8.1: Climate Change and the Combustion of Fossil Fuels
- Page ID
- 392395
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
 |
Prince George's Community College |
 |
| Unit I: Atoms Unit II: Molecules Unit III: States of Matter Unit IV: Reactions Unit V: Kinetics & Equilibrium Unit VI: Thermo & Electrochemistry Unit VII: Nuclear Chemistry |
||
- To use thermochemical concepts to discuss environmental issues.
Our contemporary society requires the constant expenditure of huge amounts of energy to heat our homes, provide telephone and cable service, transport us from one location to another, provide light when it is dark outside, and run the machinery that manufactures material goods. The United States alone consumes almost 106 kJ per person per day, which is about 100 times the normal required energy content of the human diet. This figure is about 30% of the world’s total energy usage, although only about 5% of the total population of the world lives in the United States.
In contrast, the average energy consumption elsewhere in the world is about 105 kJ per person per day, although actual values vary widely depending on a country’s level of industrialization. In this section, we describe various sources of fossil fuel energy and their impact on the environment.
Driven by environmental concerns about climate change and pollution, the world is undergoing a transformation from fossil fuels to renewable resources such as solar, and wind. The role that hydro and nuclear energy will play is uncertain and especially in the later case a policy rather than a scientific issue.
Fossil Fuels
According to the law of conservation of energy, energy can never actually be “consumed”; it can only be changed from one form to another.
Fossil fuels, coal, oil and natural gas are the result of anaerobic decay of dead plants and animals laid down hundreds of millions of years ago, most of which took place well before the dinosaurs strode the earth. Fossil fuels slowly formed as further geological layers compressed and heated the dead organic matter. The energy content of fossil fuels results from the transformation of sunlight into vegetation and the chemical transformation brought about by anaerobic cooking at high pressures and temperatures over geological times.
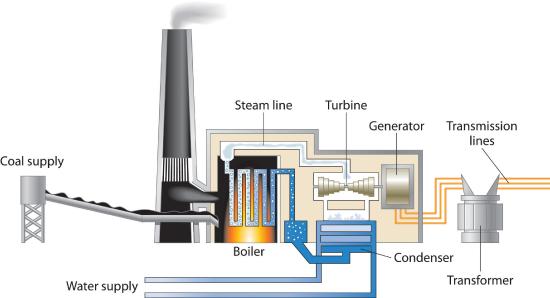
Figure 15.7.1 represents a plant for generating electricity using oil or coal where the fuel is burned in a boiler, superheating steam which then powers a turbine for electrical generation. Oil derived fuels are seldom used in large power plants but diesel is used commonly in small electrical generators either in remote locations or as back up for when electrical distribution systems fail. Natural gas fueled power plants burn the fuel directly in the turbine which is similar to a jet engine. Coal power plants can convert ~40% of the energy released from combustion to electricity. In comparison, nuclear power plants can be more than 50% efficient and gas turbines can approach 60% mostly due to higher operating temperatures. Co-generation, using the plant to produce not only electricity but also heat for industrial or other purposes can raise overall efficiency by 10 - 15 % or so.
Figure 15.7.1 Electricity from Coal A coal-powered electric power plant uses the combustion of coal to produce steam, which drives a turbine to produce electricity.
The total expenditure of energy in the world each year is about 3 × 1017 kJ. Today, more than 80% of this energy is provided by the combustion of fossil fuels: oil, coal, and natural gas (The sources of the energy consumed in the United States in 2009 are shown in Figure 15.7.2.) but as Table 15.7.1 from the Wikipedia shows, energy usage is a complex issue. Petroleum dominates as a source of energy for transportation because gasoline is easy to transport, but is very little used for electrical generation, whereas 91% of coal is used for electrical generation. The other major use of coal is as a reducing agent for metal refining from ores. The former is called thermal coal, the latter metallurgical coal.
