Metals Analysis by X-ray Fluorescence
- Page ID
- 142686
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)X-ray Fluorescence Spectroscopy
At the end of this assignment students will be able to:
- Describe the process of XRF and explain how element specific information can be obtained using this technique.
- Differentiate between qualitative and quantitative analysis using XRF.
- Compare and contrast the benefits and challenges of quantitative analysis using XRF.
- Apply the knowledge gained to determine the role of XRF in the analysis of heavy metals in
Lake Nakuru
Purpose
The purpose of this unit is to introduce the fundamental principles of x-ray fluorescence spectroscopy (XRF), apply this method to the analysis of simulated data for heavy metals in suspended particulate and sediment samples from
Recommended reading prior to completion of this exercise:
- Chapter 10 on spectroscopic techniques in the eText Analytical Chemistry 2.0 by David Harvey found in the ASDL collection.1 http://www.asdlib.org/onlineArticles/ecourseware/Analytical%20Chemistry%202.0/Text_Files.html
- The JASDL Article "Introduction to Energy-Dispersive X-ray Fluorescence (XRF) – an Analytical Chemistry Perspective" by Pete Palmer.2 Note: you may find it convenient to download this material as a PowerPoint presentation. http://www.asdlib.org/onlineArticles/ecourseware/Palmer/ASDL%20Intro%20to%20XRF.pdf
Introduction
What analytical chemist hasn't longed for a Star Trek tricorder? In addition to the medical and engineering tricorders, a variety of other tricorders were "used" on the original TV series for geological (GEO) and metallurgical (MET) and Biological (BIO) measurements. Landing on a new planet, the tricorder gave the Star Trek crew the ability to rapidly scan the elemental composition of surrounding rocks, evaluate alloys of structures and survey animal and plant life. To those of us burdened by collection of representative samples that must be transported back to the laboratory for laborious processing and sample preparation steps, and finally an instrumental analysis – the ability to get instantaneous elemental composition was as fantastic as transporters, phasers and Klingon cloaking devices. With recent developments of handheld XRF devices this fantasy has become a scientific reality. In this module we will examine the scientific principles that underlie the method of XRF, examine its advantages and limitations and apply this method to the analysis of sediment samples from
X-ray fluorescence is a luminescence-based field-portable method that can provide rapid elemental analyses, relatively inexpensively. The battery operated devices shown below operate essentially in a point and acquire mode that allows measurements to be made outside the laboratory, for example determining whether heavy metals are present in toys or other consumer products, or sorting scrap metal alloys for recycling.4 The non-destructive nature of this method makes it ideal for the compositional analysis of priceless art and antiquities.

XRF instruments are also available for use in a more standard laboratory spectrometer format in which the sample is inserted into the instrument. These benchtop instruments can be more sensitive and accurate, but generally are also more expensive.
How can x-rays generate fluorescence emission? In molecular fluorescence measurements, UV-visible light is used to excite valence electrons into an excited state.1 Fluorescence occurs when these excited state electrons relax back to the ground state emitting photons. Compared with the light used for excitation, the wavelength corresponding to the maximum fluorescence intensity is red shifted (i.e. at a longer wavelength and lower energy) because the excited electrons relax to the ground vibrational and rotational states of the excited electronic state before emitting a photon.1 Atomic fluorescence is a similar phenomenon, except that the electrons are excited thermally in a flame or plasma and fluorescence results when these excited electrons relax.
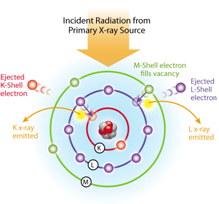
X-ray fluorescence occurs by an analogous but subtly different process illustrated in Figure 2. X-ray radiation is much higher energy than UV-visible light. X-rays are referred to as ionizing radiation because the energy of an x-ray photon can provide enough energy to eject an electron from an inner shell atomic orbital creating a positive ion. As shown in Figure 2, higher energy outer shell electrons fill the vacancies in the lower energy orbitals created by electron ejection. The excess energy of the electron that fills the vacancy is emitted as a secondary x-ray photon, generating the fluorescence signal. As in molecular fluorescence, the photons emitted by fluorescence are lower in energy than the source radiation. Also, because the energy (and hence wavelength) of the fluorescence depends on differences in energy of the inner shell atomic orbitals that do not participate in molecular bonds, it is characteristic of the elemental composition of the sample independent of its chemical form.
