22.9: Carbon
- Page ID
- 21906
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)↵
The elements of group 14 show a greater range of chemical behavior than any other family in the periodic table. Three of the five elements—carbon, tin, and lead—have been known since ancient times. For example, some of the oldest known writings are Egyptian hieroglyphics written on papyrus with ink made from lampblack, a finely divided carbon soot produced by the incomplete combustion of hydrocarbons (Figure \(\PageIndex{1}\)). Activated carbon is an even more finely divided form of carbon that is produced from the thermal decomposition of organic materials, such as sawdust. Because it adsorbs many organic and sulfur-containing compounds, activated carbon is used to decolorize foods, such as sugar, and to purify gases and wastewater.

Preparation and General Properties of Carbon
Elemental carbon, for example, ranks only 17th on the list of constituents of Earth’s crust. Pure graphite is obtained by reacting coke, an amorphous form of carbon used as a reductant in the production of steel, with silica to give silicon carbide (SiC). This is then thermally decomposed at very high temperatures (2700°C) to give graphite:
\[\mathrm{SiO_2(s)}+\mathrm{3C(s)}\xrightarrow{\Delta}\mathrm{SiC(s)}+\mathrm{2CO(g)} \label{\(\PageIndex{1}\)} \]
\[\mathrm{SiC(s)}\xrightarrow{\Delta}\mathrm{Si(s)}+\mathrm{C(graphite)} \label{\(\PageIndex{2}\)} \]
One allotrope of carbon, diamond, is metastable under normal conditions, with a ΔG°f of 2.9 kJ/mol versus graphite. At pressures greater than 50,000 atm, however, the diamond structure is favored and is the most stable form of carbon. Because the structure of diamond is more compact than that of graphite, its density is significantly higher (3.51 g/cm3 versus 2.2 g/cm3). Because of its high thermal conductivity, diamond powder is used to transfer heat in electronic devices.
The most common sources of diamonds on Earth are ancient volcanic pipes that contain a rock called kimberlite, a lava that solidified rapidly from deep inside the Earth. Most kimberlite formations, however, are much newer than the diamonds they contain. In fact, the relative amounts of different carbon isotopes in diamond show that diamond is a chemical and geological “fossil” older than our solar system, which means that diamonds on Earth predate the existence of our sun. Thus diamonds were most likely created deep inside Earth from primordial grains of graphite present when Earth was formed (Figure \(\PageIndex{2}\)). Gem-quality diamonds can now be produced synthetically and have chemical, optical, and physical characteristics identical to those of the highest-grade natural diamonds.

Reactions and Compounds of Carbon
Carbon is the building block of all organic compounds, including biomolecules, fuels, pharmaceuticals, and plastics, whereas inorganic compounds of carbon include metal carbonates, which are found in substances as diverse as fertilizers and antacid tablets, halides, oxides, carbides, and carboranes. Like boron in group 13, the chemistry of carbon differs sufficiently from that of its heavier congeners to merit a separate discussion.
The structures of the allotropes of carbon—diamond, graphite, fullerenes, and nanotubes—are distinct, but they all contain simple electron-pair bonds. Although it was originally believed that fullerenes were a new form of carbon that could be prepared only in the laboratory, fullerenes have been found in certain types of meteorites. Another possible allotrope of carbon has also been detected in impact fragments of a carbon-rich meteorite; it appears to consist of long chains of carbon atoms linked by alternating single and triple bonds, (–C≡C–C≡C–)n. Carbon nanotubes (“buckytubes”) are being studied as potential building blocks for ultramicroscale detectors and molecular computers and as tethers for space stations. They are currently used in electronic devices, such as the electrically conducting tips of miniature electron guns for flat-panel displays in portable computers.
