6.11E: Structure - Zinc Blende (ZnS)
- Page ID
- 2592
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)ZnS has a unique structure type compared to other molecules, having different types of unique structures. ZnS can have a zinc blende structure which is a "diamond-type network" and at a different temperature, ZnS can become the wurtzite structure type which has a hexagonal type symmetry. Structure-wise, the zinc blende structure is more thermodynamically favored, however, because of the wurtzite structures slow construction, both forms of ZnS can be found.
Summary: Zinc blend is a compound that comes in two forms: sphalerite and wurtzite. These are characterized by a 1:1 stoichiometric ratio of Zinc to Sulfur. It maintains a tetrahedral arrangement in both forms.
Introduction
Zinc sulfide (ZnS) is a unique compound that forms two types of crystalline structures. These two polymorphs are wurtzite and zincblende (also known as sphalerite). Wurtzite has a hexagonal structure, while zincblende is cubic. It is characterized by single bonds between each atom and maintenance of a 1:1 zinc to sulfur ratio.
sphalerite:
| Site | Zn | S |
|---|---|---|
| Central | 4 | 0 |
| Face | 0 | 6(1/2) = 3 |
| Corner | 0 | 8(1/8) = 1 |
| Total | 4 | 4 |
Since the number of atoms in a single unit cell of Zn and S is the same, it is consistent with the formula ZnS.
The ionic radius for Zn2+ is 74pm and for S2- is 190pm. Therefore the ratio between cationic and anionic radii in zinc blend is 0.39 (74pm/190 pm) .This suggests a tetrahedral ion arrangement and four nearest neighbors from standard crystal structure prediction tables. Therefore, four sulfur atoms surround each zinc atom and four zinc atoms surround each sulfur atom.2 The coordination number, the number of of electron pairs donated to a metal by its ligands, for both zinc an sulfur is four.1,2 The difference between wurtzite and zincblende lies in the different arrangements of layers of ions.2
Zincblende (Sphalerite)
Zincblende is characterized as a cubic closet packing (ccp), also known as face-centered cubic, structure.1,4 This crystal lattice structure is shown in Figures 1 & 2 below.
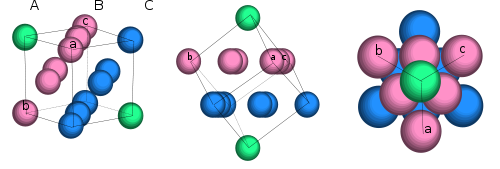
Fig. 1. A break down of cubic closest packing. (Author: Maghémite Date: May 5, 2008. Licensed under the Creative Commons Attribution-Share Alike 3.0 Unported, 2.5 Generic, 2.0 Generic and 1.0 Generic license.)
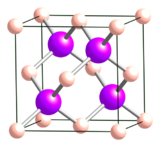
Fig. 2. A representation of ccp structure. (from Public Domain)
Notice how only half of the tetrahedral sites are occupied.
Thermal stability
Density tends to decrease as temperature increases. In this case, since ccp structures are more dense than hcp structures, so a conversion from sphalerite to wurtzite occurs naturally over time at a rate similar to that of diamond to graphite. The sphalerite structure is favored at 298k by 13kJ/mol, but at 1296K the transition to wurtzite occurs.3
Wurtzite
Wurtzite has a hexagonal closest packing structure (hcp), which is characterized by 12 ions in the corners of each unit that create a hexagonal prism (seen in Fig. 3).2 As discussed previously, zincblende slowly transforms to wurtzite due to thermodynamic stability.
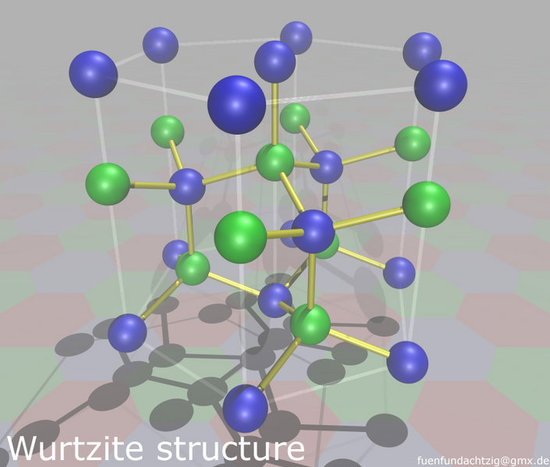
Fig 3. HCP structure of wurtzite. (Creator: Alexander Mann Date: 01/14/2006 Licensed under the Creative Commons Attribution-Share Alike 2.0 Germany license)
Calculating density of a crystal structure
Density = Mass of unit cell / volume of unit cell.
where:
Mass of unit cell = Number of atoms in a unit cell x the mass of each atom
volume of unit cell = a3 x 10-30
Contributors and Attributions
- Emma Mele, UC Davis Animal Biology

