Reference Electrodes
- Page ID
- 78002
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)It should be clear by now that at least two electrodes are necessary to make a potential measurement. As Kissinger and Bott have so perfectly expressed, “electrochemistry with a single electrode is like the sound of one hand clapping” (http://currentseparations.com/issues/20-2/20-2d.pdf). In potentiometry, those two electrodes are generally called the indicator electrode and the reference electrode. The indicator electrode possesses some characteristic that allows it to selectively respond to changes in the activity of the analyte being measured. For the measured potential to have meaning in this context, the reference electrode must be constructed so that its composition is fixed and its response is stable over time, with observed changes in measured potential due solely to changes in analyte concentration.
You are probably familiar with tables of standard reduction potentials from a general chemistry course. The standard reduction potential, or E0, allows you to predict the ease with which a half-cell reaction occurs relative to other half-reactions. (For a review of standard potentials and electrochemical cells, you can access the companion “Concepts” module). Values of E0 are most often reported as the potential measured in an electrochemical cell for which the standard hydrogen electrode is used as a reference.
The standard hydrogen electrode, or SHE, is composed of an inert solid like platinum on which hydrogen gas is adsorbed, immersed in a solution containing hydrogen ions at unit activity. The half-cell reaction for the SHE is given by
\[\ce{2H+ (aq) + 2 e- \rightleftharpoons H2 (g)}\]
and the half-cell potential arbitrarily assigned a value of zero (E0 = 0.000 V).
Practical application of the SHE is limited by the difficulty in preparing and maintaining the electrode, primarily due to the requirement for H2 (g) in the half-cell. Most potentiometric methods employ one of two other common reference half-cells – the saturated calomel electrode (SCE) or the silver-silver chloride electrode (Ag/AgCl).
1. Saturated Calomel Electrode (SCE)
The SCE is a half cell composed of mercurous chloride (Hg2Cl2, calomel) in contact with a mercury pool. These components are either layered under a saturated solution of potassium chloride (KCl) or within a fritted compartment surrounded by the saturated KCl solution (called a double-junction arrangement). A platinum wire is generally used to allow contact to the external circuit. The half reaction is described by
\[\ce{Hg2Cl2 (s) + 2 e- \rightleftharpoons 2 Hg} (l ) + \ce{2 Cl-} \textrm{(sat’d)}\]
with an E0 value of +0.244 V. A common arrangement for the SCE is shown below, left side. In this arrangement, a paste is prepared of the calomel and solution that is saturated with KCl.
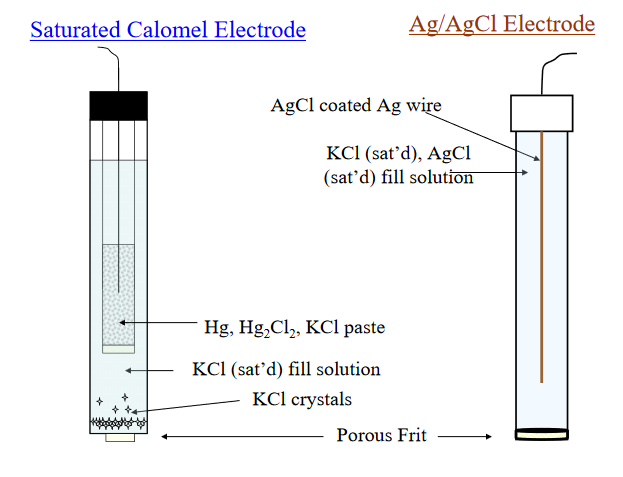
The solution over the paste is also saturated with KCl, with some solid KCl crystals present. Contact to the measurement cell is made through a porous glass frit or fiber which allows the movement of ions, but not the bulk solution. In many electrodes designed for potentiometry, the reference half cell is contained within the body of the sensing electrode. This arrangement is referred to as a “combination” electrode.
2. Silver/Silver Chloride (Ag/AgCl)
The silver/silver chloride reference electrode is composed of a silver wire, sometimes coated with a layer of solid silver chloride, immersed in a solution that is saturated with potassium chloride and silver chloride. The pertinent half reaction is
\[\ce{AgCl (s) + e- ⇔ Ag (s) + Cl-} \textrm{(sat’d)}\]
with a value for E0 of +0.222 V. The actual potential of the half-cell prepared in this way is +0.197 V vs SHE, which arises because in addition to KCl, AgCl also contributes to the chloride activity, which is not exactly unity. A schematic of the Ag/AgCl reference electrode is shown at right in the previous figure.
Both the SCE and the Ag/AgCl reference electrodes offer stable half-cell potentials that do not change over time or with temperature. In addition, the loss of electrolyte to evaporation does not change the saturated nature of the solution, nor the potential. One must be aware that the contact junctions of the half cells by nature slowly leak fill solution into the external solution in which they are found. As such, there are instances where measurements of certain ions, like chloride, might be affected by the ions introduced to the measurement solution by leakage. The doublejunction design prevents this problem by placing a second solution between the reference half cell and the measurement solution. This of course adds a second junction potential, a topic covered elsewhere in this module.


