27.5: Introduction to Addition Reactions: Reactions of Alkenes
- Page ID
- 24392
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The most common chemical transformation of a carbon-carbon double bond is the addition reaction. A large number of reagents, both inorganic and organic, have been found to add to this functional group, and in this section we shall review many of these reactions.
- Addition of Lewis Acids (Electrophilic Reagents)
- Addition of Strong Brønsted Acids
- Rearrangement of Carbocations
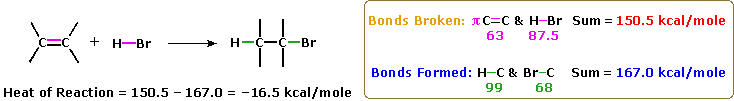
A large number of reagents, both inorganic and organic, have been found to add to this functional group, and in this section we shall review many of these reactions. A majority of these reactions are exothermic, due to the fact that the C-C pi-bond is relatively weak (ca. 63 kcal/mole) compared to the sigma-bonds formed with the atoms or groups of the reagent. Remember, the bond energies of a molecule are the energies required to break (homolytically) all the covalent bonds in the molecule. Consequently, if the bond energies of the product molecules are greater than the bond energies of the reactants, the reaction will be exothermic. The following calculations for the addition of H-Br are typical. Note that by convention exothermic reactions have a negative heat of reaction.
Addition of Hydrogen: Hydrogenation of Alkenes
Addition of hydrogen to a carbon-carbon double bond is called hydrogenation. The overall effect of such an addition is the reductive removal of the double bond functional group. Regioselectivity is not an issue, since the same group (a hydrogen atom) is bonded to each of the double bond carbons. The simplest source of two hydrogen atoms is molecular hydrogen (H2), but mixing alkenes with hydrogen does not result in any discernible reaction. Although the overall hydrogenation reaction is exothermic, a high activation energy prevents it from taking place under normal conditions. This restriction may be circumvented by the use of a catalyst,
Ethene reacts with hydrogen in the presence of a finely divided palladium catalyst at a temperature of about 150°C. Ethane is produced.
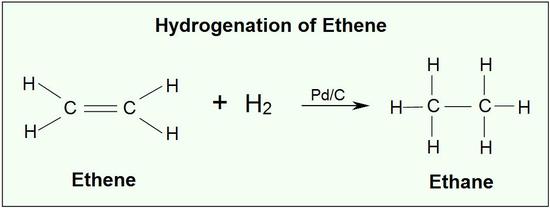
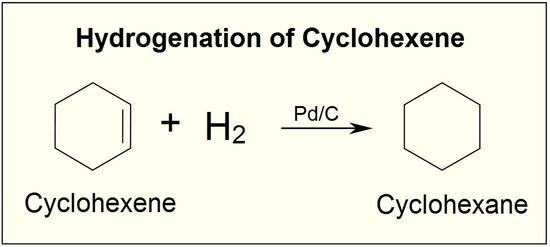
This is a fairly pointless reaction because ethene is a far more useful compound than ethane! However, what is true of the reaction of the carbon-carbon double bond in ethene is equally true of it in much more complicated cases.
Addition of Hydrogen Halides to Alkenes
All alkenes undergo addition reactions with the hydrogen halides. A hydrogen atom joins to one of the carbon atoms originally in the double bond, and a halogen atom to the other. For example, with ethene and hydrogen chloride, you get chloroethane:
![]()

With but-2-ene you get 2-chlorobutane:
![]()
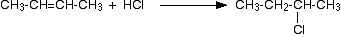
What happens if you add the hydrogen to the carbon atom at the right-hand end of the double bond, and the chlorine to the left-hand end? You would still have the same product. The chlorine would be on a carbon atom next to the end of the chain - you would simply have drawn the molecule flipped over in space. That would be different of the alkene was unsymmetrical - that's why we have to look at them separately.
Reaction rates increase in the order HF - HCl - HBr - HI. Hydrogen fluoride reacts much more slowly than the other three, and is normally ignored in talking about these reactions. When the hydrogen halides react with alkenes, the hydrogen-halogen bond has to be broken. The bond strength falls as you go from HF to HI, and the hydrogen-fluorine bond is particularly strong. Because it is difficult to break the bond between the hydrogen and the fluorine, the addition of HF is bound to be slow.
This applies to unsymmetrical alkenes as well as to symmetrical ones. For simplicity the examples given below are all symmetrical ones- but they don't have to be. Reaction rates increase as the alkene gets more complicated - in the sense of the number of alkyl groups (such as methyl groups) attached to the carbon atoms at either end of the double bond. For example:
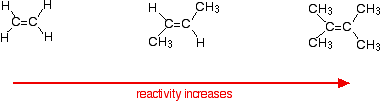
There are two ways of looking at the reasons for this - both of which need you to know about the mechanism for the reactions.
