12.2.2: Inert and Labile Complexes
- Page ID
- 385506
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Kinetics of Substitution Reactions
Kinetics is a branch of chemistry that is concerned with the rates of chemical reactions. In this section, we will discuss the rates of metal-ligand (M-L) substitution reactions.
Let's start with some examples. In the table below are three examples of ligand substitution reactions of hexaaquo metal complexes to form hexaammine complexes. These reactions are nearly identical with the exception of the metal ion. The products are thermodynamically favored in all cases.
| Reaction | Rate constant (\(k\)) | Labile or Inert |
|---|---|---|
| \(\ce{[Ni(OH2)]^{2+} + 6 NH3 <=>> [Ni(NH3)6]^{2+}} \quad\) | \(k = 10^{4} s^{-1}\) (1 ns) | Labile (happens in < 1 min) |
| \(\ce{[Cr(OH2)]^{3+} + 6 NH3 <=>> [Cr(NH3)6]^{3+}} \quad\) | \(k = 10^{-3} s^{-1}\) (6 days) | Inert (slow, takes hours) |
| \(\ce{[Cu(OH2)]^{2+} + 6 NH3 <=>> [Cu(NH3)6]^{2+}}\quad \) | \(k = 10^{8} s^{-1}\) | Very Labile (happens in seconds) |
- Kinetically Labile - Metal complexes that undergo "kinetically fast" substitution reactions are kinetically labile. Taube suggested that these are reactions in which half of the reactant is consumed in one minute or less (\(t_{1/2}\) <1 min).
- Kinetically Inert - Metal complexes that undergo "kinetically slow" substitution reactions are kinetically inert or non-labile. Taube suggested that these are reactions in which \(t_{1/2}\) >1 min.
A common pitfall is to confuse the meaning of kinetic terms, like labile and inert, with thermodynamic terms, like stable and unstable. It is important to distinguish between kinetics and thermodynamics. For example, the complex \([Fe(H_2O)Cl]^{2+}\) has a large formation constant and is thermodynamically stable; yet it is also labile. On the other hand, the complex \([Co(NH_3)_6]^{3+}\) is unstable in acidic aqueous solution and decomposes spontaneously to \([Co(H_2O)_6]^{3+}\); yet it decomposes slowly because it is inert. It is good practice to use clear terms such as "kinetically labile" or "kinetically inert" and "thermodynamically stable" and "thermodynamically unstable".
Draw the reaction coordinate diagrams for a reaction of the form \([ML_6]^{n+} + X \rightleftharpoons [ML_5X]^{n+} + L\) in the following scenarios:
- \([ML_6]^{n+}\) is theromdynamically stable and kinetically inert.
- \([ML_6]^{n+}\) is theromdynamically unstable and kinetically inert.
- \([ML_6]^{n+}\) is theromdynamically stable and kinetically labile.
- \([ML_6]^{n+}\) is theromdynamically unstable and kinetically labile.
- Answer a)
-
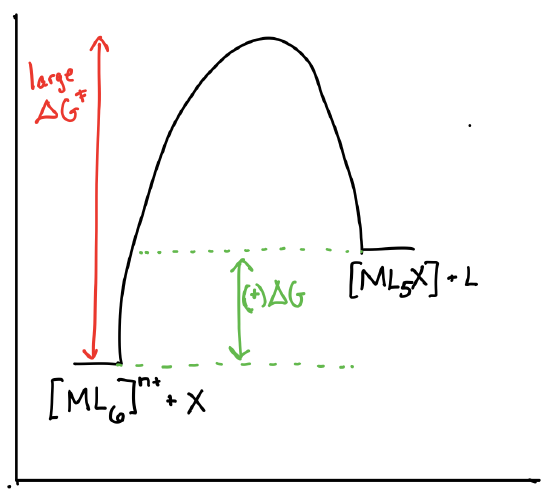
- Answer b)
-
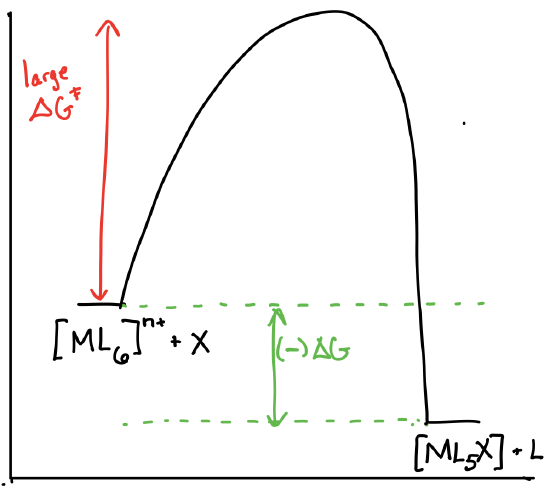
- Answer c)
-
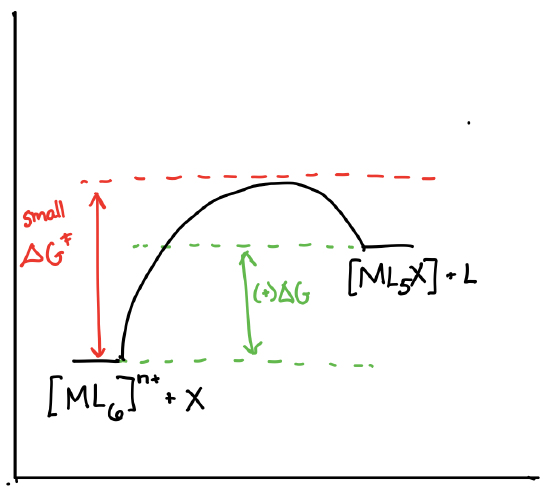
- Answer d)
-

Factors that affect rates of substitution reactions:
Some of the factors that affect the kinetic rates of ligand substitution are the same factors that affect thermodynamic stability (see Chapter 10). The same factors that make a complex stable can also make it more inert. Why are kinetic factors related to thermodynamic stability? It is because the structure and stability of the reactant complex is related to the structure and stability of the transition state. The reactant complex must change its geometry to form an intermediate or transition state. When the reactant is particularly stable, it can result in a higher activation energy associated with moving away from the stable configuration. However, it is incorrect to assume that stability is always correlated with reaction rates. Kinetic and thermodynamic factors are related, yet separate.