The material provided in the tutorial "Introduction to Energy-Dispersive X-ray Fluorescence (XRF) – an Analytical Chemistry Perspective" (reference 2) will likely be helpful in answering the questions below.
Q1. Figure 3 shows a periodic table with the relevant energies (in electron Volts; eV) for the X-ray emission of various elements. What do the letters K, L and M represent?

The characteristic x-rays listed for each element in Figure 3 are labeled as K, L, M or N to denote the shells they originated from, with x-rays originating from the K-shell having the highest energy. Another designation: \(\alpha\), \(\beta\) or \(\gamma\), is used to indicate x-rays that originated from the original shell of the transition electrons that fill the vacancy created by the ejected electron. For example, a K\(\alpha\) x-ray is produced from a transition of an electron from the L to the K shell, while a K\(\beta\) x-ray results from the transition of an electron from the M to a K shell, etc. Since within the shells there are multiple orbits of higher and lower energy electrons, a further designation is made as \(\alpha\)1, \(\alpha\)2 or \(\beta\)1, \(\beta\)2, etc. to denote transitions of electrons from these orbits into the same lower shell.
Q2. For lead, atomic absorption measurements are typically made at a wavelength of 283.3 nm. Calculate the energy in joules of a 283.3 nm photon and the L\(\alpha\) and M\(\alpha\) photons listed for lead in the Periodic Table in Figure 3. Note that the values in Figure 4 are given in keV. Offer an explanation for the relative order of the energies you calculated.
Q3. Examine the values in the periodic table in Figure 3. What are the trends in energy as you move along a row of the periodic table? What about moving down a column? Can you explain these trends in terms of differences in atomic structure? How might these differences in energy be useful in the analysis of the amounts of different elements in a complex sample?
XRF measures the energy and intensity of secondary x-rays produced, as illustrated in Figure 2. The emitted x-rays are called "secondary" because they are produced as a result of irradiation from a higher energy "primary" source. Backscattering of the x-ray source radiation also occurs and is a source of interference that spectrometers try to reduce or remove, for example using filters or polarization methods. Backscattered radiation does not interact with the sample and has the same wavelength as the source radiation but is scattered from the sample in all directions. In some cases, backscattered x-rays can be used to help normalize the data and compensate for self-absorption and differences in sample density.
The power of XRF for elemental analysis is in its ability to report on the presence of a wide range of elements, its ease of use (little sample preparation is required) and the non-destructive nature of the measurement. The results of XRF can be quantitative, however to match the accuracy and precision of alternative methods like atomic absorption spectroscopy, special care is required in the preparation and analysis of the sample.2 An example spectrum for an energy-dispersive (explained below) x-ray fluorescence (EDS-XRF) measurement is shown in Figure 4.

Q4. Locate absorption bands in Figure 4 that might be attributed to the Cr and Zn K\(\alpha\) transitions listed in Figure 3.
The EDS spectrum in Figure 4 was produced using a solid-state silicon-based detector with a digital processor that can rapidly analyze the electronic pulses caused by the incident x-rays. The detector monitors the energy and number of the x-ray photons over a defined integration time window. The signal is processed using a multichannel analyzer that accumulates the electrical signals to produce a digital spectrum. As illustrated in Figure 4, the energy in eV of the fluorescent x-rays reflects the elemental composition of the sample. The photon emission rate in terms of the number of counts per second (cps) at a given voltage is proportional to the concentration of the element producing a particular signal.
XRF measurements made with field-portable devices are most often used for qualitative analysis. The presence of a metal of concern (e.g., lead in children's toys or house paint) can be easily identified. The non-invasive nature of XRF analyses makes it an excellent screening tool for field measurement as illustrated by this short video http://www.youtube.com/watch?v=mPrN8-9t-DI. Quantitative analyses can be performed using XRF provided that care is taken in calibration and efforts are made to overcome matrix effects.2 As demonstrated in several examples in reference 2, calibration plots for XRF can compare favorably with those produced by atomic spectroscopy, however such careful quantitative experiments are typically performed in the laboratory using standard additions and comparison with a standard reference material (SRM) to validate the measurement. Although the method of standard additions typically provides more accurate quantitative results, when this method is used we can no longer consider XRF to be a nondestructive analysis method because the sample is irreparably altered by addition of the standard.
