Chemistry of Vision
- Page ID
- 475
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Vision is such an everyday occurrence that we seldom stop to think and wonder how we are able to see the objects that surround us. Yet the vision process is a fascinating example of how light can produce molecular changes. The retina contain the molecules that undergo a chemical change upon absorbing light, but it is the brain that actually makes sense of the visual information to create an image.
Introduction
Light is one of the most important resources for civilization, it provides energy as it pass along by the sun. Light influence our everyday live. Living organisms sense light from the environment by photoreceptors. Light, as waves carry energy, contains energy by different wavelength. In vision, light is the stimulus input. Light energy goes into eyes stimulate photoreceptor in eyes. However, as an energy wave, energy is passed on through light at different wavelength.
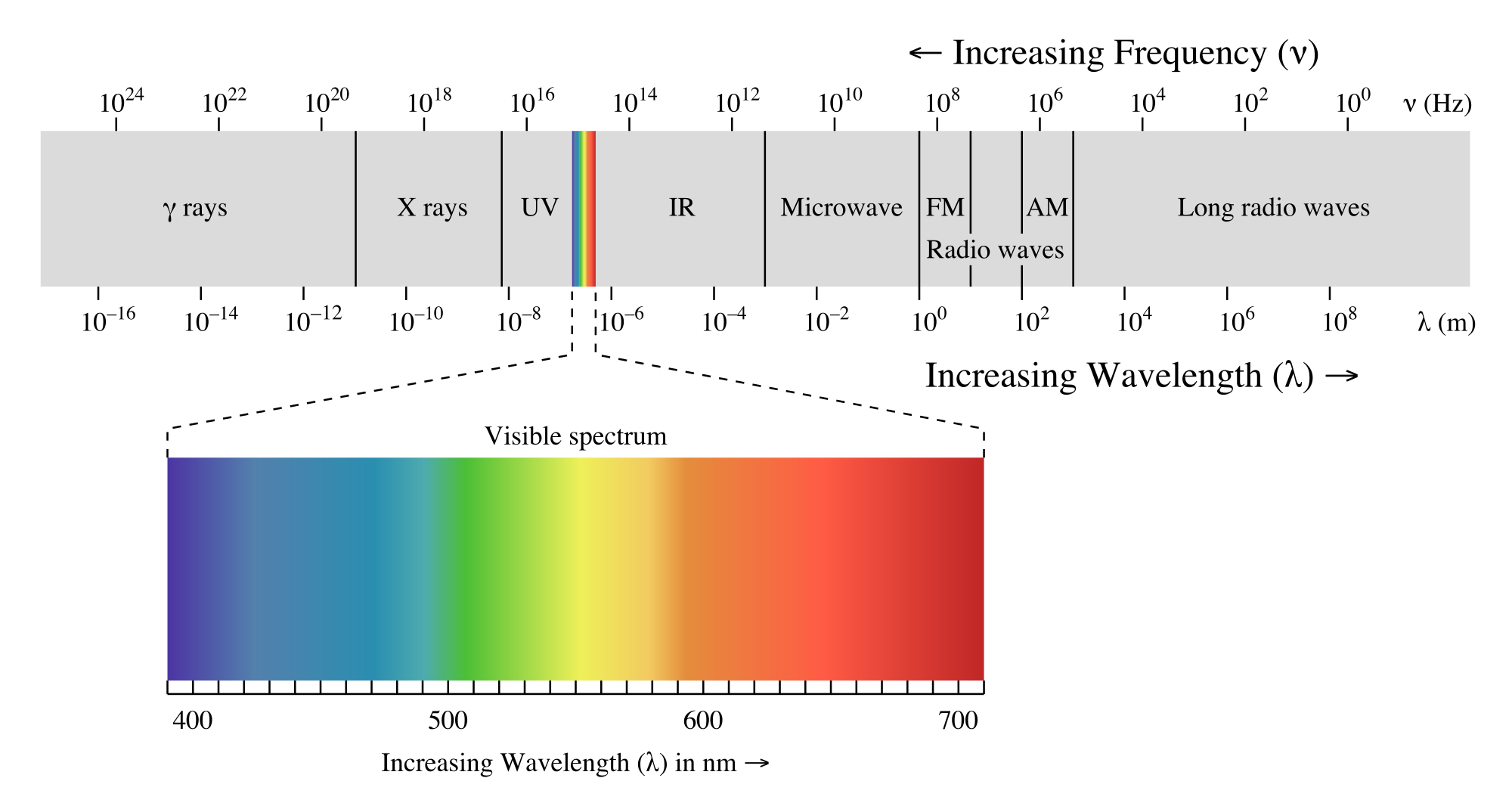
Light, as waves carry energy, contains energy by different wavelength. From long wavelength to short wavelength, energy increase. 400 nm to 700 nm is visible spectrum.
Light energy can convert chemical to other forms. Vitamin A, also known as retinol, anti-dry eye vitamins, is a required nutrition for human health. The predecessor of vitamin A is present in the variety of plant carotene. Vitamin A is critical for vision because it is needed by the retina of eye. Retinol can be convert to retinal, and retinal is a chemical necessary for rhodopsin. As light enters the eye, the 11-cis-retinal is isomerized to the all-"trans" form.
Mechanism of Vision
The molecule cis-retinal can absorb light at a specific wavelength. When visible light hits the cis-retinal, the cis-retinal undergoes an isomerization, or change in molecular arrangement, to all-trans-retinal. The new form of trans-retinal does not fit as well into the protein, and so a series of geometry changes in the protein begins. The resulting complex is referred to a bathrhodopsin (there are other intermediates in this process, but we'll ignore them for now).
The reaction above shows Lysine side-chain from the opsin react with 11-cis-retinal when stimulated. By removing the oxygen atom from the retinal and two hydrogen atom from the free amino group of the lysine, the linkage show on the picture above is formed, and it is called Schiff base.
Signal Transduction Pathway
As the protein changes its geometry, it initiates a cascade of biochemical reactions that results in changes in charge so that a large potential difference builds up across the plasma membrane. This potential difference is passed along to an adjoining nerve cell as an electrical impulse. The nerve cell carries this impulse to the brain, where the visual information is interpreted.
The light image is mapped on the surface of the retina by activating a series of light-sensitive cells known as rods and cones or photoreceptors. The rods and cones convert the light into electrical impulses which are transmitted to the brain via nerve fibers. The brain then determines, which nerve fibers carried the electrical impulse activate by light at certain photoreceptors, and then creates an image.
The retina is lined with many millions of photoreceptor cells that consist of two types: 7 million cones provide color information and sharpness of images, and 120 million rods are extremely sensitive detectors of white light to provide night vision. The tops of the rods and cones contain a region filled with membrane-bound discs, which contain the molecule cis-retinal bound to a protein called opsin. The resulting complex is called rhodopsin or "visual purple".
In human eyes, rod and cones react to light stimulation, and a series of chemical reactions happen in cells. These cells receive light, and pass on signals to other receiver cells. This chain of process is class signal transduction pathway. Signal transduction pathway is a mechanism that describe the ways cells react and response to stimulation.
References
- Biochemistry, L. Stryer (W.H. Freeman and Co, San Francisco, 1975).
- The Cambridge Guide to the Material World, Rodney Cotterill (Cambridge University Press, Cambridge, 1985)
Contributors
- {{template.ContribOphardt()}}