Table 15.7.1: Energy useage in the United States for 2008
| Supply Sources | Percent of Source | Demand Sectors | Percent of Sector |
|---|---|---|---|
| Petroleum 37.1% |
71% Transportation 23% Industrial 5% Residential and Commercial 1% Electric Power |
Transportation 27.8% |
95% Petroleum 2% Natural Gas 3% Renewable Energy |
| Natural Gas 23.8% |
3% Transportation 34% Industrial 34% Residential and Commercial 29% Electric Power |
Industrial 20.6% |
42% Petroleum 40% Natural Gas 9% Coal 10% Renewable Energy |
| Coal 22.5% |
8% Industrial <1% Residential and Commercial 91% Electric Power |
Residential and Commercial 10.8% |
16% Petroleum 76% Natural Gas 1% Coal 1% Renewable Energy |
| Renewable Energy 7.3% |
11% Transportation 28% Industrial 10% Residential and Commercial 51% Electric Power |
Electric Power 40.1% |
1% Petroleum 17% Natural Gas 51% Coal 9% Renewable Energy 21% Nuclear Electric Power |
| Nuclear Electric Power 8.5% |
100% Electric Power |
Figure 15.7.2 Energy Consumption in the United States by Source, 2009
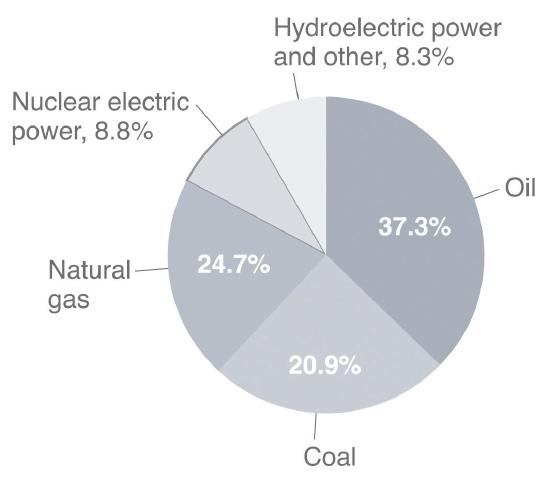
More than 80% of the total energy expended is provided by the combustion of fossil fuels, such as oil, coal, and natural gas.
Coal
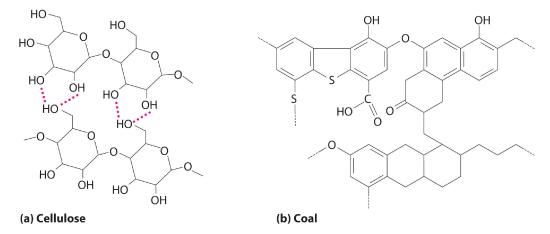
CoalA complex solid material derived primarily from plants that died and were buried hundreds of millions of years ago and were subsequently subjected to high temperatures and pressures. It is used as a fuel. was primarily laid down from the large swamp forests of the Carboniferous Period. Coal deposits are found today where those forests were Coal is a complex solid material derived primarily from plants that died and were buried hundreds of millions of years ago and were subsequently subjected to high temperatures and pressures. Because plants contain large amounts of cellulose, derived from linked glucose units, the structure of coal is more complex than that of petroleum (Figure 15.7.3 ). In particular, coal contains a large number of oxygen atoms that link parts of the structure together, in addition to the basic framework of carbon–carbon bonds. It is impossible to draw a single structure for coal; however, because of the prevalence of rings of carbon atoms (due to the original high cellulose content), coal is more similar to an aromatic hydrocarbon than an aliphatic one.
Figure 15.7.3 The Structures of Cellulose and Coal (a) Cellulose consists of long chains of cyclic glucose molecules linked by hydrogen bonds. (b) When cellulose is subjected to high pressures and temperatures for long periods of time, water is eliminated, and bonds are formed between the rings, eventually producing coal. This drawing shows some of the common structural features of coal; note the presence of many different kinds of ring structures.
There are four distinct classes of coal (Table 15.7.2); their hydrogen and oxygen contents depend on the length of time the coal has been buried and the pressures and temperatures to which it has been subjected. Lignite, with a hydrogen:carbon ratio of about 1.0 and a high oxygen content, has the lowest ΔHcomb. Lignite is extensively mined in Germany and Poland. Anthracite, in contrast, with a hydrogen:carbon ratio of about 0.5 and the lowest oxygen content, has the highest ΔHcomb and is the highest grade of coal. Anthracite is the first choice for metallurgical refining. The most abundant form in the Western United States in anthracite while that in the Eastern United States is bituminous coal, which has a high sulfur content because of the presence of small particles of pyrite (FeS2). Combustion of coal releases the sulfur in FeS2 as SO2, which is a major contributor to acid rain. Table 15.7.3 compares the ΔHcomb per gram of oil, natural gas, and coal with those of selected organic compounds.
