16.3: Self-Ionization of Water and the pH Scale
- Page ID
- 24292
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To understand the autoionization reaction of liquid water.
- To know the relationship among pH, pOH, and \(pK_w\).
Because of its highly polar structure, liquid water can act as either an acid (by donating a proton to a base) or a base (by using a lone pair of electrons to accept a proton). For example, when a strong acid such as HCl dissolves in water, it dissociates into chloride ions (\(Cl^−\)) and protons (\(H^+\)). The proton, in turn, reacts with a water molecule to form the hydronium ion (\(H_3O^+\)):
\[\underset{aicd}{\ce{HCl(aq)}} + \underset{base}{\ce{H2O(l)}} \rightarrow \underset{acid}{\ce{H3O^{+}(aq)}} + \underset{base}{\ce{Cl^{-}(aq)}} \label{16.3.1a}\]
In this reaction, \(\ce{HCl}\) is the acid, and water acts as a base by accepting an \(\ce{H^{+}}\) ion. The reaction in Equation \(\ref{16.3.1a}\) is often written in a simpler form by removing \(\ce{H2O}\) from each side:
\[ \ce {HCl (aq) \rightarrow H^{+} (aq) + Cl^{-} (aq)} \label{16.3.1b}\]
In Equation \(\ref{16.3.1b}\), the hydronium ion is represented by \(\ce{H^{+}(aq)}\), although free \(\ce{H^{+}}\) ions do not exist in liquid water as this reaction demonstrates:
\[ \ce{H^{+}(aq) + H2O(l) \rightarrow H3O^{+}(aq)}\]
Water can also act as an acid, as shown in Equation \(\ref{16.3.2}\). In this equilibrium reaction, \(H_2O\) donates a proton to \(NH_3\), which acts as a base:
\[\underset{aicd}{\ce{H2O(aq)}} + \underset{base}{\ce{NH3(aq)}} \rightleftharpoons \underset{acid}{\ce{NH^{+}4 (aq)}} + \underset{base}{\ce{OH^{-}(aq)}} \label{16.3.2}\]
Water is thus termed amphiprotic, meaning that it can behave as either an acid or a base, depending on the nature of the other reactant. Notice that Equation \(\ref{16.3.2}\) is an equilibrium reaction as indicated by the double arrow and hence has an equilibrium constant associated with it.
The Ion-Product Constant of Pure Water
Because water is amphiprotic, one water molecule can react with another to form an \(OH^−\) ion and an \(H_3O^+\) ion in an autoionization process:
\[\ce{2H2O(l) <=> H3O^{+}(aq) + OH^{−}(aq)} \label{16.3.3}\]
In pure water, a very small fraction of water molecules donate protons to other water molecules to form hydronium ions and hydroxide ions:
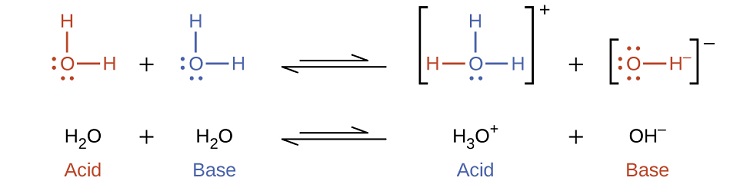
The equilibrium constant \(K\) for this reaction can be written as follows:
\[K_{a}=\dfrac{a_{H_3O^+}·a_{OH^-}}{a_{H_2O}^2} \approx \frac{[H_{3}O^{+}][HO^{-}]}{(1)^{2}}=[H_{3}O^{+}][HO^{-}] \label{16.3.4}\]
where \(a\) is the activity of a species. Because water is the solvent, and the solution is assumed to be dilute, the activity of the water is approximated by the activity of pure liquid water, which is defined as having a value of 1. The activity of each solute is approximated by the molarity of the solute.
It is a common error to claim that the molar concentration of the solvent is in some way involved in the equilibrium law. This error is a result of a misunderstanding of solution thermodynamics. For example, it is often claimed that Ka = Keq[H2O] for aqueous solutions. This equation is incorrect because it is an erroneous interpretation of the correct equation Ka = Keq(\(\textit{a}_{H_2O}\)). Because \(\textit{a}_{H_2O}\) = 1 for a dilute solution, Ka = Keq(1), or Ka = Keq.