Although all the carbon tetrahalides (CX4) are known, they are generally not obtained by the direct reaction of carbon with the elemental halogens (X2) but by indirect methods such as the following reaction, where X is Cl or Br:
\[CH_{4(g)} + 4X_{2(g)} \rightarrow CX_{4(l,s)} + 4HX_{(g)} \label{\(\PageIndex{7}\)} \]
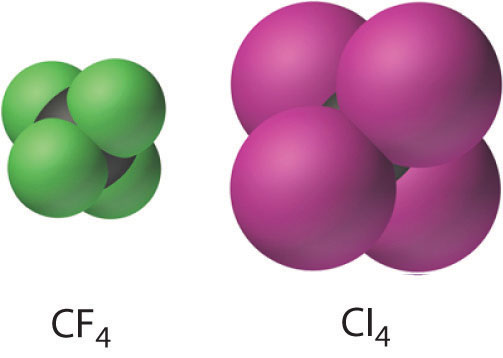
The carbon tetrahalides all have the tetrahedral geometry predicted by the valence-shell electron-pair repulsion (VSEPR) model, as shown for CCl4 and CI4. Their stability decreases rapidly as the halogen increases in size because of poor orbital overlap and increased crowding. Because the C–F bond is about 25% stronger than a C–H bond, fluorocarbons are thermally and chemically more stable than the corresponding hydrocarbons, while having a similar hydrophobic character. A polymer of tetrafluoroethylene (F2C=CF2), analogous to polyethylene, is the nonstick Teflon lining found on many cooking pans, and similar compounds are used to make fabrics stain resistant (such as Scotch-Gard) or waterproof but breathable (such as Gore-Tex).
The stability of the carbon tetrahalides decreases with increasing size of the halogen due to increasingly poor orbital overlap and crowding.
Carbon reacts with oxygen to form either CO or CO2, depending on the stoichiometry. Carbon monoxide is a colorless, odorless, and poisonous gas that reacts with the iron in hemoglobin to form an Fe–CO unit, which prevents hemoglobin from binding, transporting, and releasing oxygen in the blood (see Figure 23.26). In the laboratory, carbon monoxide can be prepared on a small scale by dehydrating formic acid with concentrated sulfuric acid:
\[\mathrm{HCO_2H(l)}\xrightarrow{\mathrm{H_2SO_4(l)}}\mathrm{CO(g)}+\mathrm{H_3O^+(aq)}+\mathrm{HSO^-_4}\label{\(\PageIndex{8}\)} \]
Carbon monoxide also reacts with the halogens to form the oxohalides (COX2). Probably the best known of these is phosgene (Cl2C=O), which is highly poisonous and was used as a chemical weapon during World War I:
\[\mathrm{CO(g)}+\mathrm{Cl_2(g)}\xrightarrow{\Delta}\textrm{Cl}_2\textrm{C=O(g)}\label{\(\PageIndex{9}\)} \]
Despite its toxicity, phosgene is an important industrial chemical that is prepared on a large scale, primarily in the manufacture of polyurethanes.
Carbon dioxide can be prepared on a small scale by reacting almost any metal carbonate or bicarbonate salt with a strong acid. As is typical of a nonmetal oxide, CO2 reacts with water to form acidic solutions containing carbonic acid (H2CO3). In contrast to its reactions with oxygen, reacting carbon with sulfur at high temperatures produces only carbon disulfide (CS2):
\[\mathrm{C(s)}+\mathrm{2S(g)}\xrightarrow{\Delta}\mathrm{CS_2(g)}\label{\(\PageIndex{1}\)0} \]
The selenium analog CSe2 is also known. Both have the linear structure predicted by the VSEPR model, and both are vile smelling (and in the case of CSe2, highly toxic), volatile liquids. The sulfur and selenium analogues of carbon monoxide, CS and CSe, are unstable because the C≡Y bonds (Y is S or Se) are much weaker than the C≡O bond due to poorer π orbital overlap.
\(\pi\) bonds between carbon and the heavier chalcogenides are weak due to poor orbital overlap.
Binary compounds of carbon with less electronegative elements are called carbides. The chemical and physical properties of carbides depend strongly on the identity of the second element, resulting in three general classes: ionic carbides, interstitial carbides, and covalent carbides. The reaction of carbon at high temperatures with electropositive metals such as those of groups 1 and 2 and aluminum produces ionic carbides, which contain discrete metal cations and carbon anions. The identity of the anions depends on the size of the second element. For example, smaller elements such as beryllium and aluminum give methides such as Be2C and Al4C3, which formally contain the C4− ion derived from methane (CH4) by losing all four H atoms as protons. In contrast, larger metals such as sodium and calcium give carbides with stoichiometries of Na2C2 and CaC2. Because these carbides contain the C2− ion, which is derived from acetylene (HC≡CH) by losing both H atoms as protons, they are more properly called acetylides. Reacting ionic carbides with dilute aqueous acid results in protonation of the anions to give the parent hydrocarbons: CH4 or C2H2. For many years, miners’ lamps used the reaction of calcium carbide with water to produce a steady supply of acetylene, which was ignited to provide a portable lantern.