There are three important factors that influence kinetic rate of substitution:
- Ligand Field Stabilization Energy (LFSE): Electron configurations that place electrons in higher-energy orbitals (particularly antibonding orbitals) result in more labile complexes. As long as there are not electrons in higher-energy orbitals, the lability correlates roughly with LFSE. The more negative the LFSE, the more inert.
- Coulombic attraction between the metal and ligand: In general, higher charge density on the metal ion or on the ligand(s) leads to stronger electrostatic attraction between metal and ligand. Stronger Coulombic attraction generally leads to slower dissociation steps and faster association steps. The effect that these have on the reaction rate depends on elementary steps that take part in the rate-determine step(s).
- Denticity: Multi-dentate ligands create particularly inert complexes as a result of the kinetic chelate effect.
All three considerations are described in more detail below.
In which compound from each pair would you expect the strongest ionic bonds? Why?
a) LiF vs KBr
b) CaCl2 vs. KCl
- Answer a
-
The ions in LiF are both smaller than in KBr, so the force of attraction between the ions in LiF is greater because of the smaller separation between the charges.
- Answer b
-
Calcium has a 2+ charge in CaCl2, whereas potassium has only a + charge, so the chloride ions are more strongly attracted to the calcium than to the potassium.
Taube's observations of metal complex substitution rates
Henry Taube (Nobel Prize, 1983) tried to understand kinetic lability by comparing the factors that govern bond strengths to observations about the rates of reaction of coordination complexes. He saw some things that were unsurprising. He also drew some new conclusions based on ligand field theory.
Taube observed that many M+1 ions (M = metal) are more labile than many M+3 ions, in general. That isn't too surprising, since metal ions function as electrophiles (Lewis acids) and ligands function as nucleophiles (Lewis bases) in forming coordination complexes. In other words, metals with higher charges ought to be stronger Lewis acids, and so they should bind ligands more tightly. However, there were exceptions to that general rule. For example, Taube also observed that \(\ce{Mo+5}\) compounds are more labile than \(\ce{Mo+3}\) compounds. So, there must be more going on here than just the effects of electrostatic attraction.
Another factor that governs ionic bond strengths is the size of the ion. Typically, ions with smaller atomic radii form stronger bonds than ions with larger radii. Taube observed that Al3+, V3+, Fe3+ and Ga3+ ions are all about the same size. All these ions exchange ligands at about the same rate. That isn't surprising, because they have the same charge and the same radius. However, Cr3+ is also about the same size as those ions and it also has the same charge, but it is much less labile. Once again, there are exceptions to our regular expectations based on simple electrostatic considerations. Furthermore, 4d and 5d transition metals (Y\(\rightarrow\)Cd, and Ac\(\rightarrow\)Hg) are much more inert than 3d transition metals (Sc\(\rightarrow\)Zn). This is unexpected when we consider size; the 4d and 5d metals are much larger than the 3d metals. This unexpected behavior tells us that electrostatics alone cannot predict lability.