In this reaction, one water molecule acts as an acid and one water molecule acts as a base. Thus, this reaction actually can be designated as the \(K_a\) of water and as the \(K_b\) of water. It is most common, however, to designate this reaction and the associated law of mass action as the \(K_w\) of water:
\[K_{w}=[H_{3}O^{+}][HO^{-}] \label{16.3.5}\]
When pure liquid water is in equilibrium with hydronium and hydroxide ions at 25 °C, the concentrations of the hydronium ion and the hydroxide ion are equal:
\[[H_3O^+] = [OH^−] = 1.003 \times 10^{−7}\; M \label{16.3.6}\]
Thus the number of dissociated water molecules is very small indeed, approximately 2 ppb.
Substituting the values for \([H_3O^+]\) and \([OH^−]\) at 25°C into this expression
\[K_w=(1.003 \times 10^{−7})(1.003 \times 10^{−7})=1.006 \times 10^{−14} \label{16.3.7}\]
The slight ionization of pure water is reflected in the small value of the equilibrium constant; at 25 °C.Thus, to three significant figures, \(K_w = 1.01 \times 10^{−14}\; M\) at room temperature. Like any other equilibrium constant, \(K_w\) varies with temperature, ranging from \(1.15 \times 10^{−15}\) at 0°C to \(4.99 \times 10^{−13}\) at 100°C.
In pure water, the concentrations of the hydronium ion and the hydroxide ion are equal, and the solution is therefore neutral. If \([H_3O^+] > [OH^−]\), however, the solution is acidic, whereas if \([H_3O^+] < [OH^−]\), the solution is basic. For an aqueous solution, the \(H_3O^+\) concentration is a quantitative measure of acidity: the higher the \(H_3O^+\) concentration, the more acidic the solution. Conversely, the higher the \(OH^−\) concentration, the more basic the solution. In most situations that you will encounter, the \(H_3O^+\) and \(OH^−\) concentrations from the dissociation of water are so small (\(1.003 \times 10^{−7} M\)) that they can be ignored in calculating the \(H_3O^+\) or \(OH^−\) concentrations of solutions of acids and bases, but this is not always the case.
What are the hydronium ion concentration and the hydroxide ion concentration in pure water at 25 °C?
Solution
The autoionization of water yields the same number of hydronium and hydroxide ions. Therefore, in pure water, \(\ce{[H_3O^+]} = \ce{[OH^- ]}\). At 25 °C:
\[\begin{align*} K_\ce{w} &=\ce{[H_3O^+][OH^- ]} \\[4pt]&=\ce{[H_3O^+]^2} \\[4pt] &=\ce{[OH^- ]^2} \\[4pt] &=1.0 \times 10^{−14} \end{align*}\]
So:
\[\ce{[H_3O^+]}=\ce{[OH^- ]}=\sqrt{1.0 \times 10^{−14}} =1.0 \times 10^{−7}\; M \nonumber\]
The hydronium ion concentration and the hydroxide ion concentration are the same, and we find that both equal \(1.0 \times 10^{−7}\; M\).
The ion product of water at 80 °C is \(2.4 \times 10^{−13}\). What are the concentrations of hydronium and hydroxide ions in pure water at 80 °C?
- Answer
-
\(\ce{[H_3O^+]} = \ce{[OH^- ]} = 4.9 \times 10^{−7}\; M\)
It is important to realize that the autoionization equilibrium for water is established in all aqueous solutions. Adding an acid or base to water will not change the position of the equilibrium. Example \(\PageIndex{2}\) demonstrates the quantitative aspects of this relation between hydronium and hydroxide ion concentrations.
A solution of carbon dioxide in water has a hydronium ion concentration of \(2.0 \times 10^{−6}\; M\). What is the concentration of hydroxide ion at 25 °C?
Solution
We know the value of the ion-product constant for water at 25 °C:
\[\ce{2 H_2O}_{(l)} \rightleftharpoons \ce{H_3O^+}_{(aq)} + \ce{OH^-}_{(aq)} \nonumber\]
and
\[K_\ce{w}=\ce{[H3O+][OH^- ]}=1.0 \times 10^{−14} \nonumber\]
Thus, we can calculate the missing equilibrium concentration.
