Photochemical Changes in Opsin
- Page ID
- 477
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Photochemical events in vision involve the protein opsin and the cis/trans isomers of retinal. Opsin does not absorb visible light, but when it is bonded with 11-cis-retinal to form rhodopsin, which has a very broad absorption band in the visible region of the spectrum. The peak of the absorption is around 500 nm, which matches the output of the sun closely.
Upon absorption of a photon of light in the visible range, cis-retinal can isomerize to all-trans-retinal. The shape of the molecule changes as a result of this isomerization. The molecule changes from an overall bent structure to one that is more or less linear. All of this is the result of trigonal planar bonding (120 o bond angles) about the double bonds.
Protein Geometry Changes Following Retinal Isomerization
As we shall see below, the isomerization of retinal has an important effect on special proteins in the rod cell: the isomerization event actually causes the proteins to change their shape. This shape change ultimately leads to the generation of a nerve impulse. Hence, the next step in understanding the vision process for monochrome vision is to describe these proteins, and how they change their shape after retinal isomerizes.
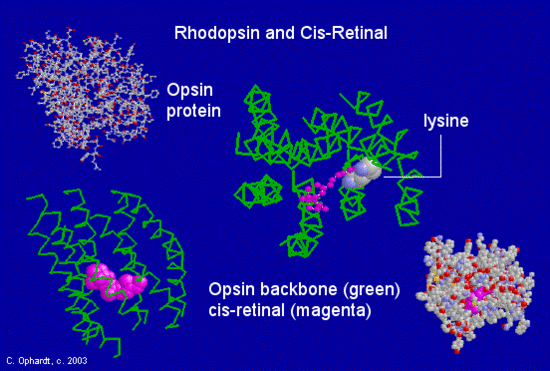
Opsin consists of 348 amino acids, covalently linked together to form a single chain. This chain has seven hydrophobic, or water-repelling, alpha-helical regions that pass through the lipid membrane of the pigment-containing discs. This region consists primarily of nonpolar amino acids, which do not attract the polar water molecule. The cis-retinal is situated among these alpha helixes in the hydrophobic region. It is covalently linked to Lysine 296, one of the amino acids in the opsin peptide chain. The linkage is as a Schiff base reaction.
When the cis-retinal absorbs a photon it isomerizes to the all-trans configuration without (at first) any accompanying change in the structure of the protein. Rhodopsin containing the all-trans isomer of retinal is known as bathorhodopsin. However, the trans isomer does not fit well into the protein, due to its rigid, elongated shape. While it is contained in the protein, the all-trans-retinal adopts a twisted conformation, which is energetically unfavorable. The molecule undergoes a series of shape changes to try and better fit the binding site. Therefore, a series of changes in the protein occurs to expel the trans-retinal from the protein.
These rapid movements of the retinal are transferred to the protein, and from there into the lipid membrane and nerve cells to which it is attached. This generates nerve impulses which travel along the optic nerve to the brain, and we perceive them as visual signals - sight. The free all-trans-retinal is then converted back into the cis form by a series of enzyme-catalyzed reactions, whereupon is reattaches to another opsin ready for the next photon to begin the process again.
Activated rhodopsin causes electrical impulses in the following way:
- The cell membrane (outer layer) of a rod cell has an electric charge. When light activates rhodopsin, it causes a reduction in cyclic GMP, which causes this electric charge to increase. This produces an electric current along the cell. When more light is detected, more rhodopsin is activated and more electric current is produced.
- This electric impulse eventually reaches a ganglion cell, and then the optic nerve.
- The nerves reach the optic chasm, where the nerve fibers from the inside half of each retina cross to the other side of the brain, but the nerve fibers from the outside half of the retina stay on the same side of the brain.
- These fibers eventually reach the back of the brain (occipital lobe). This is where vision s interpreted and is called the primary visual cortex. Some of the visual fibers go to other parts of the brain to help to control eye movements, response of the pupils and iris, and behavior.
Outside Links
- Rhodopsin changes - NEUROBIOLOGY, Molecules, Cells and Systems, Gary G. Matthews
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College; Virtual Chembook

