6.4: Boron Hydrides
- Page ID
- 212647
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Borane and diborane
Borane (BH3) formed in the gaseous state from decomposition of other compounds, (6.4.2) but cannot be isolated except as a Lewis acid-base complex (6.4.1). As such many borane adducts are known.
\[ \text{BH}_3 \text{ + PPh}_3 \rightarrow \text{H}_3\text{B-PPh}_3\]
In the absence of a Lewis base the dimeric diborane (B2H6) is formed. Diborane is generally synthesized by the reaction if BF3 with a hydride source, such as NaBH4, (6.4.4), or LiAlH4, (6.4.3).
\[\text{H}_3\text{B-PF}_3 \xrightarrow{\Delta} \text{BH}_3 \text{ + PF}_3\]
\[ \text{3 LiAlH}_4 \text{ + 4 BF}_3 \rightarrow \text{2 B}_2\text{H}_6 \uparrow \text{ + 3 LiAlF}_4\]
The structure of diborane (Figure \(\PageIndex{1}\) a) is considered to be electron deficient, and has been confirmed by IR spectroscopy and electron diffraction. The four terminal B-H bonds are normal covalent bonds, however, the bridging B-H-B unit consists of two three-centered two-electron bonds, each ordinarily considered to be formed by the combination of two boron sp3 orbitals and one hydrogen s orbital (Figure \(\PageIndex{1}\) b). However, a consideration of the H-B-H bond angle associated with the terminal hydrides (120°) it is perhaps better to consider the BH2 fragment to be sp2 hybridized, and the B-H-B bridging unit to be a linear combination of one sp2 orbital and one p orbital from each boron atom with the two hydrogen s orbitals. Diborane represents the archetypal electron deficient dimeric compound, of which Al2Me3 is also a member of this class of electron deficient molecules.
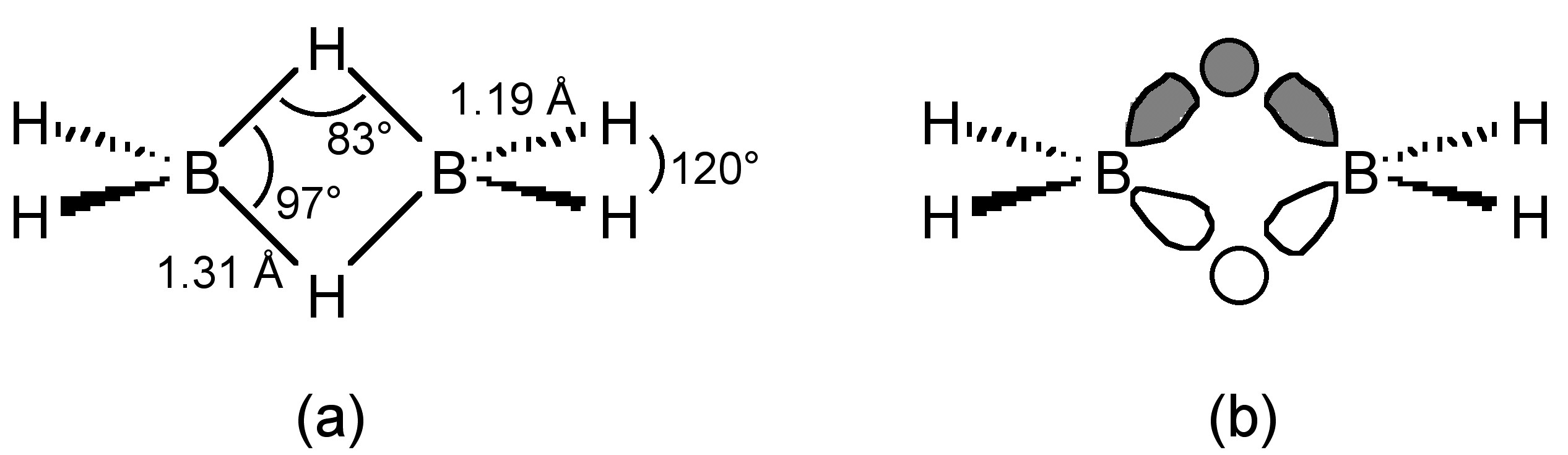
B2H6 is spontaneously and highly exothermically inflammable above 25 °C (ΔH = -2137.7 kJ/mol), (6.4.5). It is often used as one of a range of solvate forms for safety for both its flammability and toxicity.
