen carbonates. There are two ways of explaining the increase in thermal stability down the Group. The hard way is in terms of the energetics of the process; the simple way is in terms of the polarizing ability of the positive ions.
Explaining the trend in terms of the polarizing ability of the positive ion
A small positive ion has a large amount of charge packed into a small volume of space; this is especially true if it has a charge greater than +1. An ion with a high charge density has a marked distorting effect on any negative ions which happen to be nearby. A larger positive ion has the same charge spread over a larger volume of space. Its charge density is therefore lower, and it causes less distortion to nearby negative ions.
The structure of the carbonate ion
The molecular structure of carbonate is given below:
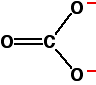
This figure shows two carbon-oxygen single bonds and one double bond, with two oxygen atoms each carrying a negative charge. However, experimental data shows that all the carbonate bonds are identical, with the charge spread out over the whole ion (concentrated on the oxygen atoms). In other words, the charges are delocalized.
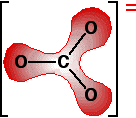
This is a more complicated version of the bonding in benzene or in ions like ethanoate. The next diagram shows the delocalized electrons. The shading shows electron density, implying a greater chance of finding electrons around the oxygen atoms than near the carbon.
Polarizing the carbonate ion
Imagine that this ion is placed next to a positive ion. The positive ion attracts the delocalized electrons in the carbonate ion towards itself. The carbonate ion becomes polarized. The diagram shows what happens with an ion from Group 2, carrying two positive charges:
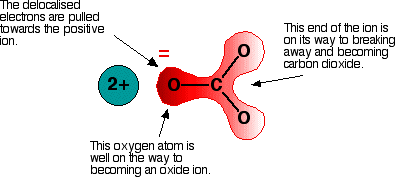
If this system is heated, the carbon dioxide breaks free, leaving a metal oxide. The amount of heat required depends on how polarized the ion was. If it is highly polarized, less heat is required than if it is only slightly polarized. If the positive ion only has one positive charge, the polarizing effect is lessened. This is why the Group 1 compounds are more thermally stable than those in Group 2. The Group 1 compound must be heated more because the carbonate ion is less polarized by a singly-charged positive ion.
The smaller the positive ion, the higher the charge density, and the greater the effect on the carbonate ion. As the positive ions get bigger down the group, they have less effect on the carbonate ions near them. To compensate, the compound must be heated more in order to force the carbon dioxide to break off and leave the metal oxide.
In other words, carbonates become more thermally stable down the group.
Extension to nitrates and hydrogen carbonates
The polarization argument exactly the same for these compounds. The small positive ions at the top of the Group polarize the nitrate or hydrogen carbonate ions to a greater extent than the larger positive ions at the bottom. Again, the Group 1 compounds need more heat than those in Group 2 because the Group 1 ions are less polarizing.
The solubility of Group 1 compounds
Group 1 compounds are more soluble than the corresponding ones in Group 2.
The carbonates
Group 2 carbonates are virtually insoluble in water. Magnesium carbonate (the most soluble Group 2 carbonate) has a solubility of about 0.02 g per 100 g of water at room temperature. By contrast, the least soluble Group 1 carbonate is lithium carbonate. A saturated solution has a concentration of about 1.3 g per 100 g of water at 20°C. The other carbonates in the group are very soluble, with solubilities increasing to an astonishing 261.5 g per 100 g of water at this temperature for cesium carbonate.
Solubility of the carbonates increases down Group 1.
The hydroxides
The least soluble hydroxide in Group 1 is lithium hydroxide, but it is still possible to make a solution with a concentration of 12.8 g per 100 g of water at 20°C. The other hydroxides in the group are even more soluble. Solubility of the hydroxides increases down Group 1. In Group 2, the most soluble is barium hydroxide—it is only possible to make a solution of concentration around 3.9 g per 100 g of water at the same temperature of 20°C .
It is difficult to explain the trends in solubility. The discussion on Group 2 of the periodic table explains why the usual explanations for these trends are not accurate.
The Group 1 hydrides
Saline (salt-like) hydrides
Group 1 metal hydrides are white crystalline solids; each contains the metal ion and a hydride ion, H-. They have the same crystal structure as sodium chloride, which is why they are called saline or salt-like hydrides. Because they can react violently with water or moist air, they are normally supplied as suspensions in mineral oil.
Preparation of the Group 1 hydrides
Group 1 hydrides are made by passing hydrogen gas over the heated metal. For example, for lithium hydride:
\[2Li + H_2 \rightarrow 2LiH \nonumber \]
Reactions of the Group 1 hydrides
Two of the most common reactions include electrolysis and reactions with water.
Electrolysis
On heating, most of these hydrides decompose into the metal and hydrogen before they melt. It is, however, possible to melt lithium hydride and to electrolyze the melt. The metal is deposited at the cathode as expected. Hydrogen is given off at the anode (the positive electrode); this is convincing evidence for the presence of the negative hydride ion in lithium hydride.
The anode equation is:
\[ 2H^- \rightarrow H_2 + 2e^- \nonumber \]
The other Group 1 hydrides can be electrolyzed in solution in various molten mixtures such as a mixture of lithium chloride and potassium chloride. These mixtures melt at lower temperatures than the pure chlorides.
Reaction with water
Group 1 hydrides react violently with water releasing hydrogen gas and producing aqueous metal hydroxide. For example, sodium hydride reacts with water to produce sodium hydroxide and hydrogen gas:
\[ NaH + H_2O \rightarrow NaOH + H_2 \nonumber \]
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


