Strengths of Covalent Bonds
- Page ID
- 53424
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Skills to Develop
- Describe the significance of bond dissociation energy
Bond dissociation energy or bond energy is the energy required to break a bond. Usually we mean bond dissociation enthalpy, the enthalpy change for this reaction:
\[A—B \rightarrow A(g) + B(g)\]
For example, H—Cl → H• (g) + Cl• (g)
The • represents an unpaired electron, called a radical. When measuring the bond enthalpy, we break the bond so that half the bonding electrons go to each atom. Breaking a bond is always an endothermic process, so the bond energy is always positive. We use the symbol D(A—B) to represent the bond enthalpy of an A—B bond.
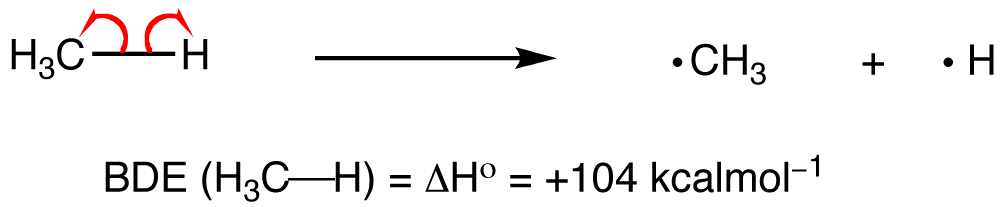
Bond energy is similar, but is the average energy required to break all the bonds in a molecule. For instance, C-H bond energy is the enthalpy of atomization of methane divided by 4. This is different from the energy required to break the first C-H bond in CH4 because breaking the second bond might be easier or harder than breaking the first.
Some Approximate Average Bond Enthalpies (kcal/mol)
| Bond | D (kcal/mol) | Bond | D (kcal/mol) |
|---|---|---|---|
| H-H | 103 | H-F | 135 |
| H-Cl | 102 | H-Br | 87 |
| H-I | 70 | H-C | 98 |
| H-O | 110 | H-N | 92 |
| C-C | 83 | C-F | 116 |
| C-Cl | 78 | C-Br | 68 |
| C-O | 86 | C-N | 73 |
| N-N | 40 | O-O | 34 |
| C=C | 144 | C≡C | 200 |
| C=O | 191 | C=N | 147 |
| O=O | 118 | N≡N | 225 |
| Cl-O | 52 | Cl-N | 72 |
| N-O | 48 | N=O | 145 |
How do we Measure Bond Dissociation Energy?
As you might guess from the discussion above about bond energy vs. bond dissociation energy, it's actually quite complicated to measure these! There are a lot of complications to consider, and you can't always measure directly the heat of reaction that you want.
Why does Bond Dissociation Energy Matter?
Having a general knowledge of bond strengths lets us understand the structures of molecules and predict reactions. For instance, when we guess the arrangement of atoms to make a Lewis structure, we will probably do better if we avoid making weak bonds. N-N and O-O single bonds are pretty weak, so usually we try not to put them in Lewis structures, unless nothing else makes sense. The structure will be better if we can make a multiple bond or put some other atom in between.
We can also use bond strength knowledge to predict what direction reactions will go and how fast they will go. Recall that we can calculate the enthalpy of a reaction by comparing the atomization energies of the reactants and products. (This is the same idea as when we calculated enthalpies of reaction using standard enthalpies of formation.) If the product has stronger bonds than the reactants, the reaction is likely to be exothermic, and exothermic reactions often go forward. If the reactant has strong bonds, you might need to use a lot of energy to get the reaction started, even if it is exothermic. Combustion reactions are an example: C-C and C-H bonds are pretty strong, so we need a spark or a match to start the burning. Another example is "nitrogen fixation": we need N to make proteins and other important molecules in our bodies, but it is very hard to get N from N2 because of the strong triple bond. This is why we have to use fertilizer in agriculture, and making nitrogen fertilizer uses roughly 1% of the world's energy. Another example is teflon or PTFE, which makes a very slippery and non-reactive coating that is used in non-stick pans, containers for reactive chemicals, etc. PTFE has only C-C and C-F bonds, both of which are very strong, so it doesn't easily react even at high temperature when you cook with it. In contrast, if a reactant has some very weak bonds, it might be very easy to start a reaction. For instance, hydrogen peroxide (HOOH) can be used as a bleach or disinfectant because the weak O-O single bond can easily break, helping it oxidize whatever is nearby. Hydrogen peroxide and hydrazine (N2H4) can both be used as rocket fuel because they react easily and exothermically (they have weak bonds, and they form products with stronger bonds).
Contributors and Attributions
Emily V Eames (City College of San Francisco)