Table 15.7.2 Properties of Different Types of Coal
| Type | % Carbon | Hydrogen:Carbon Mole Ratio | % Oxygen | % Sulfur | Heat Content | US Deposits |
|---|---|---|---|---|---|---|
| anthracite | 92 | 0.5 | 3 | 1 | high | Pennsylvania, New York |
| bituminous | 80 | 0.6 | 8 | 5 | medium | Appalachia, Midwest, Utah |
| subbituminous | 77 | 0.9 | 16 | 1 | medium | Rocky Mountains |
| lignite | 71 | 1.0 | 23 | 1 | low | Montana |
Table 15.7.3 Enthalpies of Combustion of Common Fuels and Selected Organic Compounds
| Fuel | ΔHcomb (kJ/g) |
|---|---|
| dry wood | −15 |
| peat | −20.8 |
| bituminous coal | −28.3 |
| charcoal | −35 |
| kerosene | −37 |
| C6H6 (benzene) | −41.8 |
| crude oil | −43 |
| natural gas | −50 |
| C2H2 (acetylene) | −50.0 |
| CH4 (methane) | −55.5 |
| gasoline | −84 |
| hydrogen | −143 |
Peat, a precursor to coal, is the partially decayed remains of plants that grow in the swampy areas of the Carboniferous Period. It is removed from the ground in the form of soggy bricks of mud that will not burn until they have been dried. Even though peat is a smoky, poor-burning fuel that gives off relatively little heat, humans have burned it since ancient times (Figure 15.7.4). If a peat bog were buried under many layers of sediment for a few million years, the peat would eventually be compressed and heated enough to become lignite, the lowest grade of coal; given enough time and heat, lignite would eventually become anthracite, a much better fuel.
Converting Coal to Gaseous and Liquid Fuels
As a solid, coal is much more difficult to mine and ship than petroleum (a liquid) or natural gas. Consequently, more than 75% of the coal produced each year is simply burned in power plants to produce electricity. Methods to convert coal to gaseous fuels (coal gasification) or liquid fuels (coal liquefaction) exist, but are not particularly economical unless the prices of oil and natural gas are high. With the development of fracking and the subsequent fall in oil and natural gas prices interest in these processes has fallen however they have played an important role in the past. In the most common approach to coal gasification, coal reacts with steam to produce a mixture of CO and H2 known as synthesis gas, or syngas:Because coal is 70%–90% carbon by mass, it is approximated as C in Equation 15.7.1
\( C\left ( s \right ) + H_{2}O \left ( g \right ) \rightarrow CO \left ( g \right ) + H_{2} \left ( g \right ) \; \; \; \; \Delta H= 131 \; kJ \tag{15.8.1} \)
Converting coal to syngas removes any sulfur present and produces a clean-burning mixture of gases. Syngas or town gas was used for cooking until the 1960s when natural gas pipelines were built. Because syngas contains carbon monoxide (CO) it is poisonous, which accounts for scenes in old movies where people were killed by sticking their heads into an oven and allowing the gas to flow.
Syngas is can also used as a reactant to produce methane and methanol. A promising approach is to convert coal directly to methane through a series of reactions:
\( \begin{matrix}
2C\left ( s \right )+2H_{2}O\left ( g \right ) \rightarrow \cancel{2CO\left ( g \right )}+\cancel{2H_{2}\left ( g \right )} & \Delta H_{1}=262 \; kJ \\
\cancel{CO\left ( g \right )}+\cancel{H_{2}O \left ( g \right )} \rightarrow CO_{2}\left ( g \right )+\cancel{H_{2}\left ( g \right )} & \Delta H_{2}=-41 \; kJ \\
\cancel{CO\left ( g \right )}+\cancel{3H_{2}\left ( g \right )} \rightarrow CH_{4}\left ( g \right )+\cancel{H_{2}O\left ( g \right )} & \Delta H_{3}=-206 \; kJ \\
-------------------&--------\\
2C\left ( s \right )+2H_{2}O\left ( g \right ) \rightarrow CH_{4}\left ( g \right ) + CO_{2}\left ( g \right ) & \Delta H_{3}=15 \; kJ
\end{matrix} \tag{15.8.2} \)
Techniques available for converting coal to liquid fuels are not economically competitive with the production of liquid fuels from petroleum. Current approaches to coal liquefaction use a catalyst to break the complex network structure of coal into more manageable fragments. The products are then treated with hydrogen (from syngas or other sources) under high pressure to produce a liquid more like petroleum. Subsequent distillation, cracking, and reforming can be used to create products similar to those obtained from petroleum.