EPA method 6200 describes the use of field-portable XRF (FPXRF) for screening for 26 analytes including Cr, Cu, Pb and Zn, in soil and sediment samples. To quote section 1.2 of the method:
"This method is a screening method to be used with confirmatory analysis using other techniques (e.g., flame atomic absorption spectrometry (FLAA), graphite furnace atomic absorption spectrometry (GFAA), inductively coupled plasma-atomic emission spectrometry, (ICP-AES), or inductively coupled plasma-mass spectrometry, (ICP-MS)). This method’s main strength is that it is a rapid field screening procedure. The method's lower limits of detection are typically above the toxicity characteristic regulatory level for most RCRA analytes. However, when the obtainable values for precision, accuracy, and laboratory-established sensitivity of this method meet project-specific data quality objectives (DQOs), FPXRF is a fast, powerful, cost effective technology for site characterization."
Table 1 of EPA Method 6200 lists "Interference Free Lower Limits of Detection" in silica sand for a variety of metals; the values relevant to this study are listed below.
Table 1. Interference Free Lower Limits of Detection (LLOD) in Silica Sand Taken from EPA Method 6200, pg. 25.
|
Analyte |
LLOD (mg/kg) |
|---|---|
|
Chromium (Cr) |
150 |
|
Copper (Cu) |
50 |
|
Lead (Pb) |
20 |
|
Zinc (Zn) |
50 |
Table 2 lists the levels for chromium, copper, lead, and zinc reported in
Table 2. Metal Concentrations in Lake Nakuru Sediments and Suspended Solids found in Nelson Study8
| Concentration in dry sediments (\(\mu\)g/g) | Concentration in suspended solids (\(\mu\)g/g) | |||
|---|---|---|---|---|
| Trace metal | Average (11 sites) | Range | Average | Standard deviation |
| Chromium | 67 | 10 – 280 | 8.3 | 0.82 |
| Copper | 24 | 5 – 95 | 19 | 2.9 |
| Lead | 22 | 4 – 100 | 11.7 | 0.9 |
| Zinc | 147 | 44 – 630 | 74 | 31 |
Q5. Compare the levels of chromium, copper, lead and zinc for the sediment samples in Table 2 to the limits of detection taken in Table 1 from EPA Method 6200. Do you think that XRF is a suitable method for detecting the presence each of the metals listed in
Q6. Similarly examine the values for suspended solids reported in Table 2 and compare these to the LLOD values in Table 1. Do you think that XRF would be a suitable method for detecting the presence of each of the metals in suspended solid samples from
Q7. Suppose you wanted to use XRF to quantify the levels of these metals in
Q8. The values in Table 1 are based on silica sand which has a fairly uniform size and composition. Would you expect that the samples from
Q9. A common format involving the use of XRF in analyses is to use it to screen samples in the field to select those above a certain value for collection and subsequent analysis in the laboratory using a method like atomic spectroscopy. Describe how you might incorporate XRF into a plan for sampling the sediment samples at
References
- "Spectroscopy", Chapter 10, Analytical Chemistry 2.0, David Harvey http://www.asdlib.org/onlineArticles/ecourseware/Analytical%20Chemistry%202.0/Text_Files.html
- "Introduction to Energy-Dispersive X-ray Fluorescence (XRF) – an Analytical Chemistry Perspective" by Pete Palmer. http://www.asdlib.org/onlineArticles/ecourseware/Palmer/ASDL%20Intro%20to%20XRF.pdf
- "Star Trek Next Generation Technical Manual" www.scribd.com/doc/17602666/S...-Pages#archive, accessed 6/25/2012.
- "Handheld XRF Spectrometers" www.bruker-axs.com/handheldx-...ctrometry.html, accessed 6/25/2012.
- "How XRF Works" www.tawadascientific.com/how-xrf-works.php, accessed 6/25/2012.
- "Periodic Table of the Elements and X-Ray Energies" www.bruker-axs.com/uploads/tx...y_Energies.pdf, accessed 6/26/2012.
- " EPA Method 6200 - Field Portable X-ray Fluorescence Spectrometry for the Determination of Elemental Concentrations in Soil and Sediment" http://www.epa.gov/osw/hazard/testmethods/sw846/pdfs/6200.pdf, accessed 6/28/2012.
- Nelson, Y.M.; Thampy, R.J.; Motelin, G.K.; Raini, J.A.; DiSante, C.J.; and Lion, L.W. “Model for Trace Metal Exposure in Filter-Feeding Flamingos at Alkaline Rift Valley Lake, Kenya” Environmental Toxicology and Chemistry, 1998, 17, 2302 – 2309.