\[ \text{3 NaBH}_4 \text{ + 4 BF}_4 \rightarrow \text{2 B}_2\text{H}_6 \uparrow \text{ + 3 NaBF}_4 \]
\[ \text{B}_2\text{H}_6 \text{ + 3 O}_2 \rightarrow \text{B}_2\text{O}_3 \text{ + 3 H}_2\text{O}\]
Most reactions of diborane involve the cleavage of the dimeric structure. Hydrolysis of diborane yields boric acid, (6.4.6), while alcoholysis yields the appropriate borate ester, (6.4.7). Diborane reacts with Lewis bases to form the appropriate Lewis acid-base complex, (6.4.8).
\[ \text{B}_2 \text{H}_6 \text{ + 6 H}_2\text{O} \rightarrow \text{2 B(OH)}_3 \text{ + 6 H}_2 \uparrow\]
\[ \text{B}_2 \text{H}_6 \text{ + 6 ROH} \rightarrow \text{2 B(OR)}_3 \text{ + 6 H}_2 \uparrow\]
\[ \text{B}_2 \text{H}_6 \text{ + NR}_3 \rightarrow \text{2 H}_3\text{B-NR}_3 \]
Borohydride
The borohydride anion (or more properly the tetrahydridoborate anion), BH4-, can be considered as the Lewis acid-base complex between borane and H-. A typical synthesis involves the reaction of a borate ester with a hydride source, (6.4.9).
\[\text{4 NaH + B(OMe)}_3 \rightarrow \text{NaBH}_4 \text{ + 3 NaOMe}\]