The reaction of carbon with most transition metals at high temperatures produces interstitial carbides. Due to the less electropositive nature of the transition metals, these carbides contain covalent metal–carbon interactions, which result in different properties: most interstitial carbides are good conductors of electricity, have high melting points, and are among the hardest substances known. Interstitial carbides exhibit a variety of nominal compositions, and they are often nonstoichiometric compounds whose carbon content can vary over a wide range. Among the most important are tungsten carbide (WC), which is used industrially in high-speed cutting tools, and cementite (Fe3C), which is a major component of steel.
Elements with an electronegativity similar to that of carbon form covalent carbides, such as silicon carbide (SiC; Equation \(\PageIndex{1}\) ) and boron carbide (B4C). These substances are extremely hard, have high melting points, and are chemically inert. For example, silicon carbide is highly resistant to chemical attack at temperatures as high as 1600°C. Because it also maintains its strength at high temperatures, silicon carbide is used in heating elements for electric furnaces and in variable-temperature resistors.
Carbides formed from group 1 and 2 elements are ionic. Transition metals form interstitial carbides with covalent metal–carbon interactions, and covalent carbides are chemically inert.
For each reaction, explain why the given product forms.
- CO(g) + Cl2(g) → Cl2C=O(g)
- CO(g) + BF3(g) → F3B:C≡O(g)
- Sr(s) + 2C(s) \(\xrightarrow{\Delta}\) SrC2(s)
Given: balanced chemical equations
Asked for: why the given products form
Strategy:
Classify the type of reaction. Using periodic trends in atomic properties, thermodynamics, and kinetics, explain why the observed reaction products form.
Solution
- Because the carbon in CO is in an intermediate oxidation state (+2), CO can be either a reductant or an oxidant; it is also a Lewis base. The other reactant (Cl2) is an oxidant, so we expect a redox reaction to occur in which the carbon of CO is further oxidized. Because Cl2 is a two-electron oxidant and the carbon atom of CO can be oxidized by two electrons to the +4 oxidation state, the product is phosgene (Cl2C=O).
- Unlike Cl2, BF3 is not a good oxidant, even though it contains boron in its highest oxidation state (+3). Nor can BF3 behave like a reductant. Like any other species with only six valence electrons, however, it is certainly a Lewis acid. Hence an acid–base reaction is the most likely alternative, especially because we know that CO can use the lone pair of electrons on carbon to act as a Lewis base. The most probable reaction is therefore the formation of a Lewis acid–base adduct.
- Typically, both reactants behave like reductants. Unless one of them can also behave like an oxidant, no reaction will occur. We know that Sr is an active metal because it lies far to the left in the periodic table and that it is more electropositive than carbon. Carbon is a nonmetal with a significantly higher electronegativity; it is therefore more likely to accept electrons in a redox reaction. We conclude, therefore, that Sr will be oxidized, and C will be reduced. Carbon forms ionic carbides with active metals, so the reaction will produce a species formally containing either C4− or C22−. Those that contain C4− usually involve small, highly charged metal ions, so Sr2+ will produce the acetylide (SrC2) instead.
Predict the products of the reactions and write a balanced chemical equation for each reaction.
- C(s) + excess O2(g) \(\xrightarrow{\Delta}\)
- C(s) + H2O(l) →
- NaHCO3(s) + H2SO4(aq) →
Answer
- C(s) + excess O2(g) \(\xrightarrow{\Delta}\) CO2(g)
- C(s) + H2O(l) → no reaction
- NaHCO3(s) + H2SO4(aq) → CO2(g) + NaHSO4(aq) + H2O(l)
Summary
The stability of the carbon tetrahalides decreases as the halogen increases in size because of poor orbital overlap and steric crowding. Carbon forms three kinds of carbides with less electronegative elements: ionic carbides, which contain metal cations and C4− (methide) or C22− (acetylide) anions; interstitial carbides, which are characterized by covalent metal–carbon interactions and are among the hardest substances known; and covalent carbides, which have three-dimensional covalent network structures that make them extremely hard, high melting, and chemically inert.
Contributors and Attributions
- Anonymous