Petroleum
The petroleum that is pumped out of the ground is a complex mixture of several thousand organic compounds including straight-chain alkanes, cycloalkanes, alkenes, and aromatic hydrocarbons with four to several hundred carbon atoms. The identities and relative abundences of the components vary depending on the source. So Texas crude oil is somewhat different from Saudi Arabian crude oil. In fact, the analysis of petroleum from different deposits can produce a “fingerprint” of each, which is useful in tracking down the sources of spilled crude oil. For example, Texas crude oil is “sweet,” meaning that it contains a small amount of sulfur-containing molecules, whereas Saudi Arabian crude oil is “sour,” meaning that it contains a relatively large amount of sulfur-containing molecules.
Gasoline
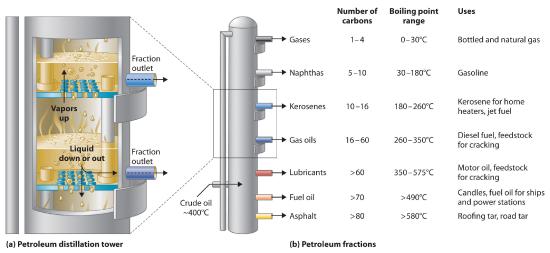
Petroleum is converted to useful products such as gasoline in three steps: distillation, cracking, and reforming. Recall that distillation separates compounds on the basis of their relative volatility, which is usually inversely proportional to their boiling points. Part (a) in Figure 15.7.5 shows a cutaway drawing of a column used in the petroleum industry for separating the components of crude oil. The petroleum is heated to approximately 400°C (750°F), at which temperature it has become a mixture of liquid and vapor. This mixture, called the feedstock, is introduced into the refining tower. The most volatile components (those with the lowest boiling points) condense at the top of the column where it is cooler, while the less volatile components condense nearer the bottom. Some materials are so nonvolatile that they collect at the bottom without evaporating at all. Thus the composition of the liquid condensing at each level is different. These different fractions, each of which usually consists of a mixture of compounds with similar numbers of carbon atoms, are drawn off separately. Part (b) in Figure 15.7.5 shows the typical fractions collected at refineries, the number of carbon atoms they contain, their boiling points, and their ultimate uses. These products range from gases used in natural and bottled gas to liquids used in fuels and lubricants to gummy solids used as tar on roads and roofs.
Figure 15.7.5: The Distillation of Petroleum. (a) This is a diagram of a distillation column used for separating petroleum fractions. (b) Petroleum fractions condense at different temperatures, depending on the number of carbon atoms in the molecules, and are drawn off from the column. The most volatile components (those with the lowest boiling points) condense at the top of the column, and the least volatile (those with the highest boiling points) condense at the bottom.
The economics of petroleum refining are complex. For example, the market demand for kerosene and lubricants is much lower than the demand for gasoline, yet all three fractions are obtained from the distillation column in comparable amounts. Furthermore, most gasolines and jet fuels are blends with very carefully controlled compositions that cannot vary as their original feedstocks did. To make petroleum refining more profitable, the less volatile, lower-value fractions must be converted to more volatile, higher-value mixtures that have carefully controlled formulas. The first process used to accomplish this transformation is cracking, in which the larger and heavier hydrocarbons in the kerosene and higher-boiling-point fractions are heated to temperatures as high as 900°C. High-temperature reactions cause the carbon–carbon bonds to break, which converts the compounds to lighter molecules similar to those in the gasoline fraction. Thus in cracking, a straight-chain alkane with a number of carbon atoms corresponding to the kerosene fraction is converted to a mixture of hydrocarbons with a number of carbon atoms corresponding to the lighter gasoline fraction. The second process used to increase the amount of valuable products is called reforming; it is the chemical conversion of straight-chain alkanes to either branched-chain alkanes or mixtures of aromatic hydrocarbons. Metal catalysts such as platinum are used to drive the necessary chemical reactions. The mixtures of products obtained from cracking and reforming are separated by fractional distillation.
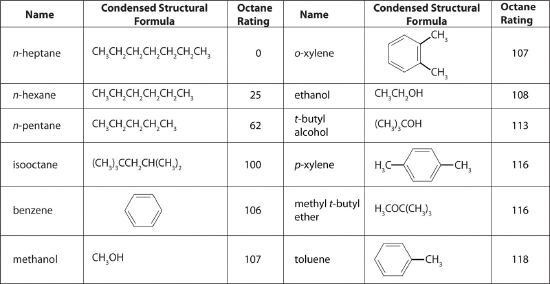
Octane Ratings
The quality of a fuel is indicated by its octane rating, which is a measure of its ability to burn in a combustion engine without knocking or pinging. Knocking and pinging signal premature combustion (Figure 15.8.6), which can be caused either by an engine malfunction or by a fuel that burns too fast. In either case, the gasoline-air mixture detonates at the wrong point in the engine cycle, which reduces the power output and can damage valves, pistons, bearings, and other engine components. The various gasoline formulations are designed to provide the mix of hydrocarbons least likely to cause knocking or pinging in a given type of engine performing at a particular level.