Taube came up with a hypothesis that could explain the seeming contradictory oservations described above: kinetic lability must be affected by d-electron configuration. This idea forms the basis of Taube's rules about lability.
For example, metals like Ni2+ and Cu2+ are very labile. The d orbital splitting diagrams for those compounds would have d electrons in the eg set. Remember, the eg set arises from interaction with the ligand donor orbitals; this set corresponds to a \(\sigma\) antibonding level.
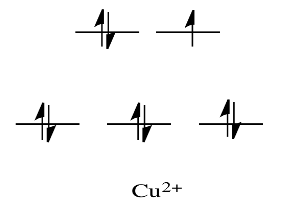
By comparison, V2+ is rather inert. The d orbital splitting diagram in this case has electrons in the t2g set, but none in the eg set.
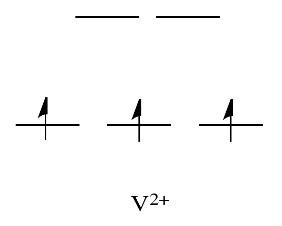
So, having electrons in the higher energy, antibonding eg level weakens the bond to the ligand, so the ligand can be replaced more easily. In the absence of those higher energy electrons, the bond to the ligand is stronger, and the ligand isn't replaced as easily.
On the other hand, metals like Ca2+, Sc3+ and Ti4+ are pretty labile. The d orbital splitting diagrams in those cases are pretty simple: there are no d-electrons at all in these ions.
That means having no electrons in these mostly non-bonding levels leaves the complex susceptible to ligand replacement. But it's hard to see why population of an orbital that is mostly non-bonding would have an effect on ligand bond strength.
Instead, this factor probably has something to do with the part of ligand substitution that we have ignored so far. Not only does one ligand need to leave, but a second one needs to bond in its place. So, having an empty orbital for the ligand to donate electrons into (or, put another way, not having electrons in the way that may complicate donation from the ligand) makes that part of the reaction easier.
Consider all possible electron configurations for octahedral complexes (\(d^0\) to \(d^{10}\), high spin and low spin cases): predict whether each case would be inert, intermediate, or labile.
- Answer
-
Kinetically Inert (Slow) Kinetically Labile (Fast) \(d^n\) electron count and ligand field strength Octahedral configurations with empty \(e_g\) orbitals:
- \(d^3\)
- low-spin \(d^4, d^5, d^6\)
Strong-field \(d^8\) (square planar)
Octahedral configurations with occupied \(e_g\) orbitals:
- \(d^1,d^2, d^7,d^9,d^10\)
- high-spin \(d^4,d^5, d^6\)
Weak-field \(d^8\) (usually tetrahedral)
Put the metal ions in order of decreasing reaction rate (from labile to inert):
a) \(Al^{3+}, Na^+, Mg^{2+}\)
b) \(Ca^{2+}, Mg^{2+}, Sr^{2+} \)
- Answer (a)
-
Most Labile to most inert: \(Na^+ > Mg^{2+} > Al^{3+}\)
These are metal ions with similar size and varying charge. They are in order of increasing charge and increasing density from left to right.
- Answer (b)
-
Most labile to most inert: \(Sr^{2+} > Ca^{2+} > Mg^{2+}\)
These metal ions have the same charge, and vary in size. They are in order of decreasing charge and increasing charge density from left to right.
Some metals, like Mn2+, can be either labile or inert, depending on whether they are high spin or low spin. Explain why using d orbital splitting diagrams.
- Answer
-
Add texts here. Do not delete this text first.
Predict whether the following metals, in octahedral complexes, are labile or not.
a) Co3+ (high spin)
b) Co3+ (low spin)
c) Fe2+ (low spin)
d) Fe2+ (high spin)
e) Zn2+
- Answer
- Answer a
-
labile (electrons in higher energy d orbital set)
- Answer b
-
not labile (all electrons in lower energy d orbitals)
- Answer c
-
not labile (all electrons in lower energy d orbitals)
- Answer d
-
labile (electrons in higher energy d orbital set)
- Answer e
-
labile (electrons in higher energy d orbital set)
Generalizations for how metal ion affects kinetics:
These are some generalizations about how the kinetics of substitution is affected by the metal ion identity and the \(d^n\) electron configuration.
