6.5.1: Host-Guest Chemistry and π-π Stacking Interactions
- Page ID
- 162915
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This section sidesteps the discussion of topics related to acid-base chemistry and indulges in a brief aside about one type of intermolecular interaction that is used in host-guest chemistry. This section will first describe the concept of host-guest chemistry, then outline the types of interactions and design principles that can be used to promote host-guest interactions, and finally conclude with a discussion of \(\pi-\pi\) interactions.
In Host-Guest Chemistry a large molecule or network material noncovalently binds a smaller molecule in a binding pocket.
In host-guest chemistry, a large molecule or network host uses noncovalent interactions to bind a smaller molecule guest in a binding pocket. Schematically this interaction is often represented as:
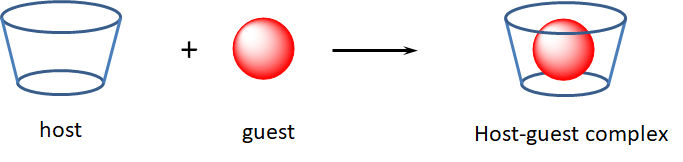
Host-guest binding is analogous to the way many biomolecule-substrate interactions occur - e.g., to how antibodies bind antigens and many enzymes bind their substrates. In this way many biomolecular systems can be said to form host-guest complexes, although that terminology is not often used in the biochemistry field. Usually when someone refers to a host-guest complex in inorganic or organic chemistry they are referring to organic or metal-organic cages that bind small organics. A simple example involves the binding of buckminsterfullerene, C60,, by calix-[5]-arenes.
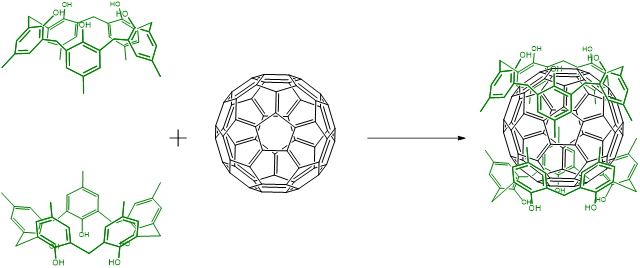
In principle any type of intermolecular interaction can hold a host-guest complex together, but whichever one you choose, it helps to pay attention to a few design principles.
The types of interactions that are used to effect host-guest binding are diverse. In the fullerene-calixarene example above, the interactions will primarily involve \(\pi\) stacking interactions between the aromatic rings of the calixarenes and those in the fullerene. Other host-guest systems use some combination of:
- hydrogen bonds
- halogen bonds
- ionic interactions whereby charged groups from the host attract oppositely charged groups of the guest
- ion-dipole interactions whereby charged groups on one component interact with the appropriate end of a polar group on the other.
- dipole-dipole interactions
- cation-\(\pi\), X-H-\(\pi\), and similar interactions in which full or partial positive charges induce a complementary dipole in a \(\pi\) system.
- \(\pi - \pi\) interactions
- dispersion forces
Many effective host-guest systems employ a few design principles to favor host-guest binding. These principles include:
- host preorganization - when conformationally flexible (-i.e., floppy) hosts bind a guest, the host will conform to the shape of the guest and so become more rigid. In other words, when a floppy host binds to a guest, a lot of conformational entropy is lost. This will make the \(\Delta S\) term for the binding free energy more negative and tends to disfavor host-guest binding. To avoid this, effective hosts minimize the loss of conformational entropy on binding. This is usually done by limiting the conformational entropy of the host to begin with by fusing multiple binding sites into a macrocyclic ring, cage, or cluster. The more rigid these are the better.
- host-guest size and shape complementarity - this simply means that complexes form when the binding pocket or cavity of the host is complementary to that of the guest. Specifically, its size and shape both fit the guest and allow for tight interactions (i.e., the guest doesn't rattle around inside). Moreover, any charged, polar, or any-directional binding sites on the host are arranged in such a way that they interact favorably with complementary sites on the guest.
- preferencing of guest desolvation via preferential solvent-solvent interactions* - \(\pi - \pi\) interactions like those which hold the calixarene-fullerene complex together are weak compared to stronger interactions like hydrogen bonds or even water-\(\pi\) bonding (a type of X-H \(\pi\) bonding. However, water-water hydrogen bonds and other interactions between H-bond-capable and polar systems are stronger still. This means that favorable water-water interactions must be broken when a nonpolar host or guest is dissolved in a very polar or ionic solution. Every dipole-induced dipole or X-H \(\pi\) interaction that takes place is a lost opportunity for solute molecules to engage in hydrogen bonding interactions. Thus, when an aqueous solution of a nonpolar host and guest are mixed, the nonpolar parts of these molecules will tend to stick together, freeing water or other polar molecules to interact with one another as they do so.**
\(\pi - \pi\) interactions occur when aromatic \(\pi\) systems bind face to face with one another and involve a combination of dispersion and dipole-induced dipole interactions.