Figure 15.76: The Burning of Gasoline in an Internal Combustion Engine. (a) Normally, fuel is ignited by the spark plug, and combustion spreads uniformly outward. (b) Gasoline with an octane rating that is too low for the engine can ignite prematurely, resulting in uneven burning that causes knocking and pinging.
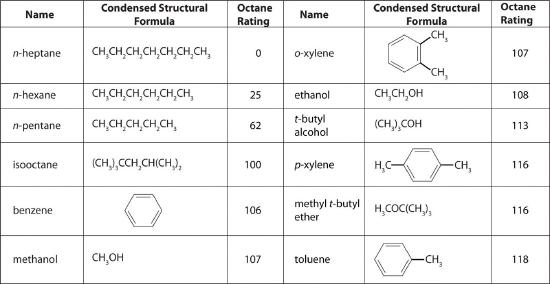
The octane scale was established in 1927 using a standard test engine and two pure compounds: n-heptane and isooctane (2,2,4-trimethylpentane). n-Heptane, which causes a great deal of knocking on combustion, was assigned an octane rating of 0, whereas isooctane, a very smooth-burning fuel, was assigned an octane rating of 100. Chemists assign octane ratings to different blends of gasoline by burning a sample of each in a test engine and comparing the observed knocking with the amount of knocking caused by specific mixtures of n-heptane and isooctane. For example, the octane rating of a blend of 89% isooctane and 11% n-heptane is simply the average of the octane ratings of the components weighted by the relative amounts of each in the blend. Converting percentages to decimals, we obtain the octane rating of the mixture:
\[0.89(100)+0.11(0)=89 \tag{4.7.1}\]
A gasoline that performs at the same level as a blend of 89% isooctane and 11% n-heptane is assigned an octane rating of 89; this represents an intermediate grade of gasoline. Regular gasoline typically has an octane rating of 87; premium has a rating of 93 or higher.
As shown in Figure 4.7.3, many compounds that are now available have octane ratings greater than 100, which means they are better fuels than pure isooctane. In addition, antiknock agents, also called octane enhancers, have been developed. One of the most widely used for many years was tetraethyllead [(C2H5)4Pb], which at approximately 3 g/gal gives a 10–15-point increase in octane rating. Since 1975, however, lead compounds have been phased out as gasoline additives because they are highly toxic.
Other enhancers, such as methyl t-butyl ether (MTBE), have been developed to take their place that combine a high octane rating with minimal corrosion to engine and fuel system parts. Unfortunately, when gasoline containing MTBE leaks from underground storage tanks, the result has been contamination of the groundwater in some locations, resulting in limitations or outright bans on the use of MTBE in certain areas. As a result, the use of alternative octane enhancers such as ethanol, which can be obtained from renewable resources such as corn, sugar cane, and, eventually, corn stalks and grasses, is increasing.
Natural Gas
Natural gas is a (mostly) combustible gas found underground. While primarily composed of methane (70-90%) the gas from each well has a different composition and the value of the other components affects the value of the gas. The gas from wells that are rich in methane is called dry and wells that have a considerable amount of higher hydrocarbons produce wet gas. The higher hydrocarbons have value above that of methane so stripping them out is important. Some wells are sour because their gas has hydrogen sulfide which must be removed before the gas can be used for heating or generating electricity.
Finally, a few wells in Texas and nearby Oklahoma have a relatively high amount of helium (0.3 - 2.7%). The Helium Act of 1925 established a national helium reserve at Cliffside near Amarillo TX. Political pressure and costs pushed laws to privatize the reserve, but other policy considerations including the need for helium for scientific research has slowed the process.
