22.4: Beneficiation
- Page ID
- 49648
Beneficiation is any process which removes the gangue minerals from ore to produce a higher grade product, and a waste stream. Beneficiation may involve physical or chemical processes. Often, as in the case of panning for gold, the desired ore or metal is denser than the gangue. The latter can be suspended in a stream of water and flushed away. The iron ore magnetite, Fe3O4, is ferrimagnetic. It can be separated from abundant deposits of taconite by grinding to a fine slurry in water. Passing this suspension over powerful electromagnets removes the Fe3O4, more than doubling the concentration of Fe. Both the physical beneficiation processes just described produce large quantities of tailings-water suspensions of the gangue. Usually the silicate and other particles are trapped in a settling basin, but in one case large quantities of taconite tailings have been dumped into Lake Superior for several decades.
Flotation
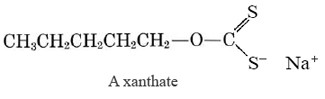
This is a partly physical and partly chemical beneficiation used to concentrate ores of copper, lead, and zinc. These metals have considerable affinity for sulfur. (the table on the metallurgy section lists sulfide ores for each of them.) The ore is ground and suspended with water in large tanks. Flotation agents such as the xanthates are added and a stream of air bubbles passed up through the tank. The sulfur-containing end of the xanthate molecule is attracted to the desired metal, while the hydrocarbon end tends to avoid water (like the hydrocarbon end of a soap molecule). The metal-containing particles are swept to the top of the tank in a froth much akin to soap bubbles. The concentrated ore may then be skimmed from the water surface. By means of flotation, ores containing as little as 0.3% copper can be concentrated to 20 to 30% copper, making recovery of the metal economically feasible.
Chemical Processes
Many ores can be chemically separated from their gangue by means of acid or base. Thus copper ore is often leached with acid which dissolves copper oxides and carbonates and leaves behind most silicates:
\[\text{CuCo}_3 + \text{H}_2\text{SO}_4(aq) \rightarrow \text{CuSO}_4 (aq) + \text{H}_2\text{O} + \text{CO}_2 \label{1} \]
In the Bayer process bauxite is dissolved in 30% NaOH:
\[\text{Al}_2\text{O}_3(s) + \text{OH}^-(aq) + \text{3H}_2\text{O}(l) \rightarrow \text{2Al(OH}^-_4(aq) \label{2} \]
and then reprecipitated by adding acid. Impurities such as Fe2O3 and SiO2 are eliminated in this way.
Many sulfide and carbonate ores are roasted in air in order to convert them into oxides, a form more suitable for further processing:
\[\text{2ZnS}(s) + \text{3O}_2(g) \rightarrow \text{2ZnO}(s) + \text{SO}_2(g)\label{3} \]
\[\text{PbCO}_3 (s) \rightarrow \text{PbO}(s) + \text{Co}_2(g) \nonumber \]
The production of SO2 in Equation \(\ref{3}\) is a notorious source of air pollution, including acid rain.