Rearrangement of the \(k_w\) expression yields that \([\ce{OH^- }]\) is directly proportional to the inverse of [H3O+]:
\[[\ce{OH^- }]=\dfrac{K_{\ce w}}{[\ce{H_3O^+}]}=\dfrac{1.0 \times 10^{−14}}{2.0 \times 10^{−6}}=5.0 \times 10^{−9} \nonumber\]
The hydroxide ion concentration in water is reduced to \(5.0 \times 10^{−9}\: M\) as the hydrogen ion concentration increases to \(2.0 \times 10^{−6}\; M\). This is expected from Le Chatelier’s principle; the autoionization reaction shifts to the left to reduce the stress of the increased hydronium ion concentration and the \(\ce{[OH^- ]}\) is reduced relative to that in pure water.
A check of these concentrations confirms that our arithmetic is correct:
\[K_\ce{w}=\ce{[H_3O^+][OH^- ]}=(2.0 \times 10^{−6})(5.0 \times 10^{−9})=1.0 \times 10^{−14} \nonumber\]
What is the hydronium ion concentration in an aqueous solution with a hydroxide ion concentration of 0.001 M at 25 °C?
- Answer
-
\[\ce{[H3O+]} = 1 \times 10^{−11} M \nonumber\]
Self-Ionization of Water (Kw): https://youtu.be/RMpO0rqUnFg
The Relationship among pH, pOH, and \(pK_w\)
The pH scale is a concise way of describing the \(H_3O^+\) concentration and hence the acidity or basicity of a solution. Recall that pH and the \(H^+\) (\(H_3O^+\)) concentration are related as follows:
\[\begin{align} pH &=−\log_{10}[\ce{H^{+}}] \label{16.3.8} \\[4pt] [\ce{H^{+}}] &=10^{−pH} \label{16.3.9} \end{align}\]
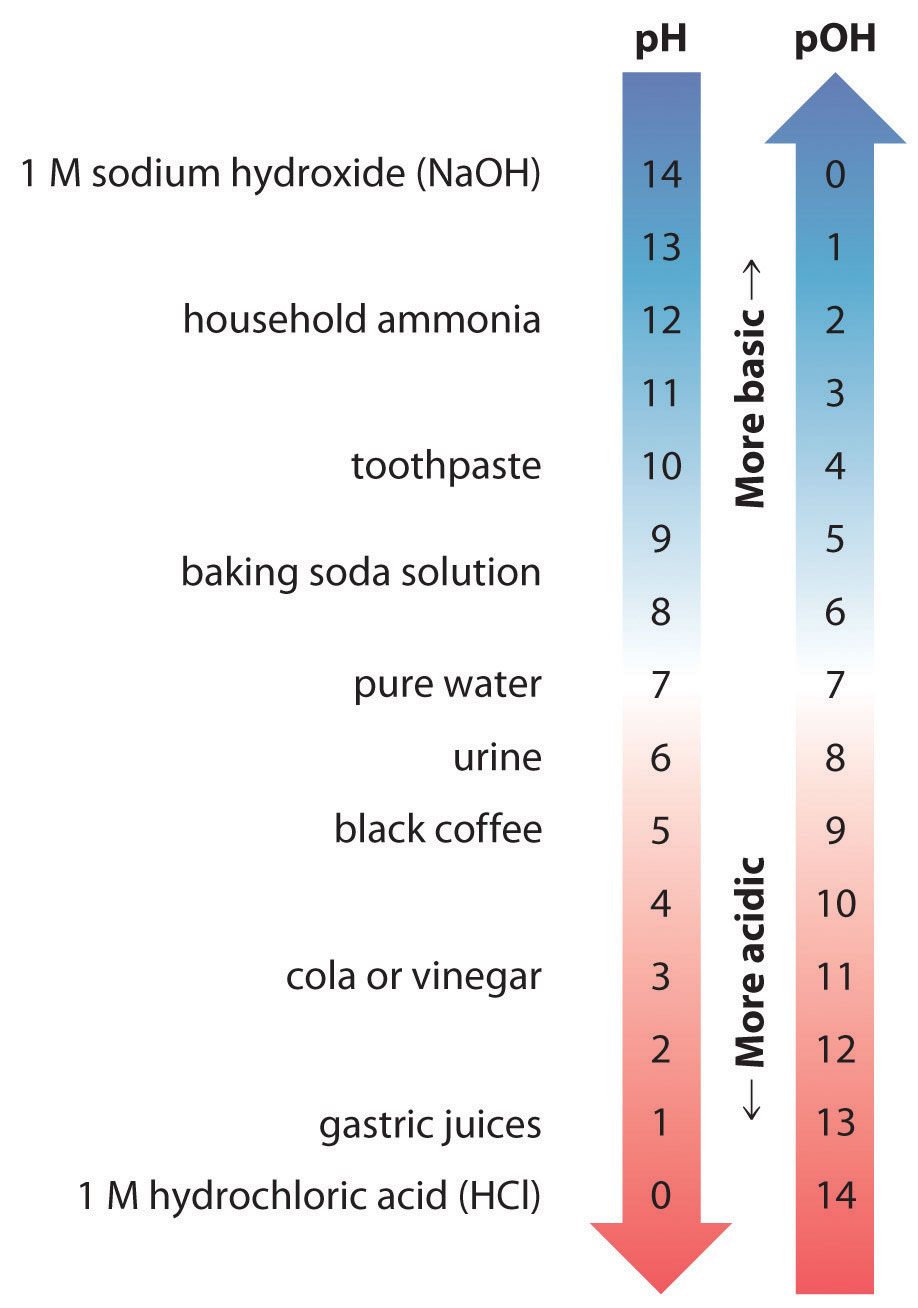
Because the scale is logarithmic, a pH difference of 1 between two solutions corresponds to a difference of a factor of 10 in their hydronium ion concentrations. Recall also that the pH of a neutral solution is 7.00 (\([H_3O^+] = 1.0 \times 10^{−7}\; M\)), whereas acidic solutions have pH < 7.00 (corresponding to \([\ce{H3O^{+}}] > 1.0 \times 10^{−7}\)) and basic solutions have pH > 7.00 (corresponding to \([\ce{H3O^{+}}] < 1.0 \times 10^{−7}\)).