Sodium borohydride is a stable white crystalline solid that is stable in dry air and is non-volatile. The boron in borohydride (BH4-) is tetrahedral. Although it is insoluble in Et2O, it is soluble in water (in which it reacts slowly), THF, ethylene glycol, and pyridine. Interestingly, NaBH4 reacts rapidly with MeOH, but dissolves in EtOH. Sodium borohydride has extensive uses in organic chemistry as a useful reducing agent in which it donates a hydride (H-).
Higher Boranes
Higher boron hydrides contain, in addition to the bridging B-H-B unit, one or more B-B bonds. The higher boranes are usually formed by the thermal decomposition of diborane, (6.4.10) and (6.4.11).
\[ \text{2 B}_2\text{H}_6 \rightarrow \text{B}_4\text{H}_{10} \text{ + H}_2 \]
\[ \text{5 B}_2\text{H}_6 \rightarrow \text{2 B}_5\text{H}_9 \text{ + 6 H}_2 \]
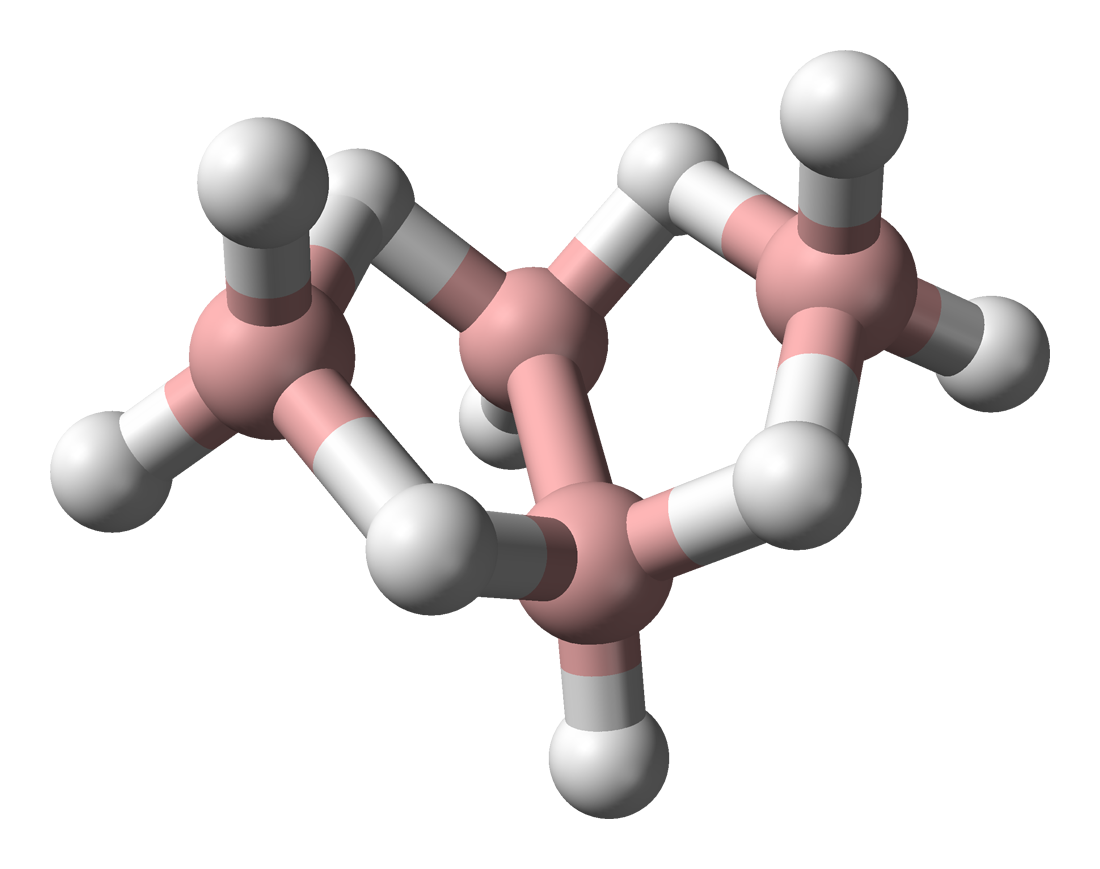
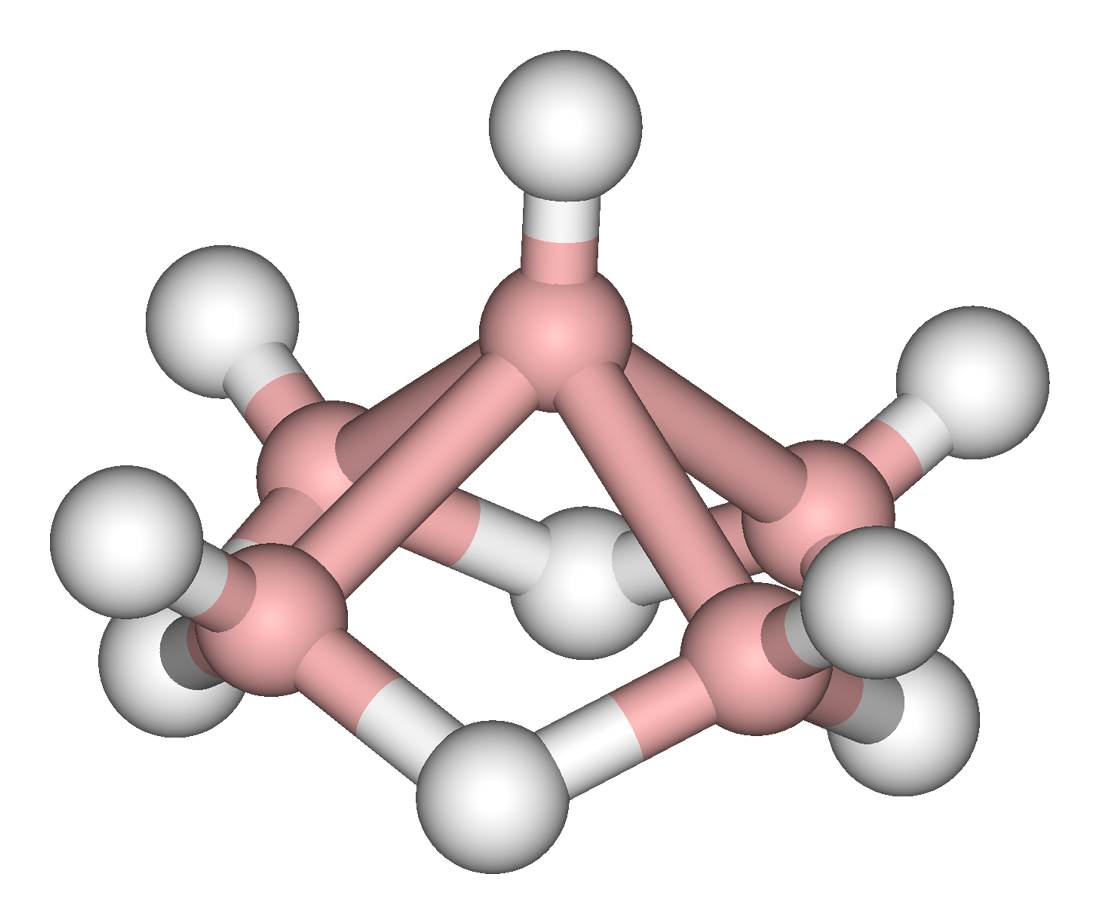
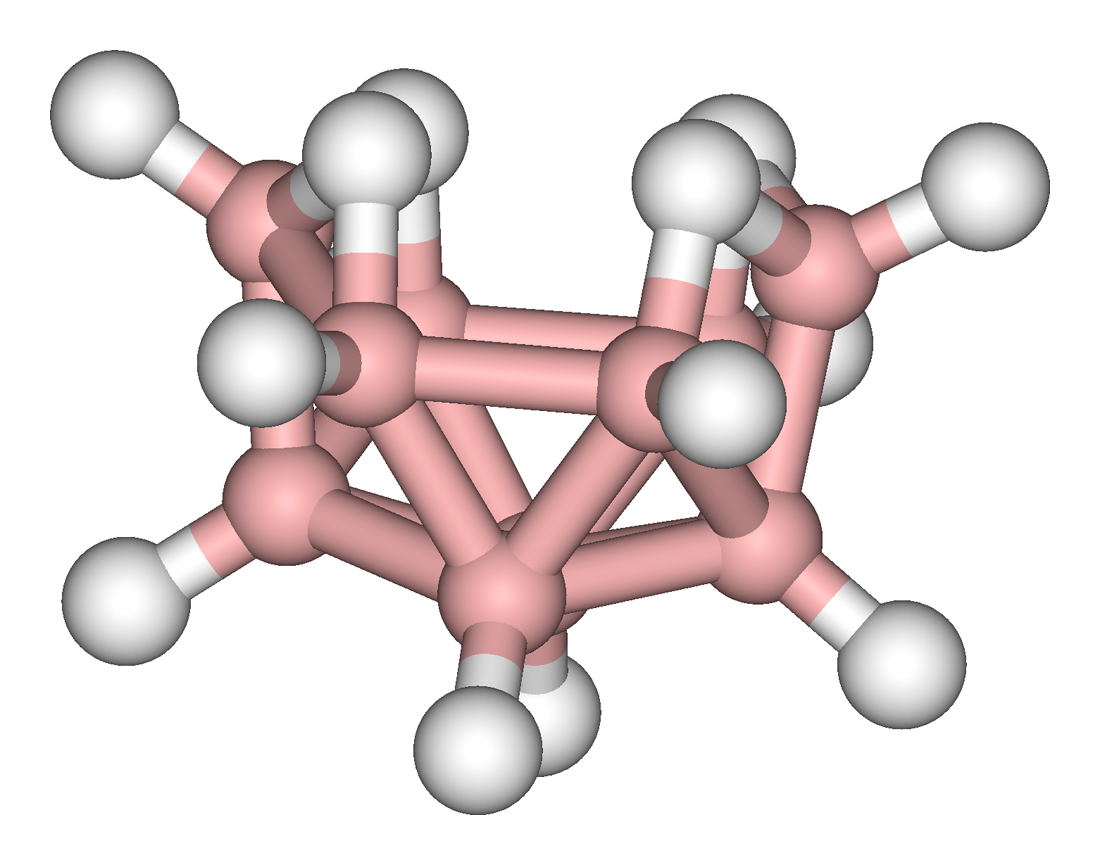
These higher boranes have ‘open’ cluster structures, e.g., Figure \(\PageIndex{2}\) - Figure \(\PageIndex{4}\). Tetraborane, or to be more precise tetraborane(10) or arachno-B4H10, is a foul-smelling toxic gas. Pentaborane (9) is a toxic liquid (with a distinctive garlic odor) that can detonate in air, and like decaborane(14) was at one time considered as a potential rocket fuel. Because simple boron compounds burn with a characteristic green flame, the nickname for these fuels in the US military was Green Dragon. Problems with using boranes as a fuel included their toxicity and the characteristic of bursting into flame on contact with the air; furthermore, the exhaust would also be toxic. The US program resulted in a stockpile of borane fuels, in particular pentaborane(9), which was not destroyed until 2000. The system for the destroying the boranes was appropriately known as Dragon Slayer.
Bibliography
- D. F. Gaines, Acc. Chem. Res., 1973, 6, 416.
- C. E. Housecroft, Boranes and metalloboranes: structure, bonding and reactivity, Ellis Horwood, Chichester (1990).
- C. F. Lane, Chem. Rev., 1976, 76, 773.