Table 15.8.4: Composition of Natural Gas
| Gas | Molecular Formula | Composition |
| Methane | CH4 | 70-95% |
| Ethane | C2H6 | 0-20% |
| Carbon Dioxide | CO2 | 0-8% |
| Nitrogen | N2 | 0-5% |
| Hydrogen Sulfide | H2S | 0-5% |
| Propane | C3H8 | Traces |
| Butane | C4H10 | Traces |
| Rare Gases | He (also Ne) | 0-3% (only in Texas) |
The purification of natural gas is a complex process with many steps as each of the impurities is stripped out
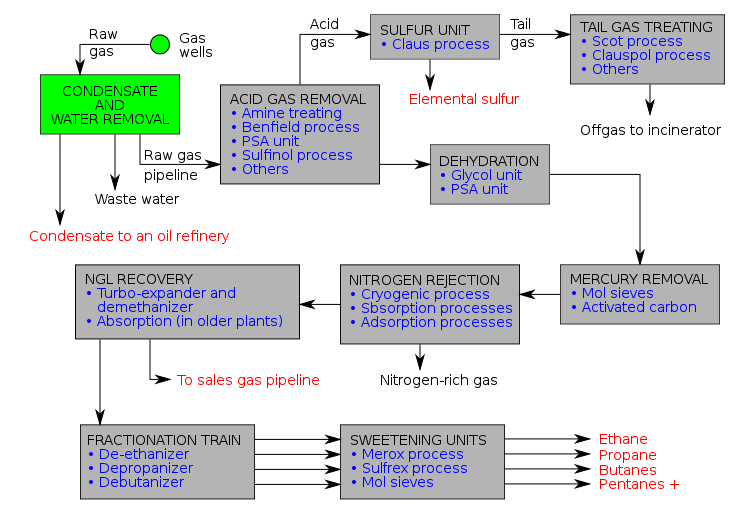
Figure 15.7.7: Purification of natural gas: Prospective Chemical Engineers can find details about each process by searching the net. The flow chart is from the Wikipedia
Gas turbine power plants to generate electricity are coming increasingly into use as fracking and other advanced drilling technologies have driven the cost of natural gas down and the supply up. While on a continental scale natural gas is transported by pipelines, natural gas can be cooled and compressed to be transported as liquified natural gas. Gas turbine power plants are small and quickly built. They can be rapidly spun up to meet peak demand. More detailed information can be found at the Department of Energy Fossil Fuel web site
The Carbon Cycle and the Greenhouse Effect
Since 1850 the burning of fossil fuels has increased the concentration of carbon dioxide in the atmosphere from 280 to just over 400 ppmV. A continued increase in the CO2 burden in the atmosphere will have serious negative effects and this requires shifting our entire energy producing economy from fossil fuels to non-carbon sources such as hydro, solar, wind and nuclear. These include sea level rise that will threaten low lying cities including but not limited to Miami Beach and Norfolk in the US, even a meter or more coupled with storm surge and high tides can cause massive damage as was seen during Hurricane Sandy. Increased carbon dioxide in the atmosphere has already measurably decreased the pH of the oceans. Sea life is adapted to a narrow range of pH.
Higher global temperatures of 2 or 3 C may not seem much, but one should keep in mind that the average global temperature during the ice ages was only ~6 C lower than it is today. During the Eemian interglacial the average temperature was only a few degrees higher than the present and the sea level was 6-9 m higher. Finally, humans are mammals who maintain a core temperature within a few degrees of 37.0 C. In hot weather we do so by evaporation of sweat however there are limits to this and by 2100 there is a significant probability even in the US that at least a few days a year will reach this limit by 2100. Given that most people on earth do not have access to air conditioning, parts of the planet may become uninhabitable. Indeed a worst case and a serious problem that merits attention.