Similar notation systems are used to describe many other chemical quantities that contain a large negative exponent. For example, chemists use an analogous pOH scale to describe the hydroxide ion concentration of a solution. The pOH and \([\ce{OH^{−}}]\) are related as follows:
\[\begin{align} pOH &=−\log_{10}[\ce{OH^{−}}] \label{16.3.10} \\[4pt] [\ce{OH^{−}}] &=10^{−pOH} \label{16.3.11} \end{align}\]
The constant \(K_w\) can also be expressed using this notation, where \(pK_w = −\log\; K_w\).
Because a neutral solution has \([OH^−] = 1.0 \times 10^{−7}\), the pOH of a neutral solution is 7.00. Consequently, the sum of the pH and the pOH for a neutral solution at 25°C is 7.00 + 7.00 = 14.00. We can show that the sum of pH and pOH is equal to 14.00 for any aqueous solution at 25°C by taking the negative logarithm of both sides of Equation \(\ref{16.3.6b}\):
\[\begin{align} −\log K_w &= pK_w \\[4pt] &=−\log([H_3O^+][OH^−]) \\[4pt] &= (−\log[H_3O^+]) + (−\log[OH^−]) \\[4pt] &= pH + pOH \label{16.3.12} \end{align}\]
Thus at any temperature, \(pH + pOH = pK_w\), so at 25°C, where \(K_w = 1.0 \times 10^{−14}\) and \(pH + pOH = 14.00\). More generally, the pH of any neutral solution is half of the \(pK_w\) at that temperature. The relationship among pH, pOH, and the acidity or basicity of a solution is summarized graphically in Figure \(\PageIndex{1}\) over the common pH range of 0 to 14. Notice the inverse relationship between the pH and pOH scales.
For any neutral solution, pH + pOH = 14.00 (at 25°C) with pH=pOH=7.
The \(k_w\) for water at 100°C is \(4.99 \times 10^{−13}\). Calculate \(pK_w\) for water at this temperature and the pH and the pOH for a neutral aqueous solution at 100°C. Report pH and pOH values to two decimal places.
Given: \(K_w\)
Asked for: \(pK_w\), \(pH\), and \(pOH\)
Strategy:
- Calculate pKw by taking the negative logarithm of \(K_w\).