Since the electron distribution in aromatic systems is relatively easily distorted, they can engage in atypically strong dispersion and dipole-induced dipole interactions called \(\pi - \pi\) stacking interactions. These interactions are so named because they occur when the planes of aromatic rings are stacked parallel to one another. This parallel stacking can occur in either a sandwich or a displaced stacking arrangement.
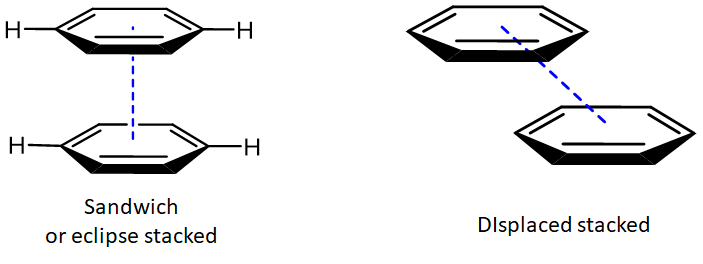
In either case the rings can be attracted to one another by a combination of dispersion and electrostatic interactions. The latter depend on the specific bonding parameters of the system and can be attractive or repulsive. The eclipsed arrangement of the benzene dimer is electrostatically disfavored, while attractions between the positive hydrogen substituents and the \(\pi\) system of an adjacent ring are attractive in the eclipsed arrangement.
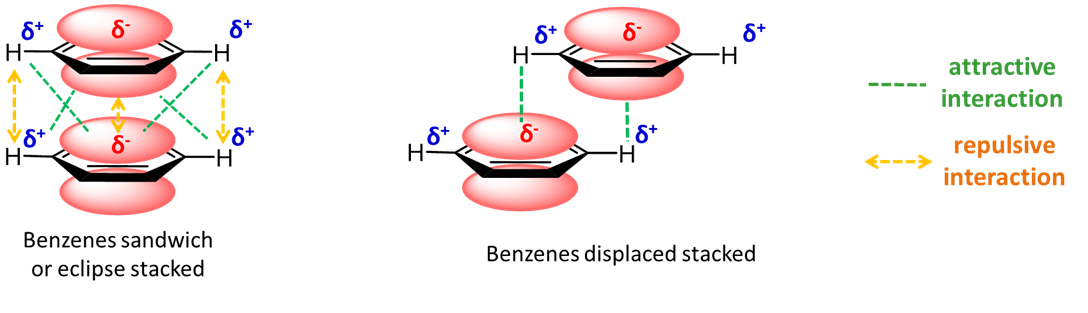
With electronegative substituents, the polarity of the ring system is reversed so that there are favorable electrostatic interactions in an eclipsed heterodimer of benzene and hexafluorobenzene.
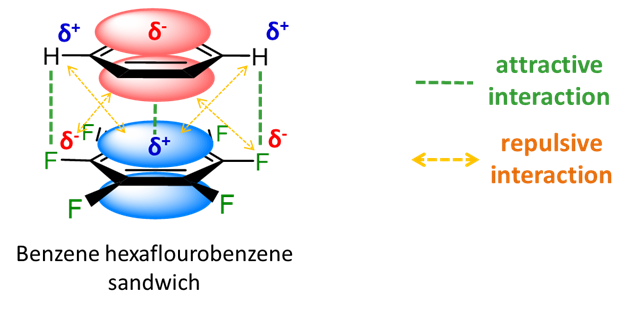
In the heterodimer of benzene and hexafluorobenzene, at last we have an example of a \(\pi\) stacking system that can more readily be described as involving a Lewis acid-base interaction. Specifically, the relatively electron-rich benzene \(\pi\) system acts as a Lewis base in donating "electron pairs" to the electron-poor hexafluorobenzene \(\pi\) system.
Notes
* The most prominent example of desolvation as a driving force is the hydrophobic effect. The hydrophobic effect governs the structure of soaps, micelles, biomolecules, and other amphiphilic or nonpolar systems in water. In the hydrophobic effect, the preference of an aqueous solution of a hydrophobic solute to maximize water-water interactions means that hydrophobic groups tend to stick together in solution.
** A good analogy for this effect is the magnetic marble analogy. When magnetic and ordinary marbles are mixed, the magnetic marbles will tend to stick together because they will attract one another strongly, not because the interactions between the nonmagnetic marbles are somehow disfavored.
† This is not to say that the stacking arrangements lead to attraction in every instance. There are arrangements in which the parallel rings are expected to repel.