Given the constraints of this text it is difficult to provide the level of detail needed to understand why this is so. A good source for those interested in learning more is David Archer's Global Warming, Understanding the Forecast and An Introduction to Modern Climate Change by Andrew Dessler
There are a few basic facts that anyone starting to learn about the issue need to know. First, that the Earth gains energy from the Sun, and that it must radiate the energy at the same rate. If more energy is absorbed than radiated the Earth will warm up, if less energy is absorbed than radiated it will cool. As discussed in Section 15.1 solar radiation follows a 5500 K blackbody distribution while radiation from the surface of the earth is also black body but at ~290K. This is shown schematically in Figure 15.8.8
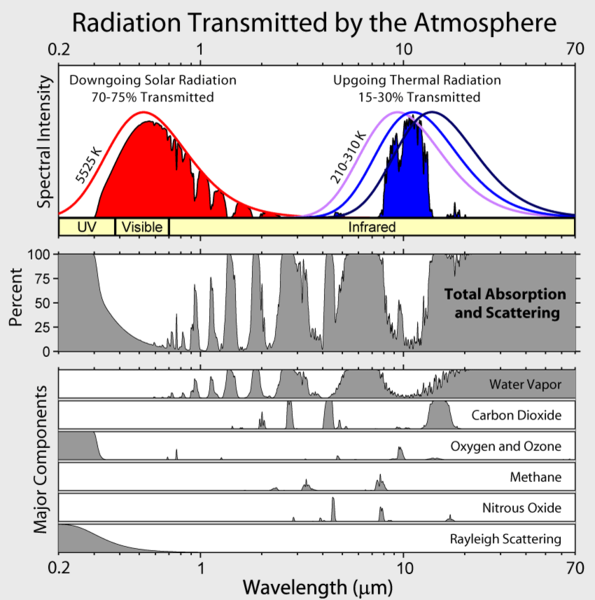
Figure 15.7.8: Origin of the greenhouse effect. The top box show the intensity of solar radiation at the top of the Earth's atmosphere to the left and the infrared emission from the atmosphere to the left. The next shows the net absorption in the atmosphere across the spectrum. The bottom show the absorption by different atmospheric species at their current concentrations. Figure from the Wikipedia
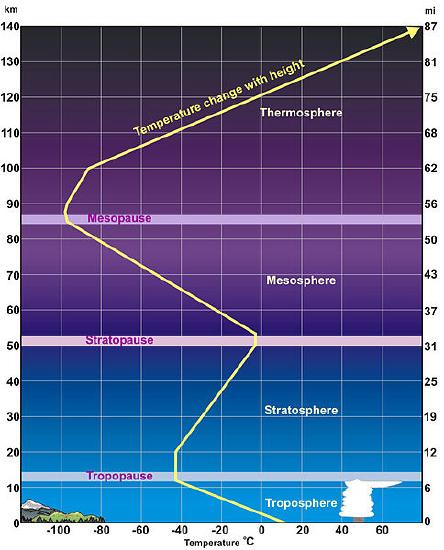
The atmosphere cools with altitude up to about 15 km where it starts warming again because of absorption of UV radiation by ozone in the stratosphere. The transition between the troposphere and the stratosphere is called the tropopause and is the coldest part of the atmosphere.
Figure 15.7.9 The thermal structure of the atmosphere. The troposphere is the lowest level,extending from the the surface to the tropopause which is at 12-15 km altitude, more in the tropics and less less at the poles. The troposphere is the section of the atmosphere relevant to the greenhouse effect. The ozone layer is found in the stratosphere and one observes the aurora borealis or northern lights in the mesosphere Taken from Wikimedia
Figure 15.7.10 shows the IR emission spectrum observed looking down on the earth from a high altitude balloon
Figure 15.7.10: High resolution far infrared emission spectrum of the atmosphere looking down from a high altitude balloon at 35 km. The strong band between 600 and 700 cm-1 is associated with CO2, the sharp band at ~ 1100 cm-1 is associated with ozone and the feature near 1300 cm-1 is due to methane. The sharp lines (the hair) are due to water vapor. Spectrum is from the Federal Earth Observation Portal and was taken by an instrument operated by NASA/Langeley Research Center
The dotted lines in Figure 15.8.9 are blackbody curves. The IR window shown schematically in blue in Figure 15.8.8 is the region between the ozone and the carbon dioxide band, where the emission from the hot, 320 K ground follows the blackbody curve with a few sharp water vapor absorption lines. At those wavelengths, in the IR window, the emission comes directly from the surface. The CO2 band extends down to about a 220 K blackbody curve. What this means is that radiation from CO2 only escapes to space from the level in the troposphere where that is the temperature. The rate of emission is proportional to T4 so the rate of emission from higher, therefore colder levels, is slower. Radiation in this area of the spectrum from the surface is blocked and only the greatly reduced emission from the upper troposphere escapes to space. The same is true for the ozone and methane bands as well as the water lines.
The net effect is that the surface must warm in order to maintain the balance between incoming solar radiation and the outgoing emission. There is a simple calculation which models the atmosphere as a one dimensional problem and calculates what the temperature of the surface would be if there were no greenhouse gases. The result is 255 K, rather cold. In fact if one attempts a more complex calculation the effective temperature without greenhouse gases would be even colder.
What happens if we increase the carbon dioxide in the atmosphere? The altitude at which the atmosphere can emit radiation to space will rise because of increased absorption by the CO2. Since in the troposphere the temperature decreases with altitude, the rate of emission from a higher level must decrease. Again, in order to maintain the balance between incoming solar radiation and the outgoing emission the surface will have to warm even more, thus the term global warming. The change is not linear with increasing CO2 but logarithmic. But, of course it is not so simple, because increasing the surface temperature will increase the water vapor pressure in the atmosphere, which will increase the temperature further.