- For a neutral aqueous solution, \([H_3O^+] = [OH^−]\). Use this relationship and Equation \ref{16.3.6b} to calculate \([H_3O^+]\) and \([OH^−]\). Then determine the pH and the pOH for the solution.
Solution:
A Because \(pK_w\) is the negative logarithm of Kw, we can write
\[pK_w = −\log K_w = −\log(4.99 \times 10^{−13}) = 12.302 \nonumber\]
The answer is reasonable: \(K_w\) is between \(10^{−13}\) and \(10^{−12}\), so \(pK_w\) must be between 12 and 13.
B Equation \ref{16.3.6b} shows that \(K_w = [H_3O^+][OH^−]\). Because \([H_3O^+] = [OH^−]\) in a neutral solution, we can let \(x = [H_3O^+] = [OH^−]\):
\[\begin{align*} K_w &= [\ce{H3O^{+}}][\ce{OH^{−}}] \\[4pt] &= (x)(x) \\[4pt] &=x^2 \\[4pt] x &=\sqrt{K_w} \\[4pt] &=\sqrt{4.99 \times 10^{−13}} \\[4pt] &=7.06 \times 10^{−7}\; M \end{align*}\]
Because \(x\) is equal to both \([\ce{H3O^{+}}]\) and \([\ce{OH^{−}}]\),
\[\begin{align*} pH = pOH &= −\log(7.06 \times 10^{−7}) \\[4pt] &= 6.15 \, \text{(to two decimal places)} \end{align*}\]
We could obtain the same answer more easily (without using logarithms) by using the \(pK_w\). In this case, we know that \(pK_w = 12.302\), and from Equation \ref{16.3.12}, we know that \(pK_w = pH + pOH\). Because \(pH = pOH\) in a neutral solution, we can use Equation \ref{16.3.12} directly, setting \(pH = pOH = y\). Solving to two decimal places we obtain the following:
\[pK_w = pH + pOH = y + y = 2y \nonumber\]
\[y=\dfrac{pK_w}{2}=\dfrac{12.302}{2}=6.15=pH=pOH \nonumber\]
Humans maintain an internal temperature of about 37°C. At this temperature, \(K_w = 3.55 \times 10^{−14}\). Calculate \(pK_w\) and the pH and the pOH of a neutral solution at 37°C. Report \(pH\) and \(pOH\) values to two decimal places.
- Answer
-
\(pK_w = 13.45\) and \(pH = pOH = 6.73\)
Introduction to pH: https://youtu.be/pQOa3bb5YEE
Summary
- For any neutral solution, \(pH + pOH = 14.00\) (at 25°C) and \(pH = \ce{1/2} pK_w\).
- Ion-product constant of liquid water: \[K_w = [H_3O^+][OH^−] \nonumber \]
- Definition of \(pH\): \[pH = −\log_{10}[H^+] \nonumber\] or \[[H^+] = 10^{−pH} \nonumber \]
- Definition of \(pOH\): \[pOH = −\log_{10}[OH^+] \nonumber\] or \[[OH^−] = 10^{−pOH} \nonumber \]
- Relationship among \(pH\), \(pOH\), and \(pK_w\): \[pK_w= pH + pOH \nonumber \]
Water is amphiprotic: it can act as an acid by donating a proton to a base to form the hydroxide ion, or as a base by accepting a proton from an acid to form the hydronium ion (\(H_3O^+\)). The autoionization of liquid water produces \(OH^−\) and \(H_3O^+\) ions. The equilibrium constant for this reaction is called the ion-product constant of liquid water (\(K_w\)) and is defined as \(K_w = [H_3O^+][OH^−]\). At 25°C, \(K_w\) is \(1.01 \times 10^{−14}\); hence \(pH + pOH = pK_w = 14.00\). The process is endothermic, and so the extent of ionization and the resulting concentrations of hydronium ion and hydroxide ion increase with temperature. For example, at 100 °C, the value for \(K_\ce{w}\) is approximately \(5.1 \times 10^{−13}\), roughly 100-times larger than the value at 25 °C.
Glossary
- acid ionization
- reaction involving the transfer of a proton from an acid to water, yielding hydronium ions and the conjugate base of the acid
- autoionization
- reaction between identical species yielding ionic products; for water, this reaction involves transfer of protons to yield hydronium and hydroxide ions
- ion-product constant for water (Kw)
- equilibrium constant for the autoionization of water
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).