- s-block metals are very labile, except for those with very high charge density (eg. \(Mg^{2+}\ is\: inert\))
- \(d^{10}\) metals are labile (eg: \(Zn^{2+}, Cu^+, Hg^{2+}\))
- Other ions with a full shell are labile (eg: \(Ln^{3+}\) of f-block)
- 3d \(M^{2+}\), when high spin, are generally labile (eg. \(Cu^{2+} is\: very\: labile\))
- 4d and 5d are usually inert due to higher LFSE (low spin, high LFSE)
- \(M^{2+}\) is more labile than the same metal as \(M^{3+}\)
- \(d^3\) and low spin \(d^6\) are inert (eg. \(Cr^{3+}, Co^{3+}, \) low spin \(Fe^{2+}\))
Chelate Complexes
In addition to considering factors related to the metal ion and charge or covalent character of metal-ligand bonds, an important consideration is the effect a chelate ligand on reaction kinetics. This effect is also discussed in more detail later in this chapter (Section 12.4.5).
Recall the Thermodynamic Chelate Effect
Chelating complexes tend to be more stable than complexes with monodentate ligands. This is called the “thermodynamic chelate effect”. The effect deserves an explanation. The explanation is the increase of entropy that occurs when two or more monodentate ligands are replaced by a chelating ligand. The entropy increases because the overall number of particles increases as the substitution takes place.
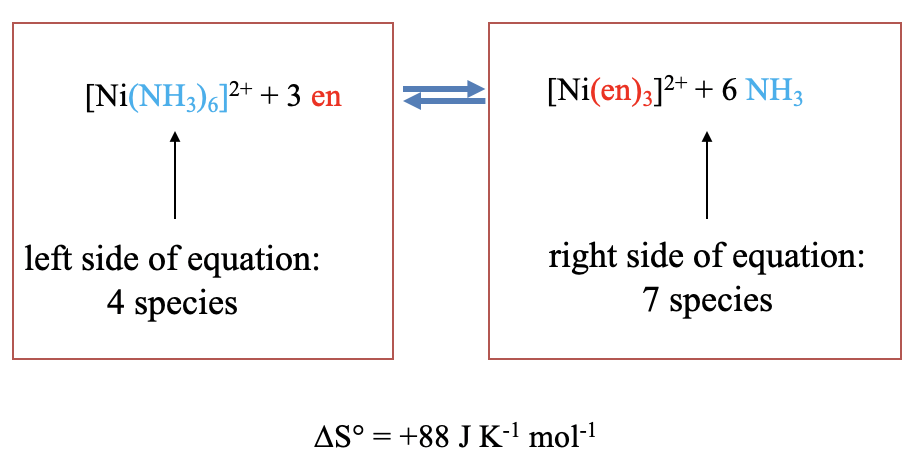
For example, the substitution of six ammine ligands in the hexaammine nickel (2+) complex by three ethylenediamine chelating ligands increases the number of molecules from four to seven, and hence the entropy increases, in this case by 88 J K-1 and mol-1 (Figure \(\PageIndex{3}\))
The Kinetic Chelate Effect
In addition to the thermodynamic chelate effect, there is the kinetic chelate effect. Chelate complexes are frequently more inert than complexes with monodentate ligands. Chelate complexes are more inert for two reasons (Figure \(\PageIndex{4}\)).
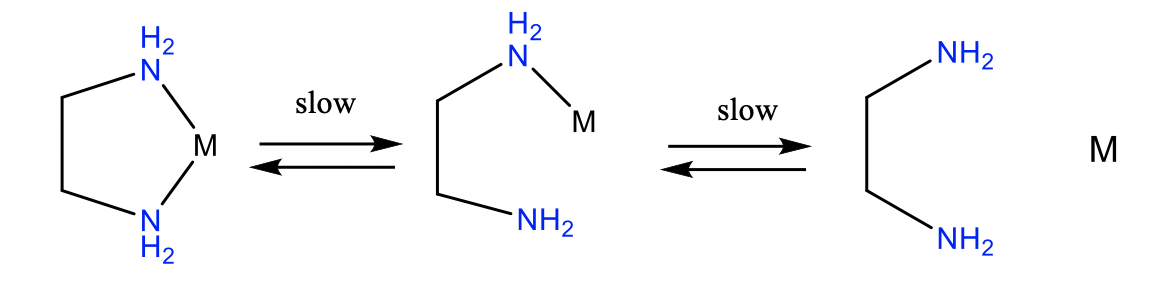
Firstly, the whole ligands needs to rotate and bend in order to cleave the first metal-ligand bond. This requires time and slows the kinetics of the bond cleavage. The second reason is that the detached donor atom cannot leave the proximity of the complex because the ligand is still attached via the other donor atom. This increases the probability of the re-formation of the metal-ligand bond which decreases the probability of both bonds being cleaved.
Chris P Schaller, Ph.D., (College of Saint Benedict / Saint John's University)
Curated or created by Kathryn Haas
Dr. Kai Landskron (Lehigh University). If you like this textbook, please consider to make a donation to support the author's research at Lehigh University: Click Here to Donate.