Alkenes react because the electrons in the \(pi\) bond attract things with any degree of positive charge. Anything which increases the electron density around the double bond will help this. Alkyl groups have a tendency to "push" electrons away from themselves towards the double bond. The more alkyl groups you have, the more negative the area around the double bonds becomes. The more negatively charged that region becomes, the more it will attract molecules like hydrogen chloride.
The more important reason, though, lies in the stability of the intermediate ion formed during the reaction. The three examples given above produce these carbocations (carbonium ions) at the half-way stage of the reaction:
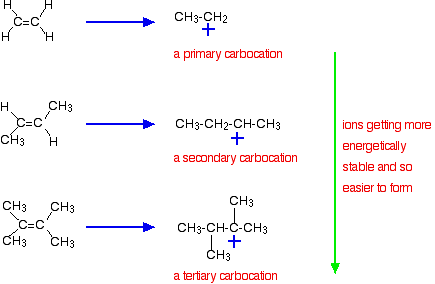
The stability of the intermediate ions governs the activation energy for the reaction. As you go towards the more complicated alkenes, the activation energy for the reaction falls. That means that the reactions become faster.
Addition to unsymmetrical alkenes
In terms of reaction conditions and the factors affecting the rates of the reaction, there is no difference whatsoever between these alkenes and the symmetrical ones described above. The problem comes with the orientation of the addition - in other words, which way around the hydrogen and the halogen add across the double bond. If HCl adds to an unsymmetrical alkene like propene, there are two possible ways it could add. However, in practice, there is only one major product.
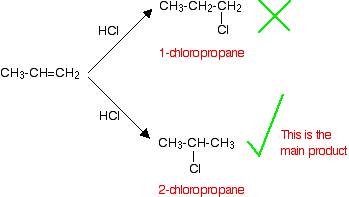
This is in line with Markovnikov's Rule.
When a compound HX is added to an unsymmetrical alkene, the hydrogen becomes attached to the carbon with the most hydrogens attached to it already.
In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count. The ones in the CH3 group are totally irrelevant.
Addition of Halogens: Halogenation of Alkenes
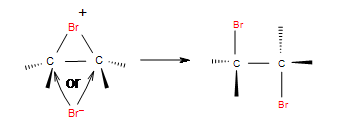
As halogen molecule, for example Br2, approaches a double bond of the alkene, electrons in the double bond repel electrons in bromine molecule causing polarization of the halogen bond. This creates a dipolar moment in the halogen molecule bond. Heterolytic bond cleavage occurs and one of the halogens obtains positive charge and reacts as an electrophile. The reaction of the addition is not regioselective but stereoselective.Stereochemistry of this addition can be explained by the mechanism of the reaction.In the first step electrophilic halogen with a positive charge approaches the double carbon bond and 2 p orbitals of the halogen, bond with two carbon atoms and create a cyclic ion with a halogen as the intermediate step. In the second step, halogen with the negative charge attacks any of the two carbons in the cyclic ion from the back side of the cycle as in the SN2 reaction. Therefore stereochemistry of the product is vicinial dihalides through anti addition.
\[\ce{R_2C=CR_2 + X_2 \rightarrow R_2CX-CR_2X}\]
Halogens that are commonly used in this type of the reaction are: \(Br\) and \(Cl\). In thermodynamical terms \(I\) is too slow for this reaction because of the size of its atom, and \(F\) is too vigorous and explosive. Solvents that are used for this type of electrophilic halogenation are inert (e.g., CCl4) can be used in this reaction. Because halogen with negative charge can attack any carbon from the opposite side of the cycle it creates a mixture of steric products.
Electrophilic addition mechanism consists of two steps. Before constructing the mechanism let us summarize conditions for this reaction. We will use Br2 in our example for halogenation of ethylene.
| Nucleophile | Double bond in alkene |
| Electrophile | Br2, Cl2 |
| Regiochemistry | not relevant |
|
Stereochemistry |
ANTI |
Step 1: In the first step of the addition the Br-Br bond polarizes, heterolytic cleavage occurs and Br with the positive charge forms a intermediate cycle with the double bond.
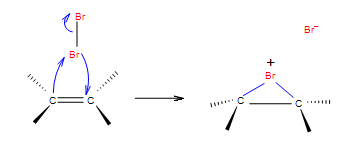
Step 2: In the second step, bromide anion attacks any carbon of the bridged bromonium ion from the back side of the cycle. Cycle opens up and two halogens are in the position anti.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