We also have to understand the flow of carbon between the atmosphere, the biosphere, the upper oceans and the deep. Observations to date show that natural emissions of CO2 from these reservoirs are in balance with absorption, while only about half of fossil fuel emissions remain in the atmosphere, the rest being absorbed by the upper ocean and the biosphere. The three upper reservoirs equilibrate in a decade or less, but flow into the deep ocean requires roughly a thousand years.
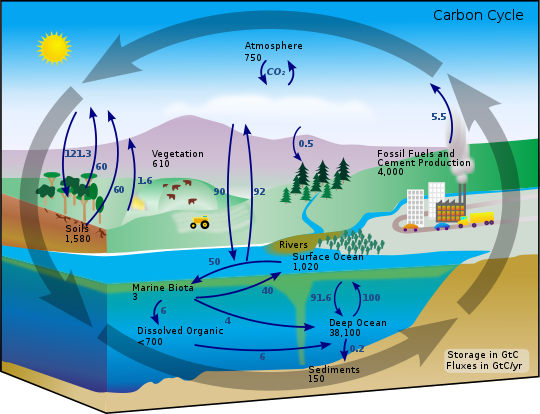
Figure 15.7.11: The carbon cycle. Figure from the Wikipedia
There is no doubt that atmospheric CO2 levels are increasing, and the major reason for this increase is the combustion of fossil fuels. An extremely conservative statement of the situation today can be found in the 2014 Synthesis Report from the IPCC, a consensus between scientists and policymakers. The report starkly states that
Cumulative emissions of CO2 largely determine global mean surface warming by the late 21st century and beyond.
and concludes that
Continued emission of greenhouse gases will cause further warming and long-lasting changes in all components of the climate system, increasing the likelihood of severe, pervasive and irreversible impacts for people and ecosystems. Limiting climate change would require substantial and sustained reductions in greenhouse gas emissions which, together with adaptation, can limit climate change risks.
The situation is serious, but we can work together to limit and even reverse damage while maintaining our standard of living in the developed world while helping the developing world to a better future. However, the issues are complex and we can only touch on some of the basics here.
Summary
More than 80% of the energy used by modern society (about 3 × 1017 kJ/yr) is from the combustion of fossil fuels. Because of their availability, ease of transport, and facile conversion to convenient fuels, natural gas and petroleum are currently the preferred fuels. Coal is primarily used for electricity generation. The combustion of fossil fuels releases large amounts of CO2 that upset the balance of the carbon cycle and result in a steady increase in atmospheric CO2 levels. Because CO2 is a greenhouse gas, which absorbs heat before it can be radiated from Earth into space, CO2 in the atmosphere results in increased surface temperatures (the greenhouse effect).
Key Takeaway
- Thermochemical concepts can be used to calculate the efficiency of various forms of fuel, which can then be applied to environmental issues.
Conceptual Problems
-
What is meant by the term greenhouse gases? List three greenhouse gases that have been implicated in global warming.
-
Name three factors that determine the rate of planetary CO2 uptake.
-
The structure of coal is quite different from the structure of gasoline. How do their structural differences affect their enthalpies of combustion? Explain your answer.
Numerical Problems
-
One of the side reactions that occurs during the burning of fossil fuels is
4FeS2(s) + 11O2(g) → 2Fe2O3(s) + 8SO2(g)- How many kilojoules of energy are released during the combustion of 10 lb of FeS2?
- How many pounds of SO2 are released into the atmosphere?
- Discuss the potential environmental impacts of this combustion reaction.
-
How many kilograms of CO2 are released during the combustion of 16 gal of gasoline? Assume that gasoline is pure isooctane with a density of 0.6919 g/mL. If this combustion was used to heat 4.5 × 103 L of water from an initial temperature of 11.0°C, what would be the final temperature of the water assuming 42% efficiency in the energy transfer?
-
A 60 W light bulb is burned for 6 hours. If we assume an efficiency of 38% in the conversion of energy from oil to electricity, how much oil must be consumed to supply the electrical energy needed to light the bulb? (1 W = 1 J/s)
-
How many liters of cyclohexane must be burned to release as much energy as burning 10.0 lb of pine logs? The density of cyclohexane is 0.7785 g/mL, and its ΔHcomb = −46.6 kJ/g.
Contributors
- Anonymous
Modified by Joshua Halpern (Howard University), Scott Sinex, and Scott Johnson (PGCC)